SEPARATE CHEMISTRY P2: ORGANIC CHEMISTRY


A GCSE Organic Chemistry revision page covering drawing alkanes, alkenes and alcohols, testing with bromine water, combustion of hydrocarbons, alcohol dehydration, combustion practicals, polymers and monomers, carboxylic acids and ester formation with examples and explanations.
CH245: Draw the structure for any alkane
AlkAnes have the general formula of CnH2n+2.
They are saturated hydrocarbons – meaning they are made up of hydrogen and carbon only and all the carbon-carbon bonds are single bonds.
To draw alkanes:
- Step 1: Draw your number of carbons (monkeys eat plastic bananas!).
- Step 2: Make sure all carbons are attached to each other with single bonds.
- Step 3: Each carbon can form 4 bonds. Make sure each carbon has 4 lines coming out of them (including the bonds already formed).
- Step 4: They are hydrocarbons, so each incomplete bond should have a hydrogen attached to it:
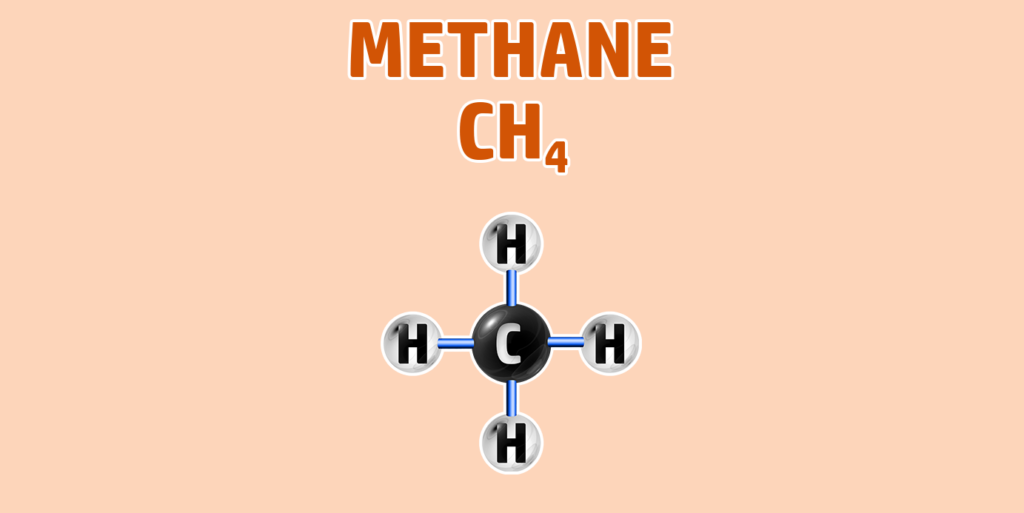
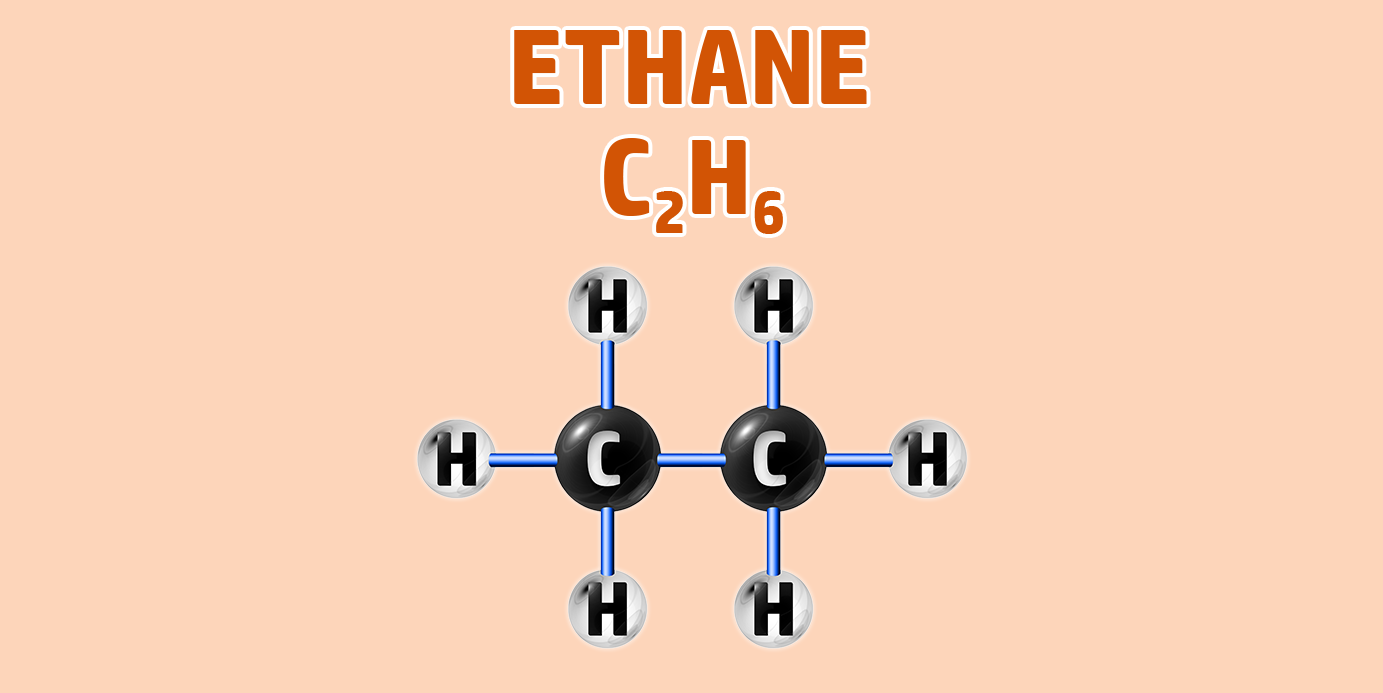
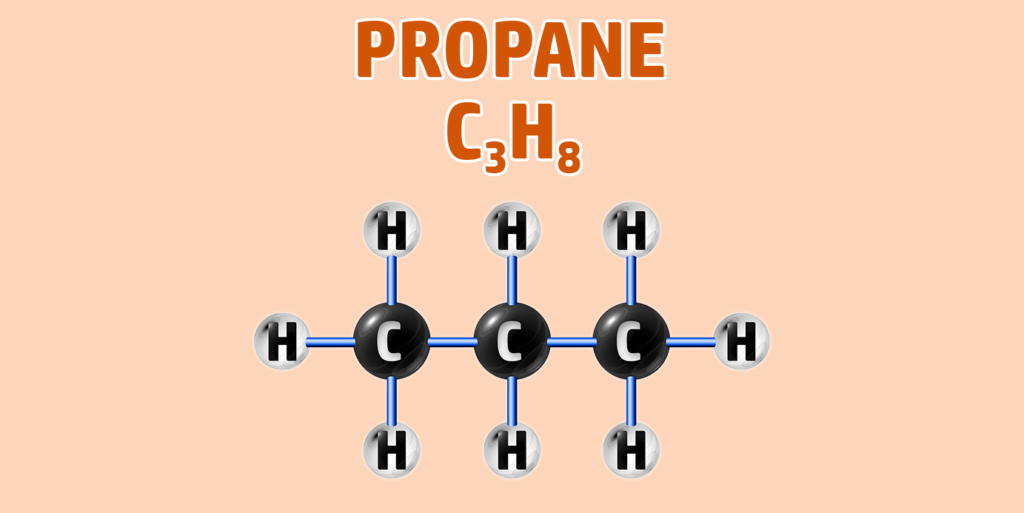
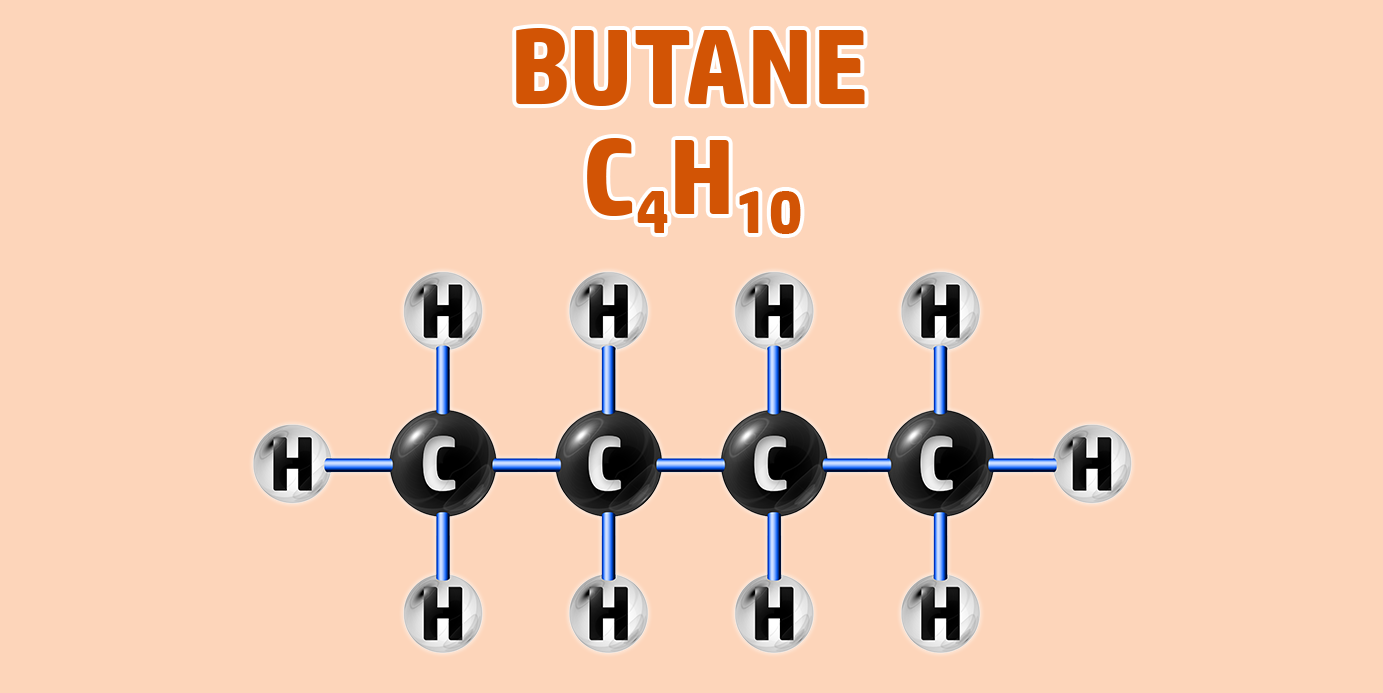
If you are trying to remember the order of the first 4 alkanes, remember Monkey Eat Plastic Bananas!
CH246: Draw the structure for any alkene
Alkenes have the general formula of CnH2n.
They are unsaturated hydrocarbons – meaning they are made up of hydrogen and carbon only and they contain at least one double bond.
The double bond is their functional group.
To draw alkenes:
- Step 1: Draw your number of carbons (monkeys eat plastic bananas!).
- Step 2: Add a double bond between the first 2 carbons. The rest are all single bonds.
- Step 3: Each carbon can form 4 bonds. Make sure each carbon has 4 lines coming out of them (including the bonds already formed).
- Step 4: Alkenes are hydrocarbons, so each incomplete bond should have a hydrogen attached to it:
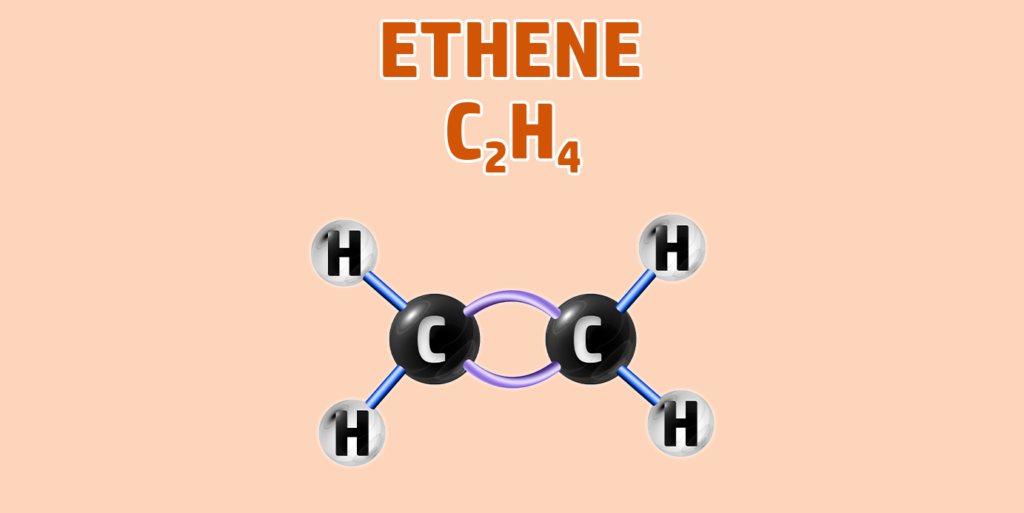
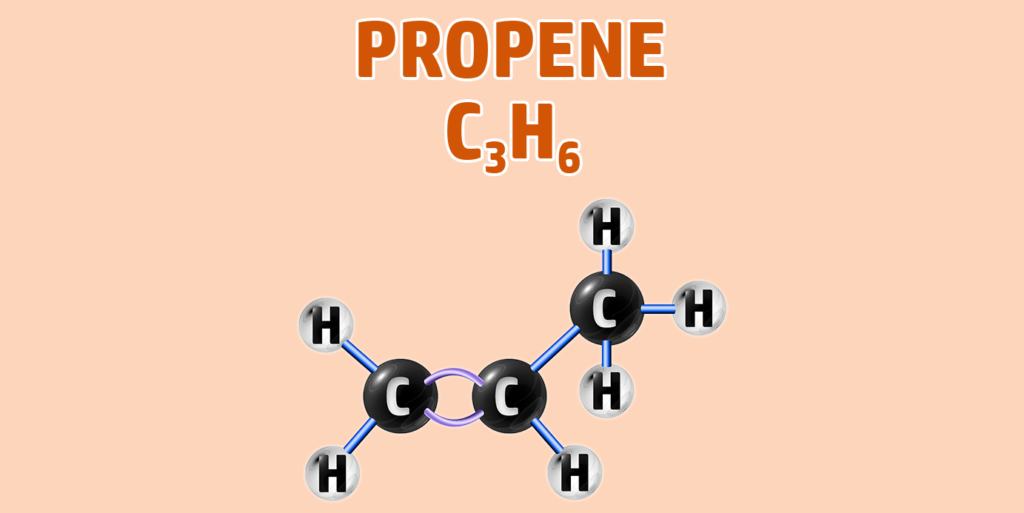
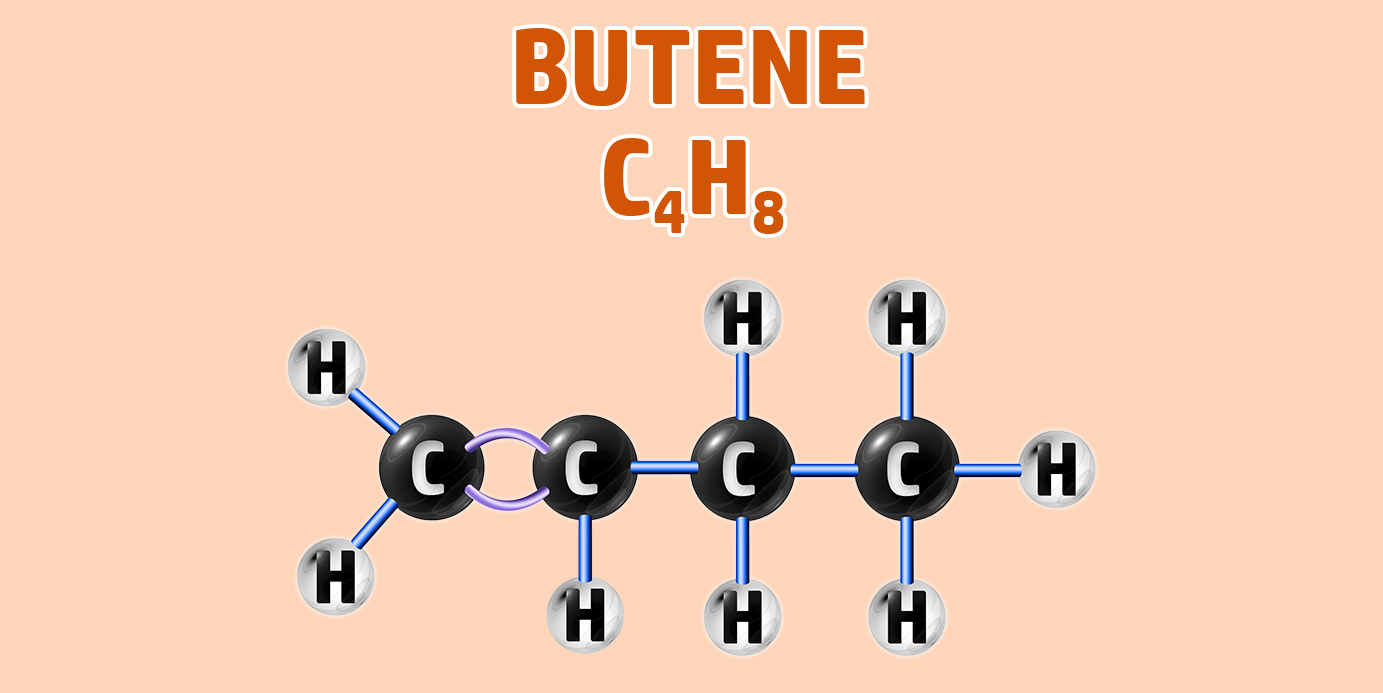
Alkenes can have isomers – which is a chemical with the same molecular formula, but different structure.
For example, butane can have two isomers – but-1-ene and but-2-ene. In but-2-ene, the carbon is attached to the second carbon:
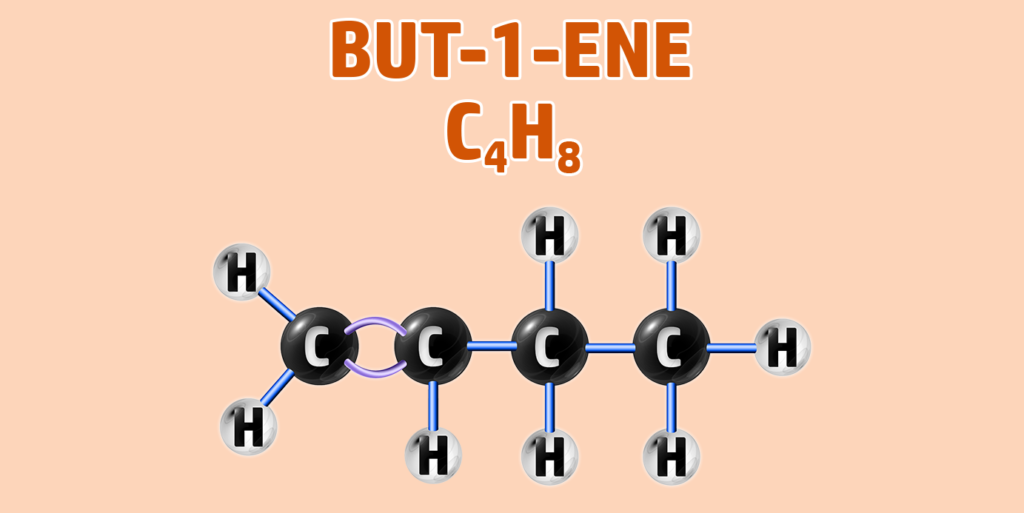
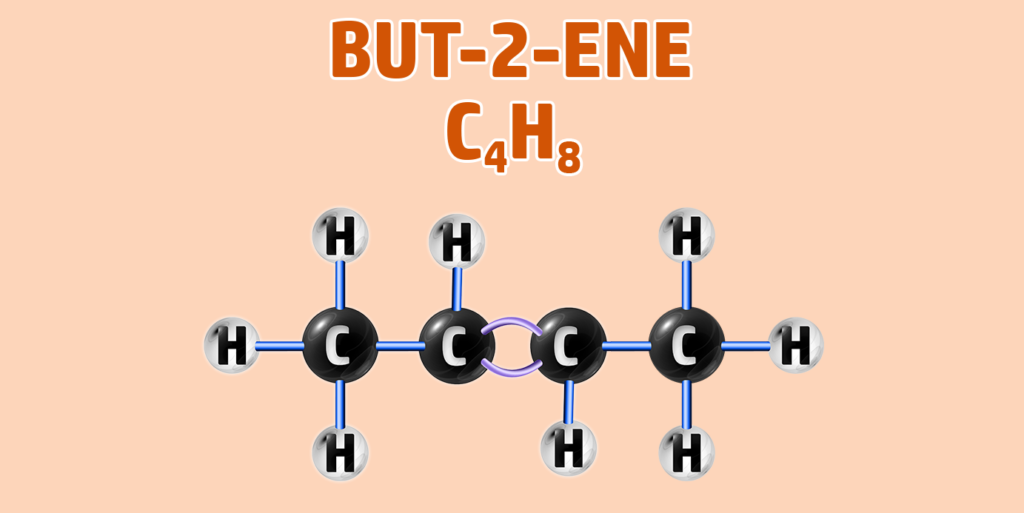
CH247: Explain the test for alkanes and alkenes
To find out if you have an alkane or an alkene, add orange bromine water.
Step 1: Alkene + Bromine Water:
In an alkEne, the bromine water will change from orange to colourless.
This is because an addition reaction has occurred.
The double bond is broken, and the bromine reacts with the alkene to form a bromo-alkane, such as 1,2-dibromoethene below:
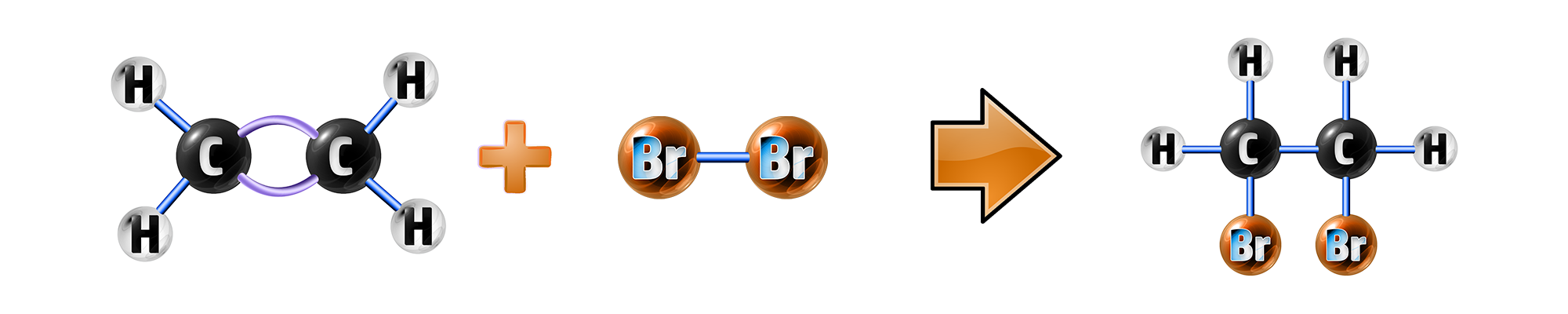
Step 2: Alkane + Bromine Water:
In an alkAne, the bromine water will stay orange. This is because there is no double bond to break, so no reaction can occur:
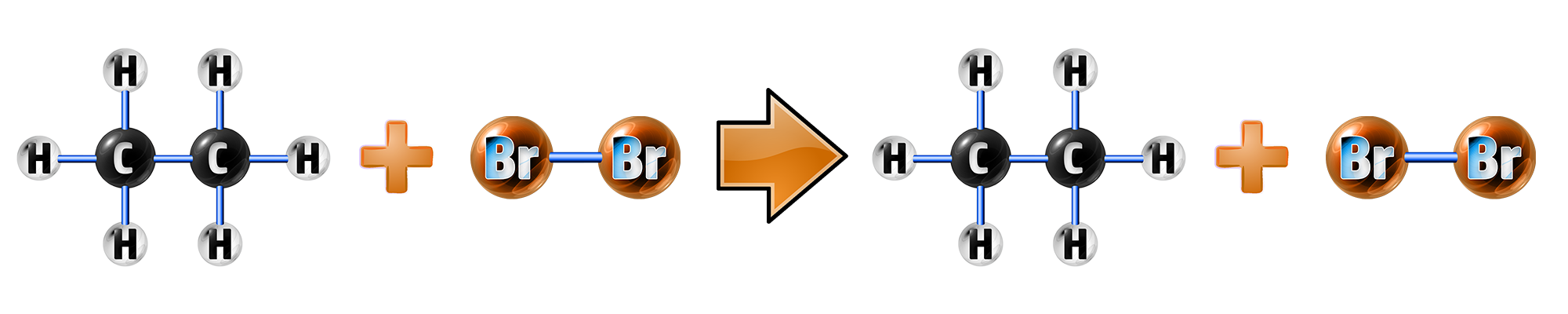
CH248: Explain the complete combustion of alkanes and alkenes
Alkanes and alkenes both react the same as any fuel – producing carbon dioxide and water vapour in complete combustion (see CH220), as well as carbon monoxide and soot in incomplete combustion (see CH221-CH223).
Example: Ethene, C2H4:
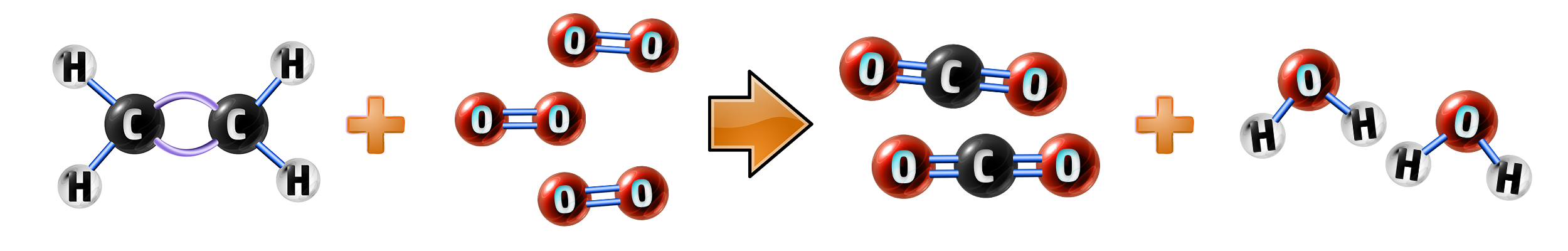
Ethene + oxygen → carbon dioxide + water
C2H4+ 3O2→ 2CO2 + H2O
Watch the video on the right for more examples.
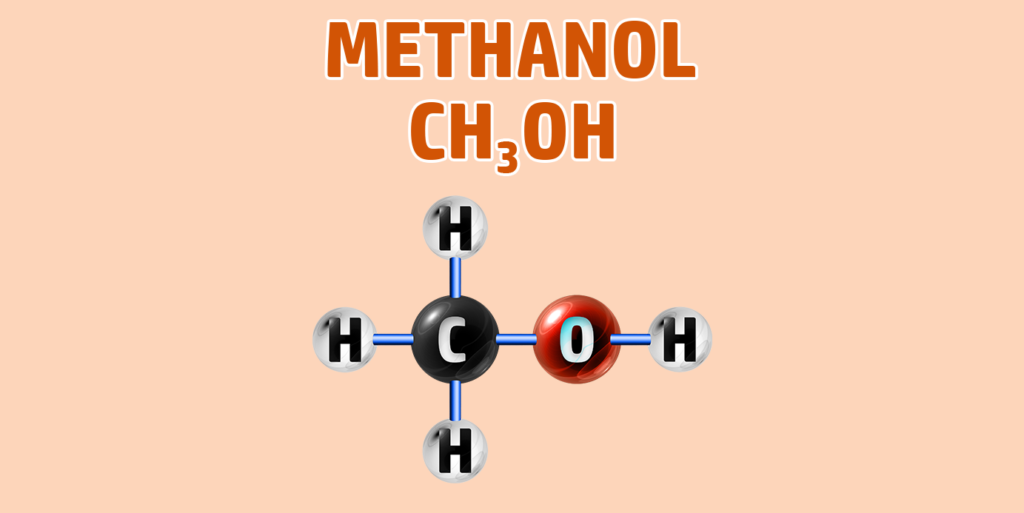
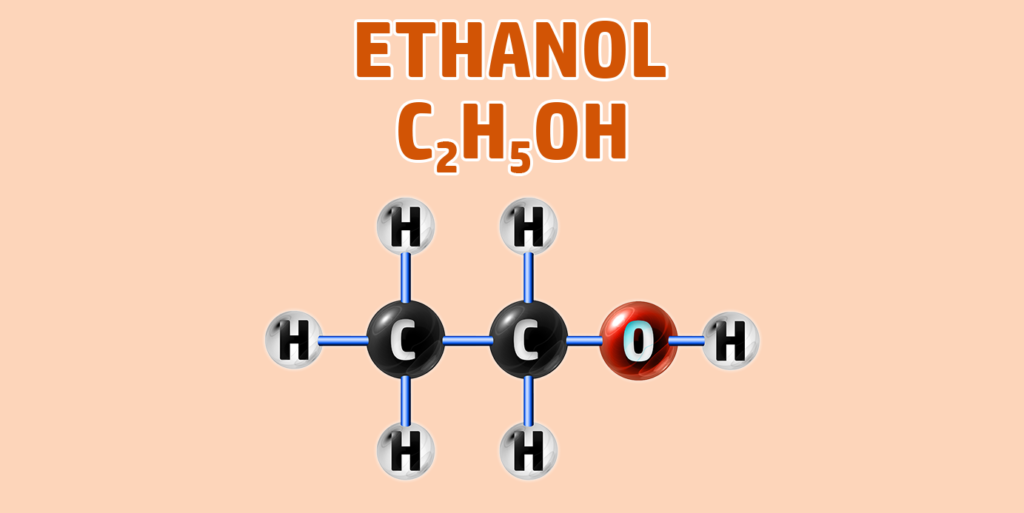
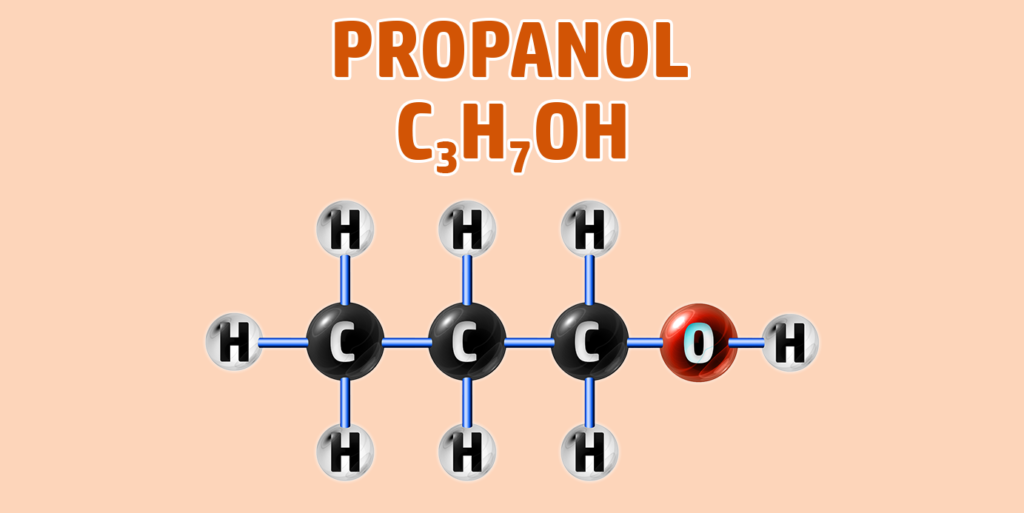
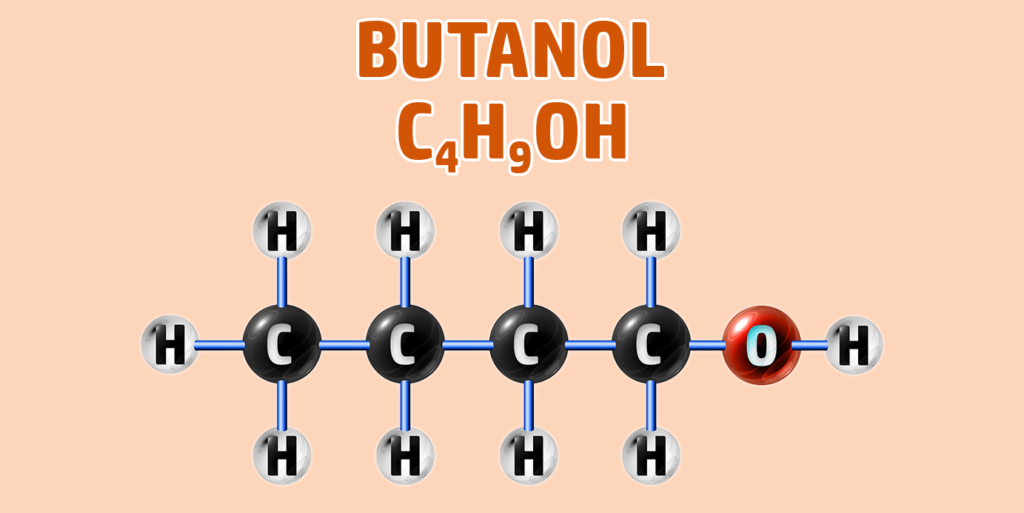
CH249: Draw the structure of any alcohol
Alcohols have the general formula of CnH2n+1OH.
They have the -OH functional group.
To draw an alcohol:
- Step 1: Draw your number of carbons (monkeys eat plastic bananas!).
- Step 2: Add single bonds between all of the carbons.
- Step 3: Each carbon can form 4 bonds. Make sure each carbon has 4 lines coming out of them (including the bonds already formed).
- Step 4: Add an -O-H group to the final carbon.
- Step 5: Place hydrogens (H) onto all other bonds.
CH250: Explain how alcohols can be dehydrated into alkenes
Alcohols can be dehydrated by applying heat and a catalyst.
Dehydration is simply the removal of water from the alcohol to produce an alkene and water:
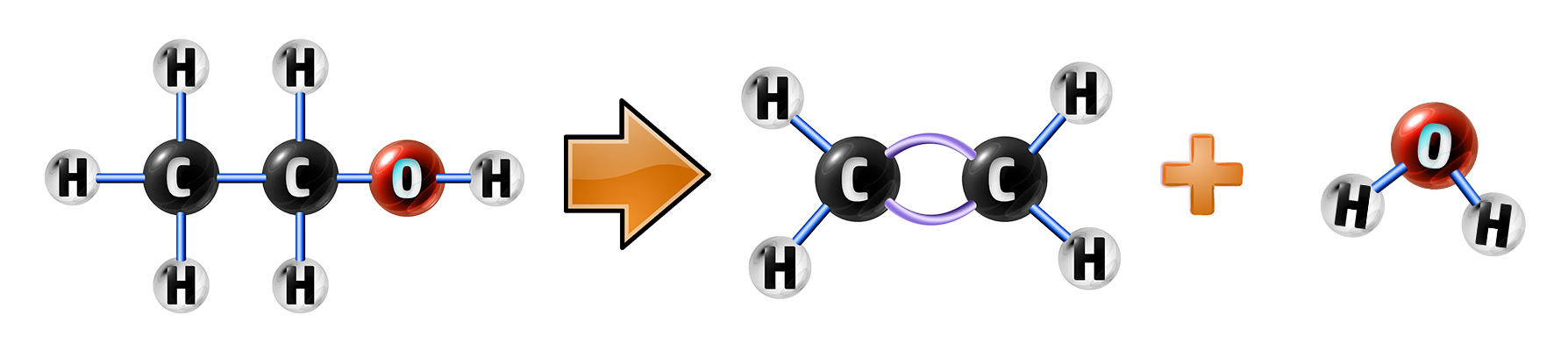
Ethanol → Ethene + Water
C2H5OH→ C2H4 + H2O
CH251: Core Practical: Investigating Combustion of Alcohols
Key Steps: Mass → Temperature → Heat → Re-measure → Repeat

- Measure the mass of a spirit burner and cap.
- Add 100cm3 of cold water to a conical flask and record the temperature.
- Light the wick and heat the water, increasing the temperature by 20oC.
- Replace the cap, re-measure the mass and work out the change in mass.
- Repeat for alcohols with different chain lengths.
Results:
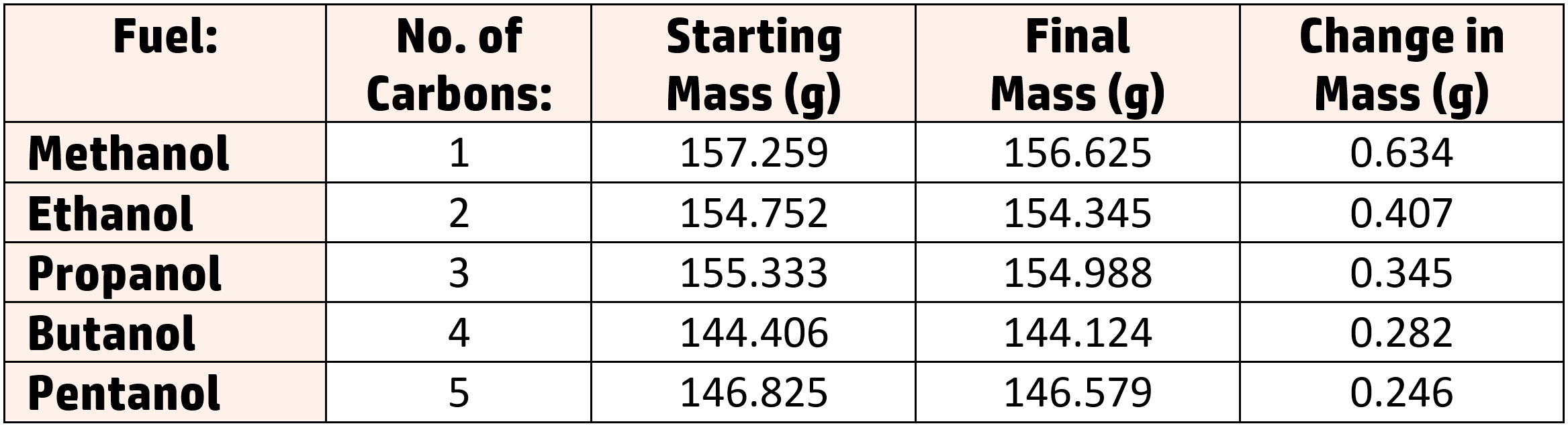
Calculations:
You may be asked to work out the temperature rise per gram for one of the fuels.
To do this, divide the change in temperature (in this experiment we increased the temperature by 20oC) by the change in mass.
Example: Calculate the temperature rise per gram when it took 0.634g of methanol to increase the temperature by 20oC.
= temperature rise ÷ change in mass
= 20oC ÷ 0.634g = 31.55oC/g
This shows that 1g of methanol will raise the temperature by 31.55oC
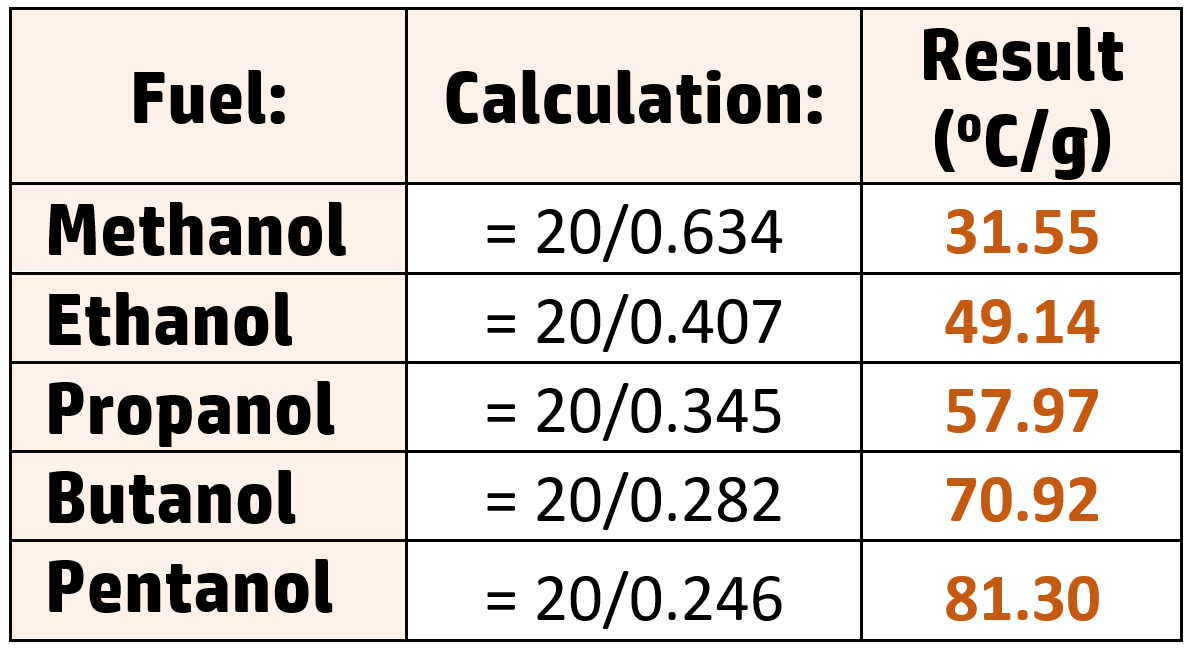
Conclusion:
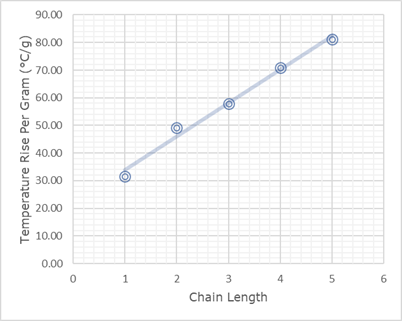
As the chain length increases, the temperature rise per gram increases.
Methanol produces a temperature rise of 31.55oC/g, whereas pentanol produces a temperature rise of 81.30oC/g.
Variables:
- Independent Variable: The (chain length of the) alcohol.
- Dependent Variable: The change in mass
- Control Variables:
- The rise in temperature (20oC).
- The distance between the wick and the conical flask.
- The volume of water (100cm3).
- The insulation around the conical flask.
Risks and management:
- Alcohols are highly flammable and can produce flammable vapours which can cause burns to the skin as well as fires. Only light the wick with a splint and keep the lab well ventilated.
- Methanol is a toxic chemical when in contact with the skin. Avoid skin contact and wash off if it does come in contact with the skin.
CH252: Explain the formation of ethanol by fermentation of glucose
Alcohol is used as a solvent in cosmetics, as well as for medicinal drugs and varnishes. Ethanol is also used as a fuel and in alcoholic drinks.
Ethanol is produced from sugars, usually from fruits. This sugar undergoes fermentation, where yeast is added and the enzymes in yeast break down the glucose into carbon dioxide and ethanol:
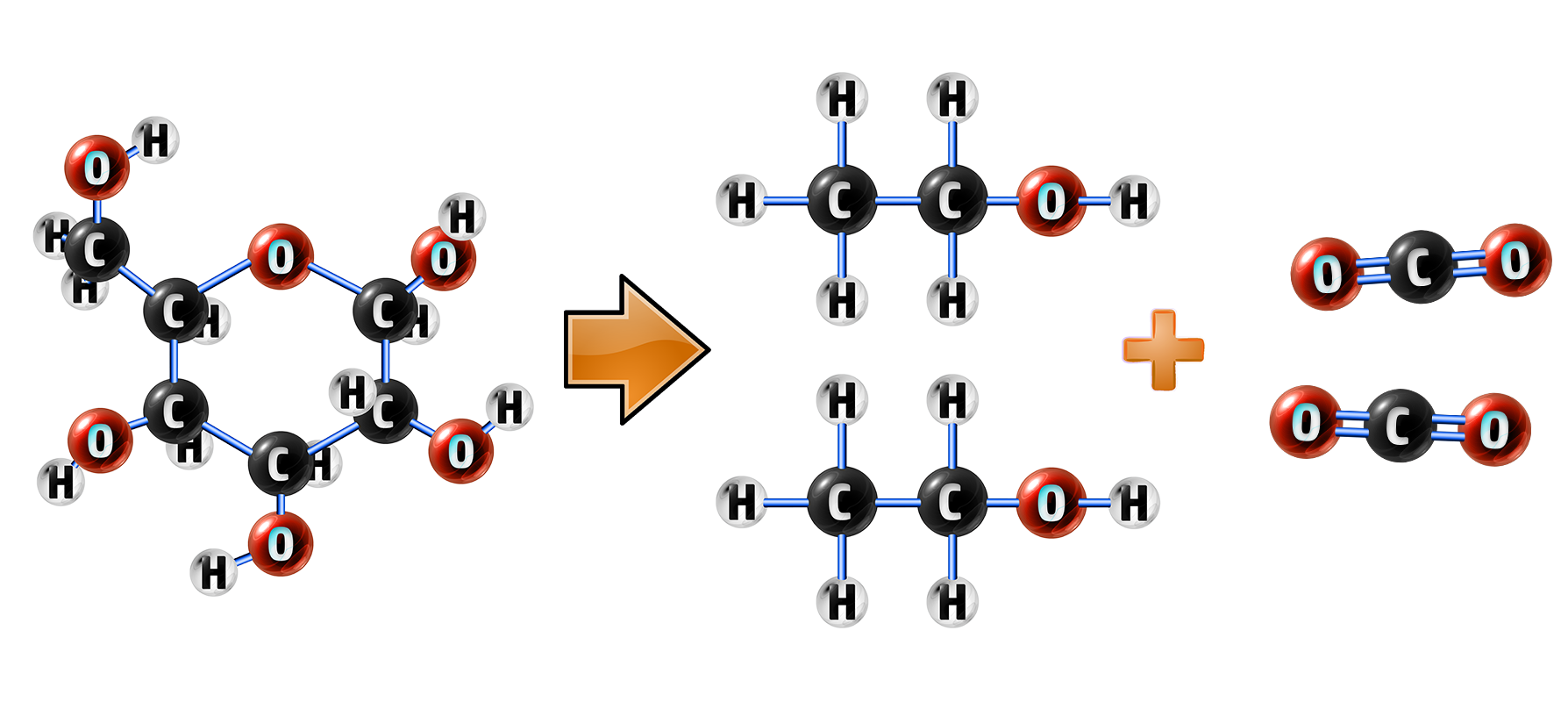
Glucose → Ethanol + Carbon Dioxide
C6H12O6 → 2C2H5OH + 2CO2
Fermentation uses anaerobic respiration, so the volume of oxygen must be controlled.
CH253: Explain how to make alcohol more concentrated
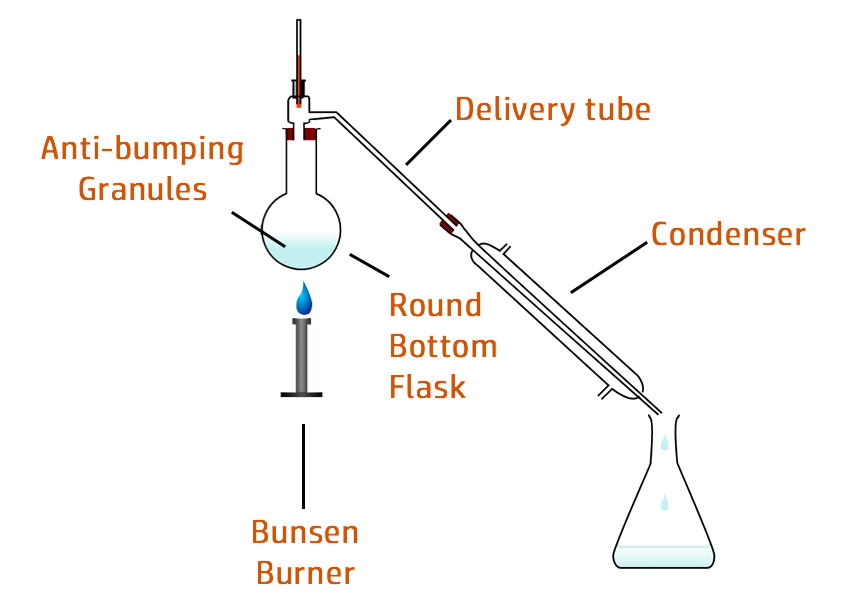
Alcohol, produced by fermentation, only produces about 15% ethanol.
Once you go above this, the higher concentrations kill the yeast.
To produce high concentrations, fractional distillation is then used to evaporate and collect the ethanol (which evaporates at 78oC), leaving behind the water which doesn’t boil until you reach 100oC.
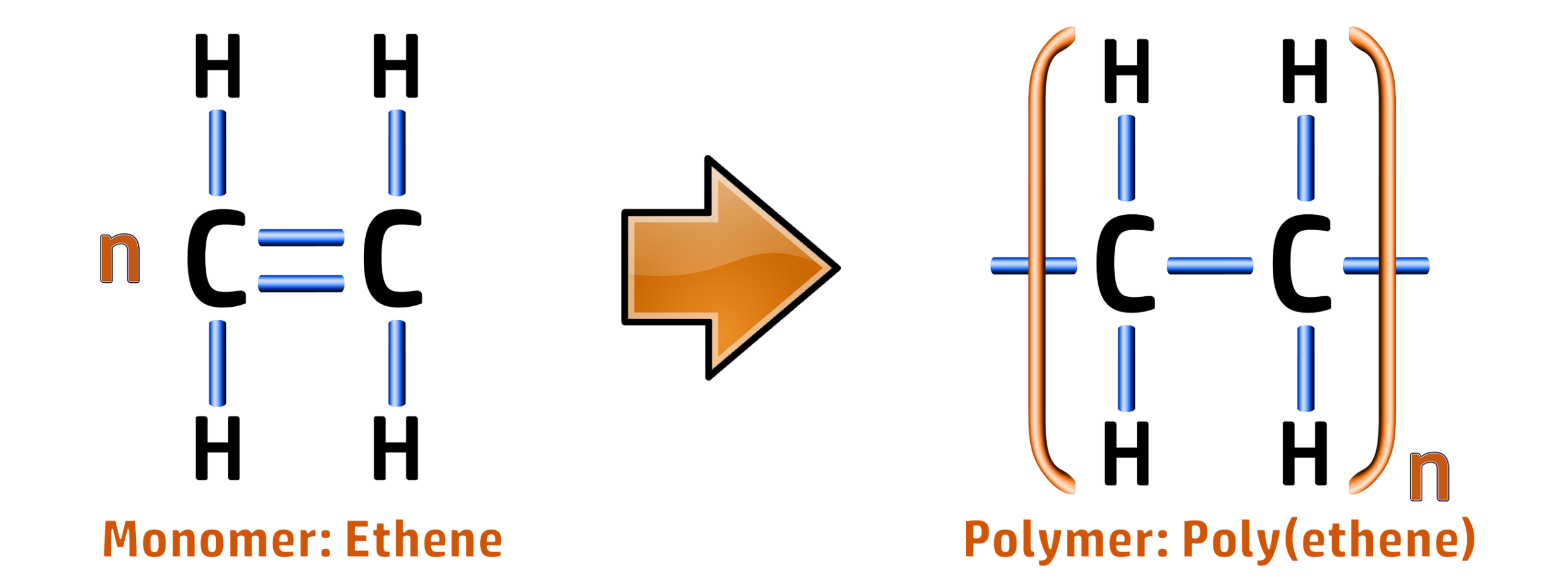
CH254: Explain how polymers form during addition polymerisation
Polymers (See CH72) can be formed in two main ways. Addition Polymerisation and Condensation polymerisation (see CH263).
The addition polymerisation of ethene occurs when a monomer (small molecule) has its double bond broken…
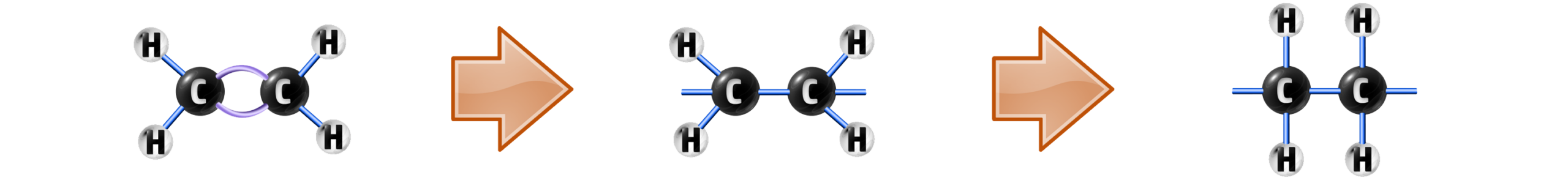
…and then many monomers are added together forming polyethene:
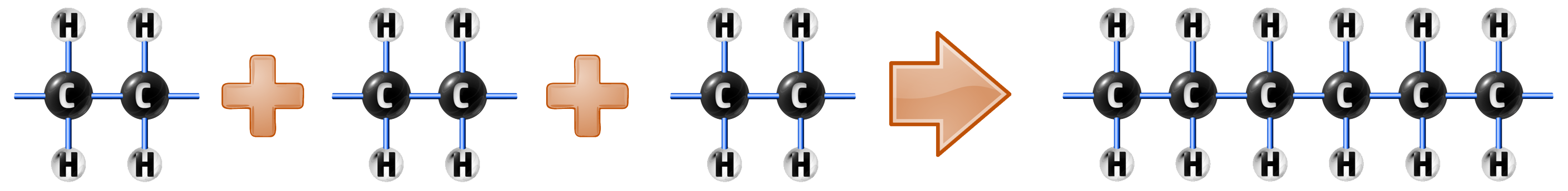
When writing out polymers, you can write them like this:
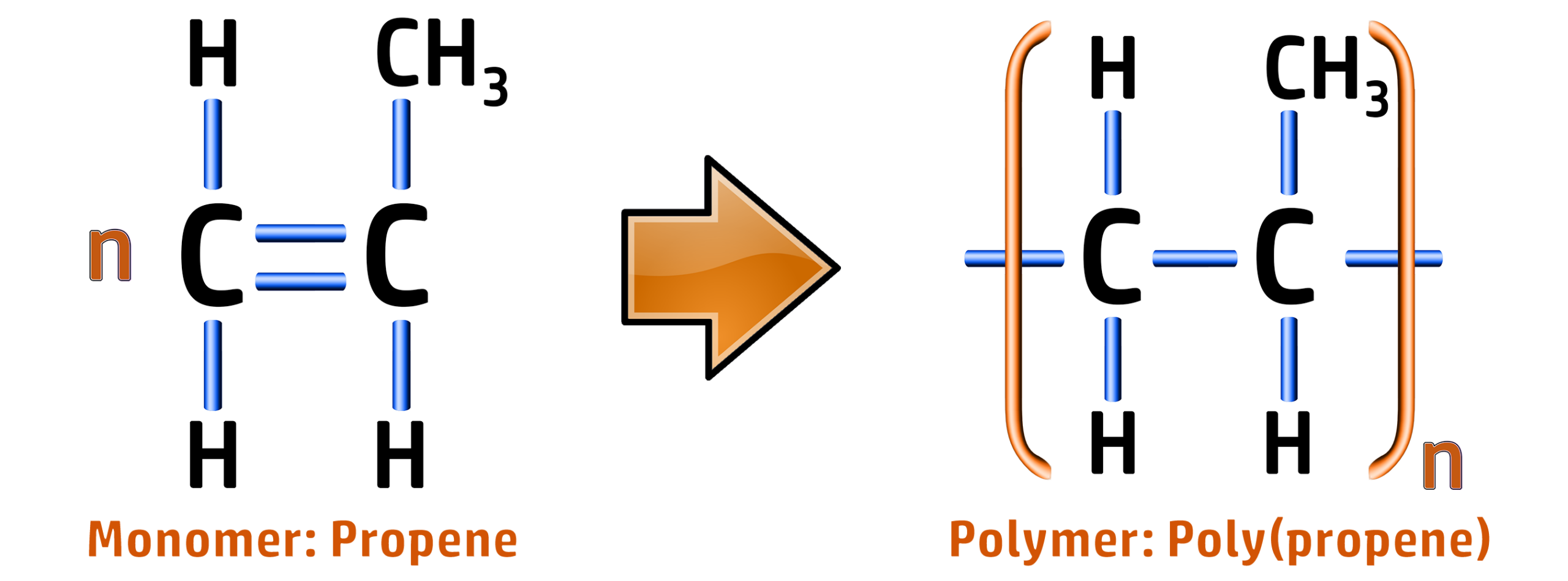
CH255: Draw repeating units for polypropene, PVC and Teflon
Poly(propene):
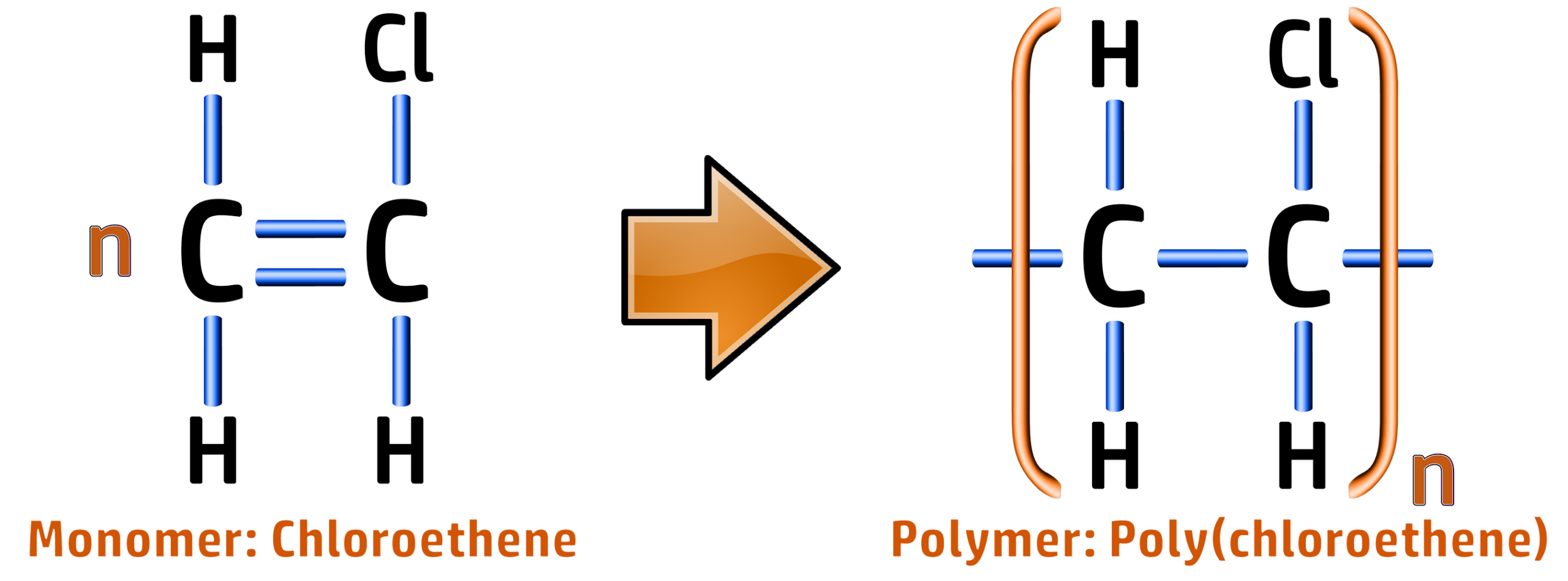
Poly(chloroethene) / PVC:
Poly(tetrafluoroethene) / Teflon::
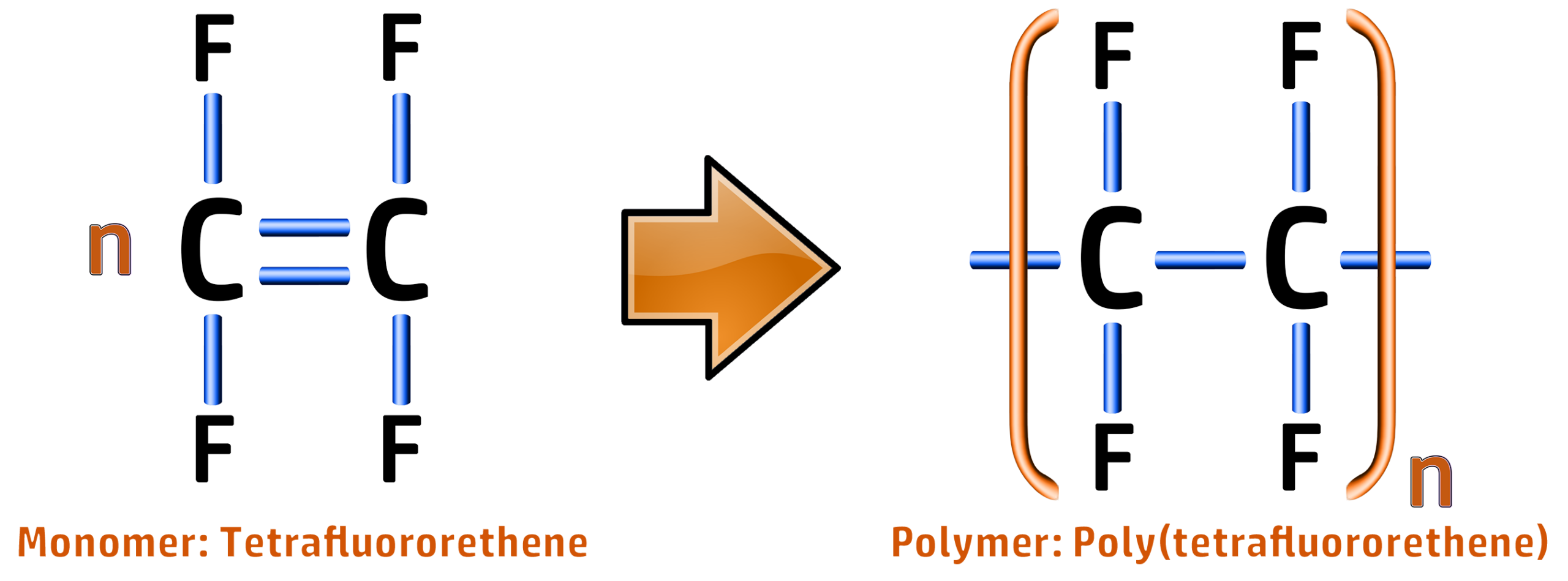
CH256: Draw monomers from polymer chains
When given a polymer and asked to draw the monomer for it, all you need to do is look at the first two carbons.
Example: Draw the monomer for the following polymer of poly(bromoethene) below:
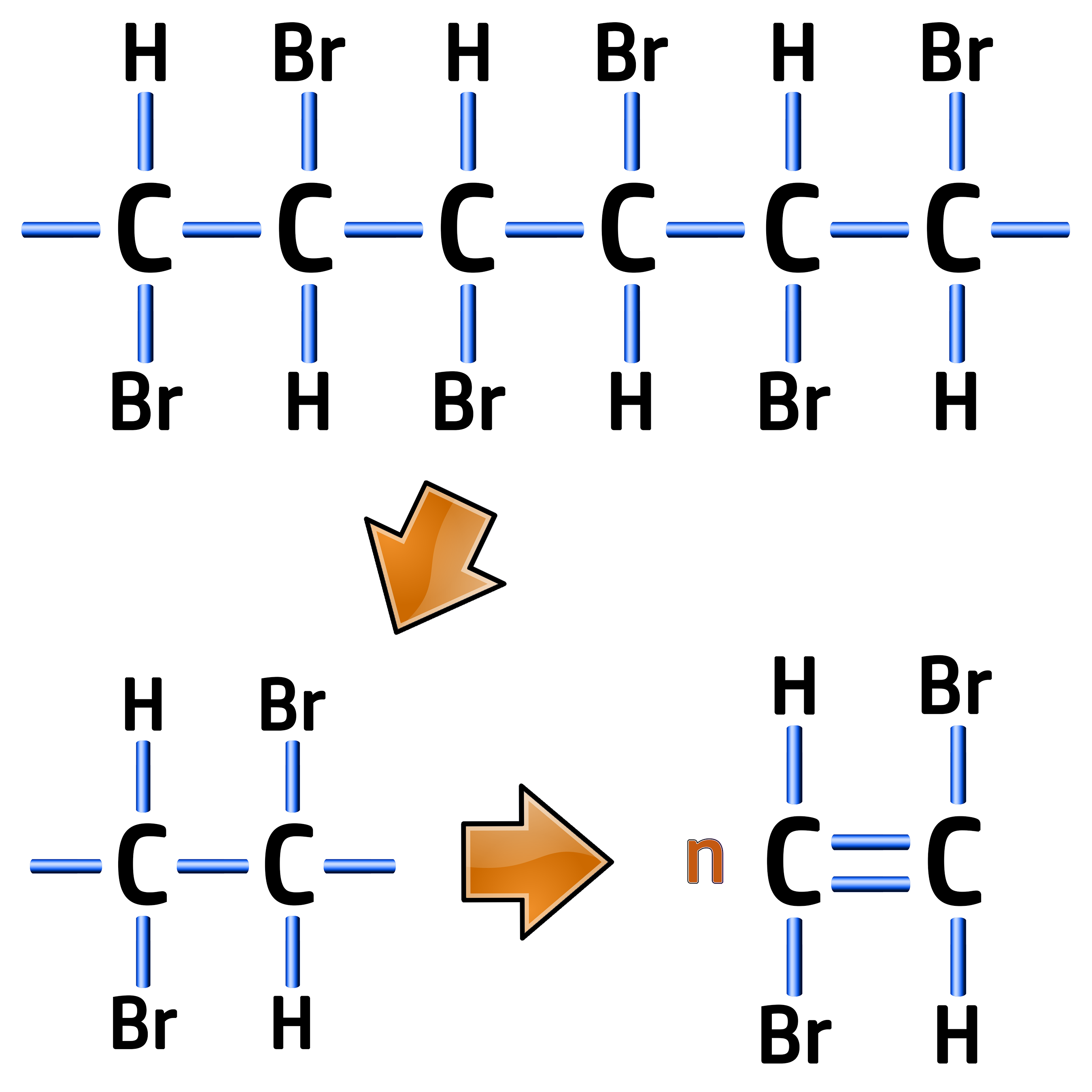
The repeating unit for an addition polymer is always two carbons wide, so you need to replicate the first two carbons making sure the elements above and below them are kept in the same order.
All you need to do is redraw the first two carbons with a double bond between them, like on the left.
CH257: Describe the uses of different polymers and link to their uses

Polymer: Poly(ethene) / Polythene
Formula: [C2H4]n
Type: Addition Polymer
Use: Plastic bags, bottles and clingfilm.
Reason: Flexible, cheap, good insulator.

Polymer: Poly(propene) / Polypropylene
Formula: [C3H6]n
Type: Addition Polymer
Use: Buckets, Crates, Bowls, Ropes and Carpets.
Reason: Flexible, does not shatter.

Polymer: Poly(chloroethene) / PVC
Formula: [C2H3Cl]n
Type: Addition Polymer
Use: Window frames, gutters, pipes, Wire insulation.
Reason: Tough, good insulator for wires.

Polymer: Poly(tetrafluoroethene) / Teflon
Formula: [C2F4]n
Type: Addition Polymer
Use: Non-stick coating for frying pans.
Reason: Tough, slippery.

Polymer: Poly(ethylene terephthalate) / PET
Formula: [C10H8O4]n
Type: Condensation Polymer
Use: Plastic furniture, carpets, fleece, bottles.
Reason: Strong, lightweight, shatter resistant.
CH258: Evaluate the different ways to dispose of polymers
Polymers are non-biodegradable which means it will take 1000’s of years for them to decompose.
There are three ways of disposing of polymers:
Method: Burying in Landfills

Advantages:
- Quick to dispose of.
Disadvantages:
- Landfills fill up
- Non-biodegradable so won’t decompose.
Method: Burning Plastics

Advantages:
- Quick to dispose of.
- Can use heat energy as a fuel.
Disadvantages:
- Burning produces carbon dioxide – which is a greenhouse gas
- Some plastics produce toxic gases.
Method: Recycling

Advantages:
- We don’t run out of our oil which is a finite resource.
- Codes can be used to help to sort the plastics more easily.
Disadvantages:
- It takes time & money to sort the plastics into groups
- Some plastics cannot be recycled and end up in landfills
- The polymers then need cleaning, chipping, and melting to form the new product.
CH259: Describe DNA as a polymer

DNA is a polymer made up of the monomers Adenine, Thymine, Guanine and Cytosine.
Starch is also a polymer made up of sugar (glucose) molecules joined together.
Proteins are also polymers made from amino acids such as glycine, right:
CH260: Draw the structure of different carboxylic acids
Carboxylic acids have the -COOH functional group.
To draw carboxylic acids:
- Step 1: Draw your number of carbons (monkeys eat plastic bananas!).
- Step 2: Put a single bond between each of the carbons..
- Step 3: Turn the final carbon into the carboxyl group to the last carbon (an =O and an -O-H group).
- Step 4: Make each other carbon up to 4 bonds and add a hydrogen to each bond:
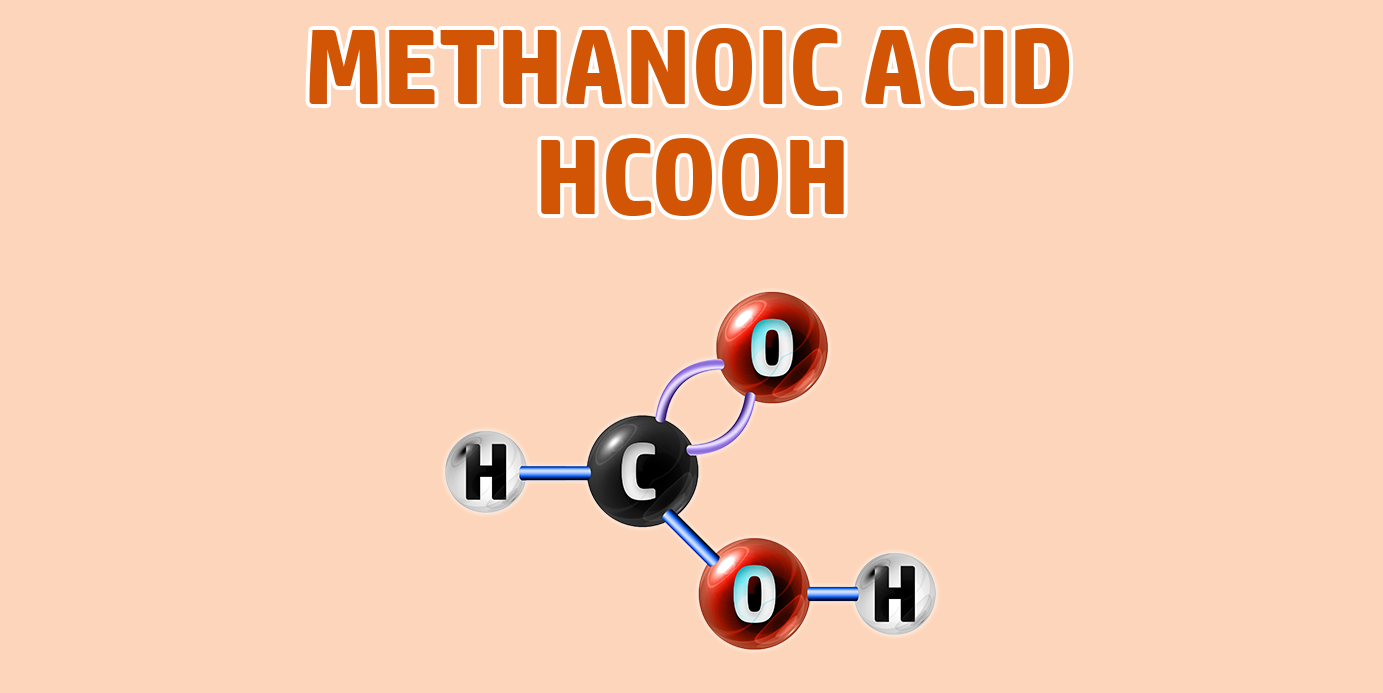
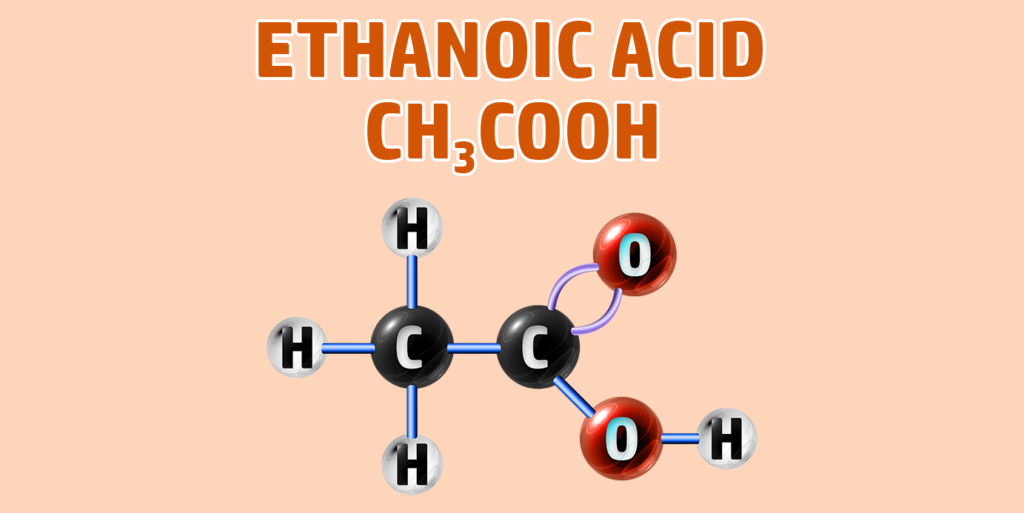
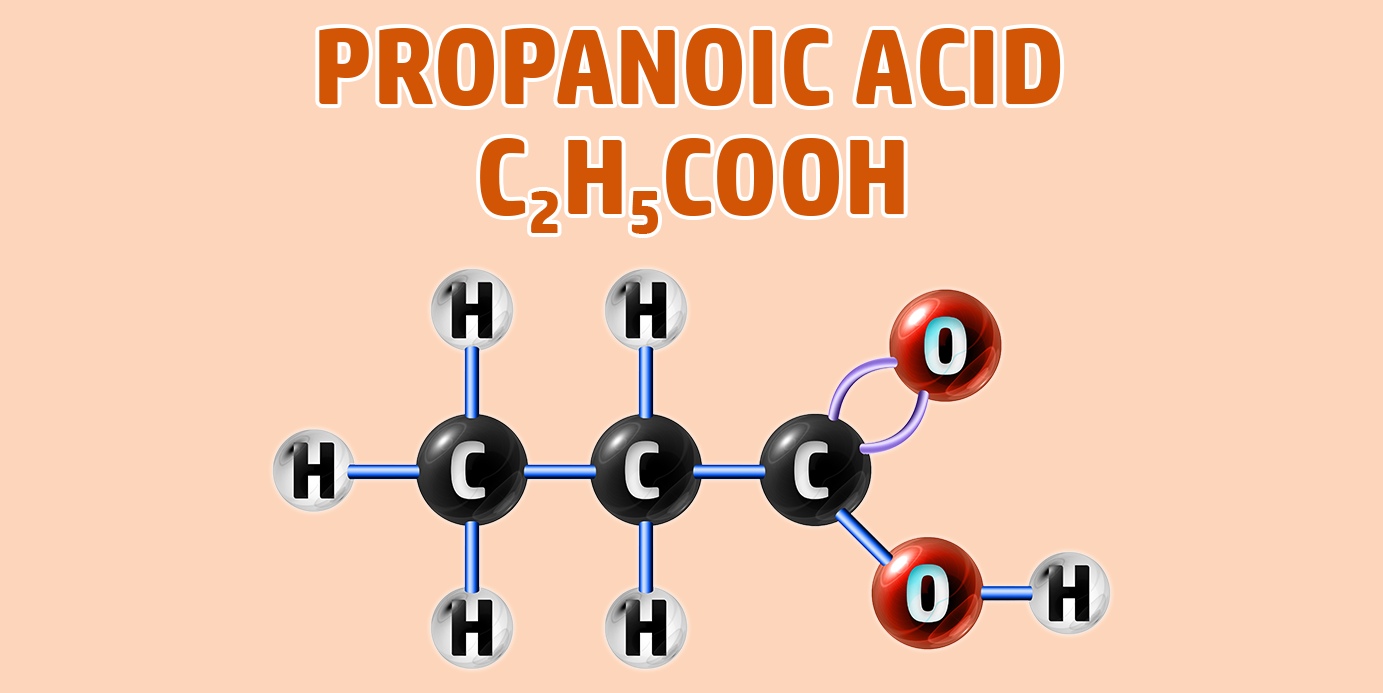
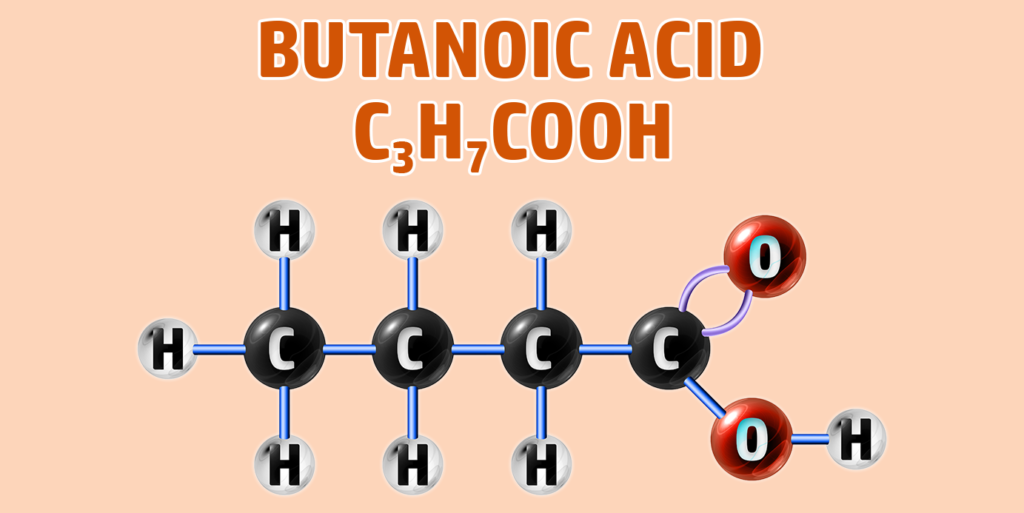
If you are trying to remember the order of the first 4 alkanes, remember Monkeys Eat Plastic Bananas!
CH261: Explain the reactions of carboxylic acids
When a carboxylic acid reacts with a metal, the salt formed takes on the name of the acid by changing the ‘-ic acid’ ending to ‘-oate’:
- Methanoic acid forms a methanoate
- Ethanoic acid forms as Ethanoate
- Propanoic acid forms a Propanoate
- Butanoic acid forms a Butanoate
Example 1: Sodium + Ethanoic acid:
- Sodium is a metal on its own, so will form hydrogen (CH116)
- Ethanoic acid will form an ethanoate, so your salt is sodium ethanoate.
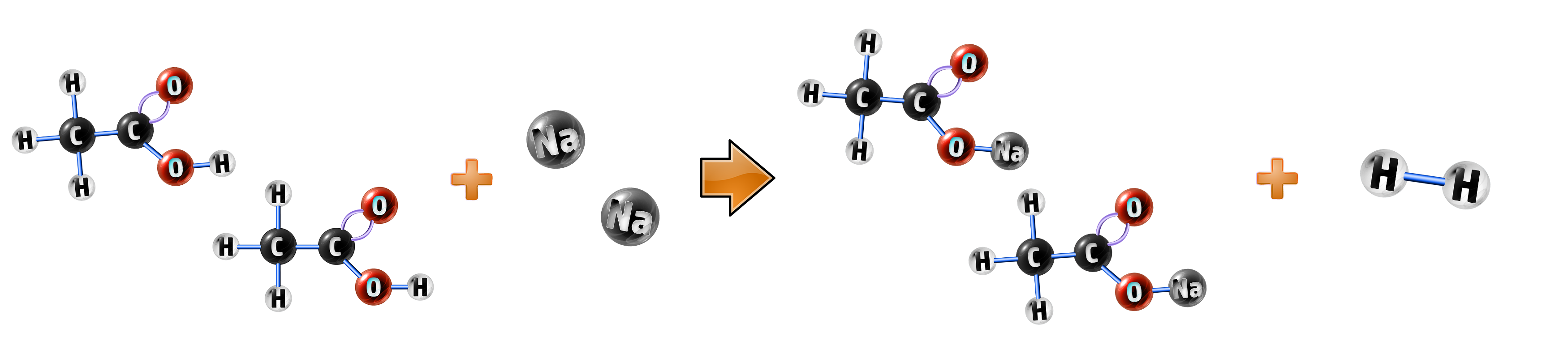
ethanoic acid + sodium → sodium ethanoate + hydrogen
2CH3COOH + 2Na → 2CH3COONa + H2
Example 2: Propanoic acid + magnesium hydroxide:
- Magnesium hydroxide will always form water (CH117)
- Propanoic acid will form a propanoate, so your salt is magnesium propanoate.
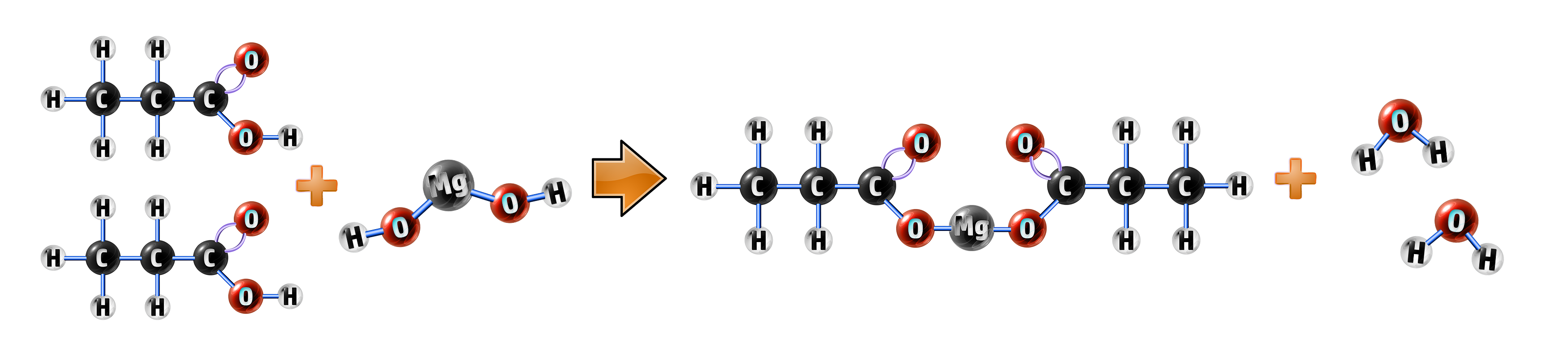
propanoic acid + magnesium hydroxide → magnesium propanoate + water
2C2H5COOH + Mg(OH)2 → (C2H5COO)2Mg + H2O
Example 3: Methanoic acid + calcium carbonate:
- Calcium carbonate will always form carbon dioxide and water (CH118)
- Methanoic acid will form a methanoate, so your salt is calcium methanoate.
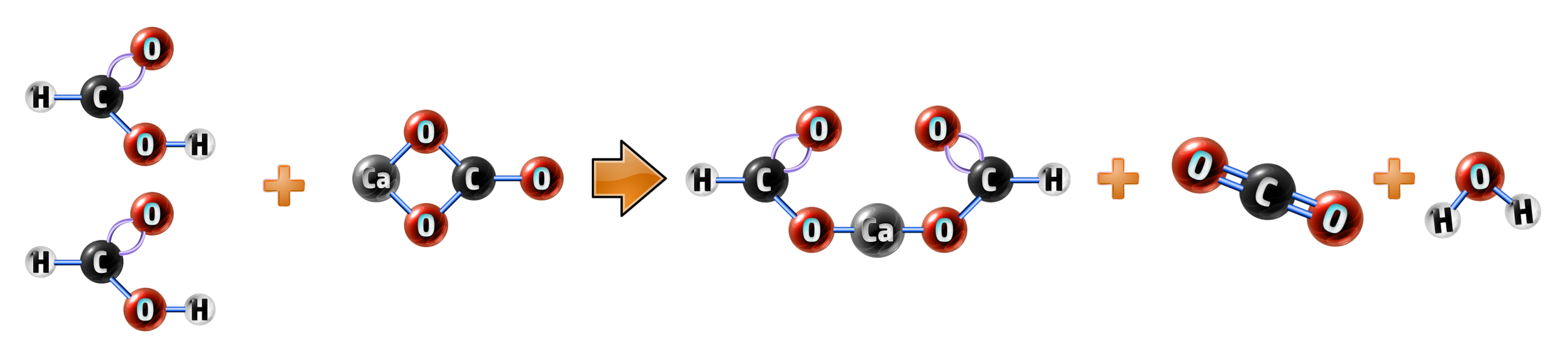
methanoic acid + calcium carbonate → calcium methanoate + carbon dioxide + water
2HCOOH + CaCO3 → (HCOO)2Ca + CO2 + H2O
CH262: Explain how alcohols can be turned into carboxylic acids
Alcohols, such as ethanol, can be oxidised using an oxidising agent to form carboxylic acids:
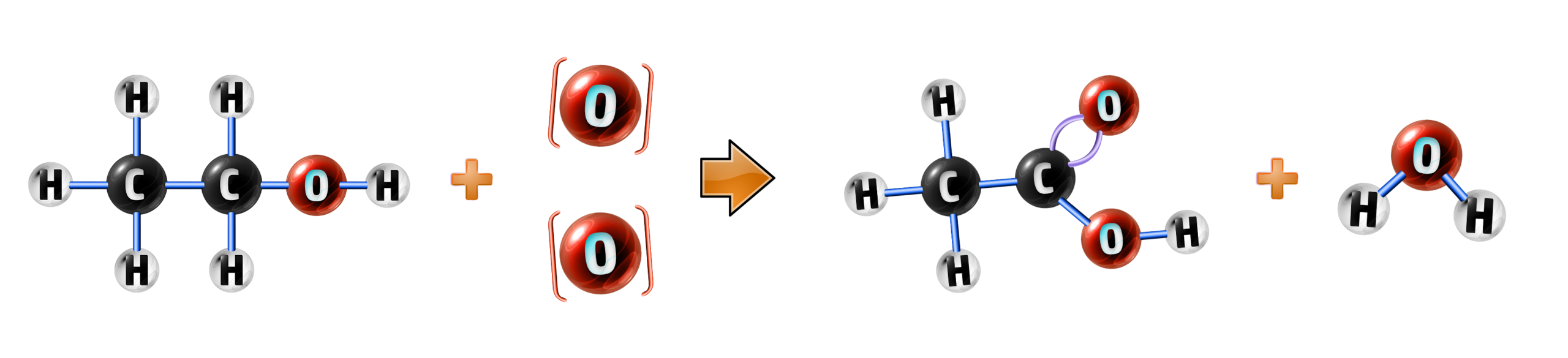
ethanol + oxidising agent → ethanoic acid + water
C2H5OH + 2[O] → CH3COOH + H2O
The oxidising agent is any chemical that can be added to ethanol to oxidise it (add oxygen). It is represented as [O] in a chemical equation.
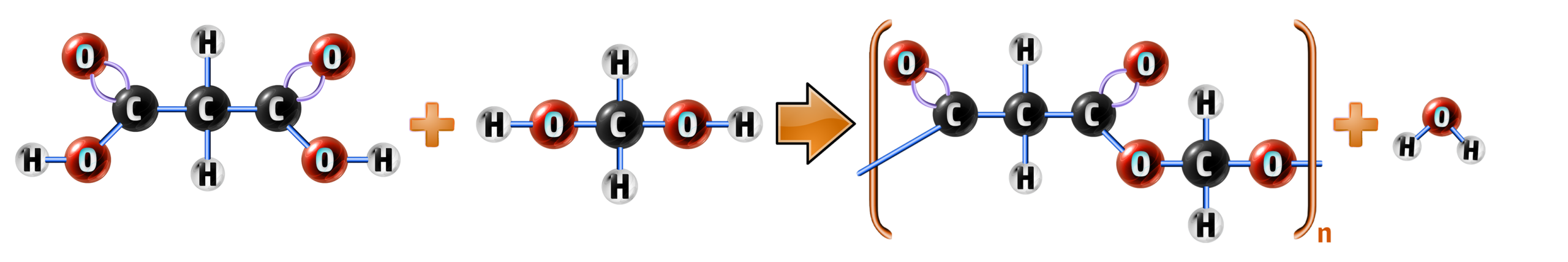
CH263: Explain how condensation polymerisation can be used to form polyesters
Condensation polymers involve a different mechanism to addition polymerisation (CH254). It involves an alcohol and a carboxylic acid and produces two products – your polymer and water:
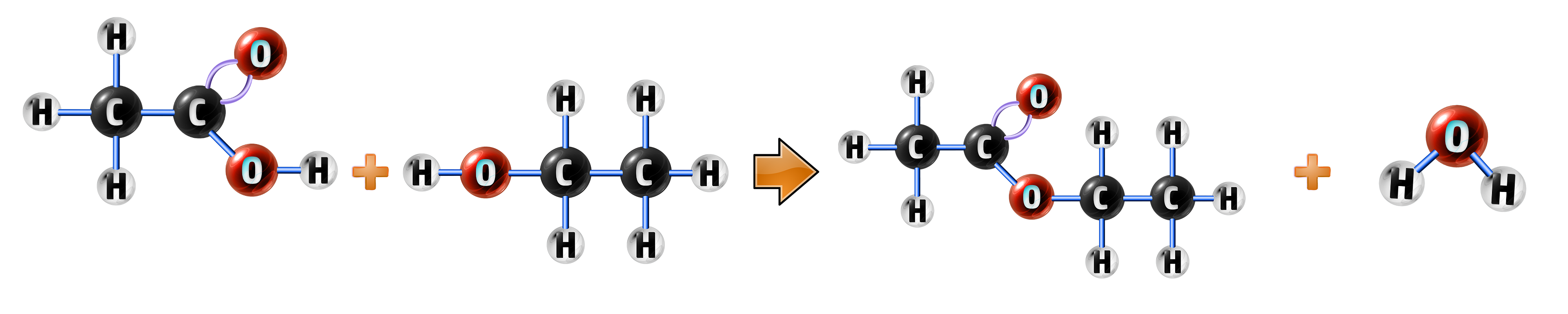
ethanoic acid + ethanol → ethyl ethanoate + water
CH3COOH + C2H5OH → CH3COOC2H5 + H2O
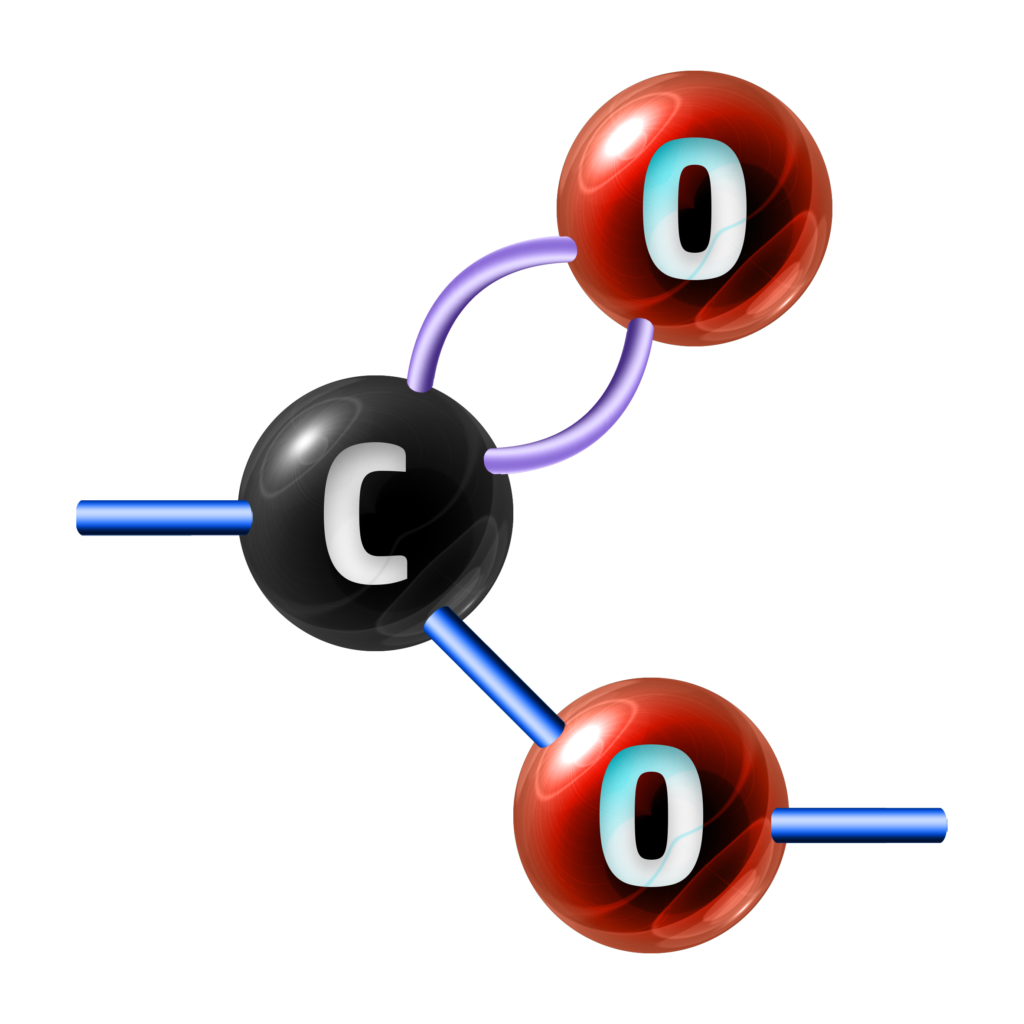
A polyester is a long-chained molecule that contains many ester links (see left).
To form a polyester, both carboxylic acid and alcohol need to have the same functional group at both sides of the molecule.
How to draw polyesters:
Example: Draw the polyester for methyl propanoate from the polymers below:
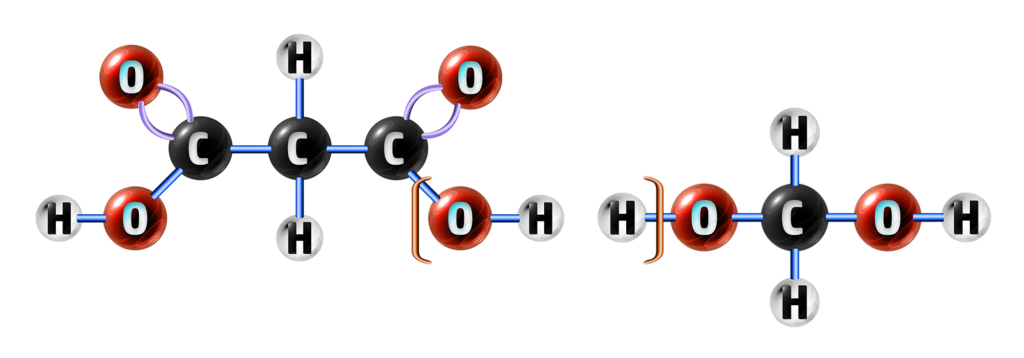
Step 1: Find the OH on the carboxylic acid and the H connected to the O on the alcohol and remove them.
This will form your water molecule.
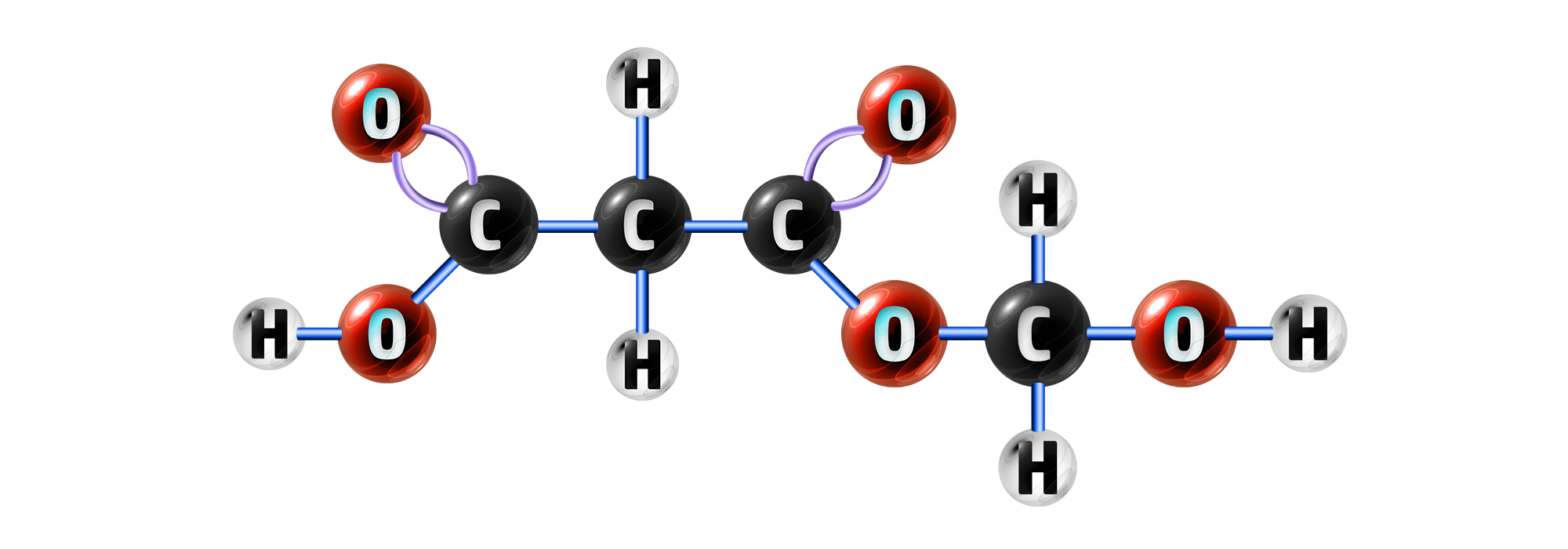
Step 2: Connect the left-over bonds from the carbon on the carboxylic acid and the oxygen on the alcohol.
This creates your ester group.
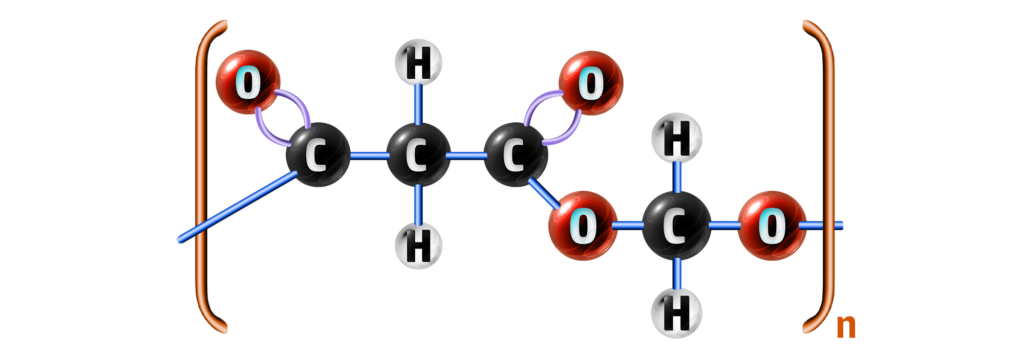
Step 3: Remove the OH from the other side of the carboxylic acid and the H from the other side of the alcohol.
Add a bracket around the polymer and you have your repeating unit.
Full equation: