SEPARATE CHEMISTRY P2: IONS AND MATERIALS


GCSE revision page on ions and materials covering flame tests, hydroxide precipitates, tests for ammonium, carbonates, sulfates and halides, an ions core practical, instrumental analysis (flame photometry), plus nanoparticles, ceramics, composites and choosing materials based on properties.
CH237: Describe why an ion test must be unique
When you carry out a test to identify which ions are present in an ionic compound, the result must be unique.
If it is not, you cannot draw a conclusion.
For example, if you added sodium hydroxide to an unknown solution and it gave a white precipitate it could be either aluminium or calcium present (see CH239). This is not unique, so you don’t know what ion is present. Therefore, you must find another way to test for that ion.
CH238: Identify the flame colours for different cations
Get a wire loop, add it to a flame and dip it in hydrochloric acid to clean it.
Add some of your sample to the cleaned wire loop and add it to your Bunsen Burner on a blue flame. Note the colour and use it to identify the ion present:
LITHIUM

RED FLAME
SODIUM

YELLOW FLAME
POTASSIUM

LILAC FLAME
CALCIUM

ORANGE-RED FLAME
COPPER

BLUE-GREEN FLAME
CH239: Identify the precipitate colours for different cations
Precipitation reactions involving sodium hydroxide often form coloured hydroxide precipitates. Dissolve your ionic substance, add your sodium hydroxide & note the colour:
IRON (II)
Fe2+
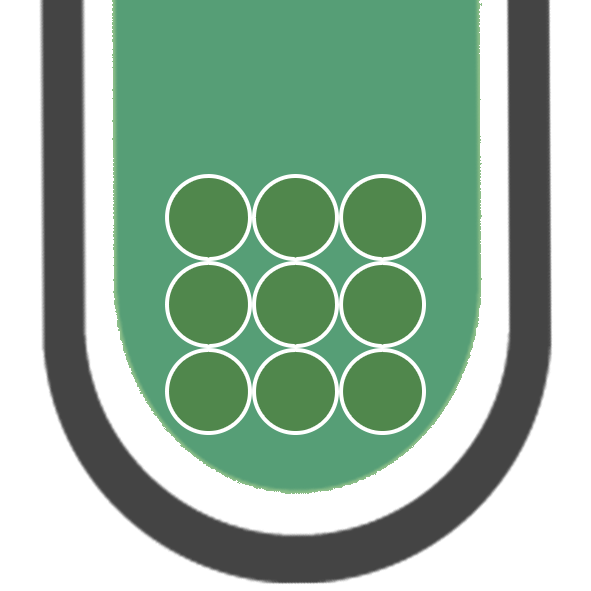
GREEN PRECIPITATE
IRON (III)
Fe3+
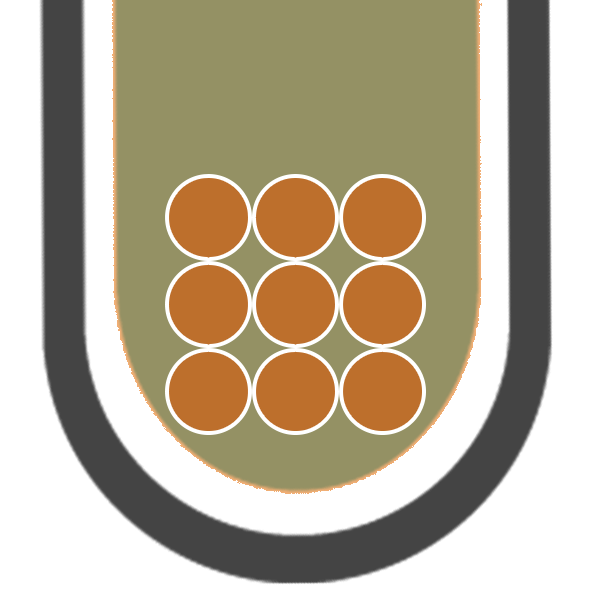
BROWN PRECIPITATE
COPPER (II)
Cu2+

BLUE PRECIPITATE
CALCIUM
Ca2+

WHITE PRECIPITATE
ALUMINIUM
Al3+

WHITE PRECIPITATE
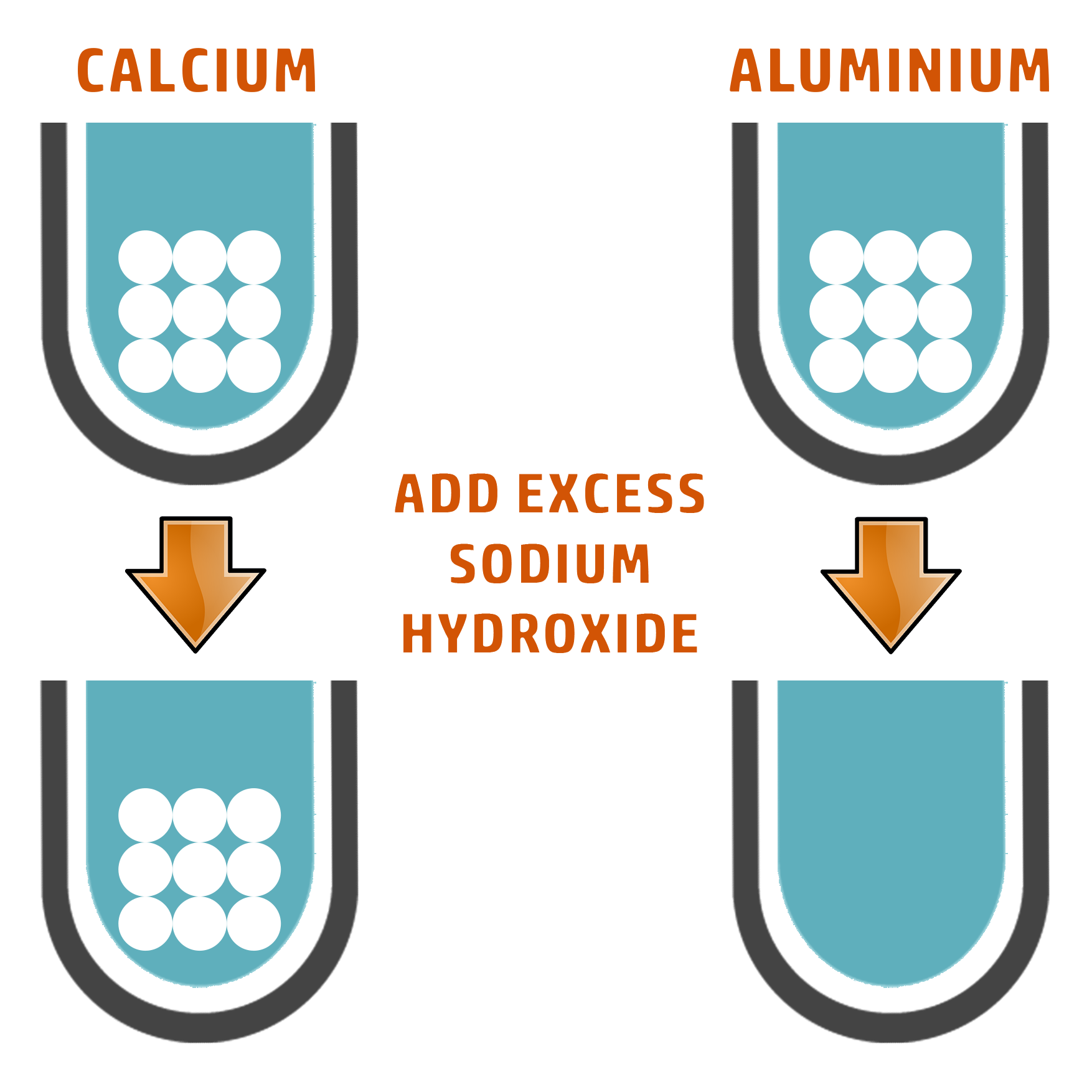
Both aluminium and calcium give a white precipitate, meaning you can’t prove which is which.
To do that, add excess sodium hydroxide.
If the precipitate dissolves, aluminium is present.
If the precipitate doesn’t dissolve, it is calcium
CH240: Identify the test for ammonia
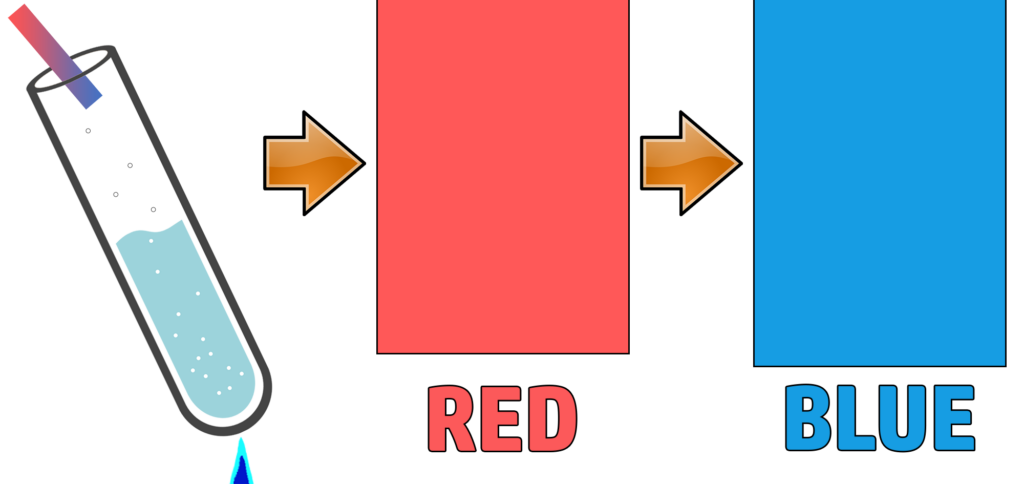
When sodium hydroxide is added to a solution containing ammonium ions, and heated, a gas (ammonia) is given off with a sharp smell.
A confirmatory test for ammonium is to add damp red litmus paper – it will go blue in the presence of ammonia.
CH241: Describe the test for carbonates, sulfates and halides
Test for carbonates, CO32-:
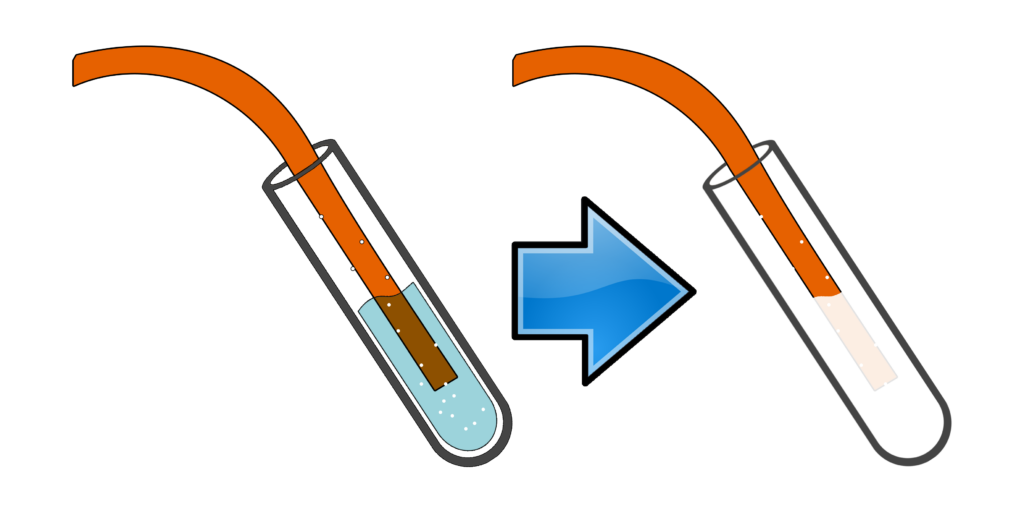
Add any acid.
Bubble the gas through limewater.
If limewater goes cloudy, you had a carbonate.
Example:
Balanced: CaCO3 + 2HCl → CaCl2 + H2O + CO2
Ionic: CO32-(aq) + 2H+ (aq) → H2O (l) + CO2 (g)
Test for sulfates, SO42-:

Add hydrochloric acid. This will acidify the solution and make sure there are no carbonate ions (no gas should form).
Add barium chloride. A precipitation reaction will occur and a white precipitate of barium sulfate will form.
Example:
Balanced: BaCl2 (aq) + MgSO4 (aq) → BaSO4 (s) + MgCl2 (aq)
Ionic: Ba2+ (aq) + SO42- (aq) → BaSO4 (s)
Test for halides, Cl-, Br- and I-:
Add nitric acid. This will acidify the solution and make sure there are no carbonate ions (no gas should form).
Add silver nitrate. A precipitation reaction will occur and a precipitate will form. The colour will tell you what precipitate you will get:

Test for Chloride, Cl-:
Adding nitric acid and silver nitrate will form a white precipitate of silver chloride.
Example:
Balanced: NaCl (aq) + AgNO3 (aq) → AgCl (s) + NaNO3 (aq)
Ionic: Cl- (aq) + Ag+ (aq) → AgCl (s)

Test for Bromide, Br-:
Adding nitric acid and silver nitrate will form a cream precipitate of silver bromide.
Example:
Balanced: NaBr (aq) + AgNO3 (aq) → AgBr (s) + NaNO3 (aq)
Ionic: Br- (aq) + Ag+ (aq) → AgBr (s)

Test for Iodide, I-:
Adding nitric acid and silver nitrate will form a yellow precipitate of silver iodide.
Example:
Balanced: NaI (aq) + AgNO3 (aq) → AgI (s) + NaNO3 (aq)
Ionic: I- (aq) + Ag+ (aq) → AgI (s)
CH242: Core Practical: Investigating Ions
You are presented with 3 ionic substances and asked to find out the names of the compounds. You carry out the following tests on each substance:
- Test 1: Flame test for cations: Add the substance to a wire loop and add to a blue Bunsen flame.
- Test 2: Precipitate test for cations: Add sodium hydroxide to the solution and note the colour of any precipitate.
- Test 3: Test for halides: Add nitric acid and a few drops of silver nitrate. Note any changes.
- Test 4: Test for sulfates: Add hydrochloric acid and a few drops of barium chloride. Note any changes.
- Test 5: Test for carbonates: Add any acid and bubble any gas through limewater.
The results of the test are below:
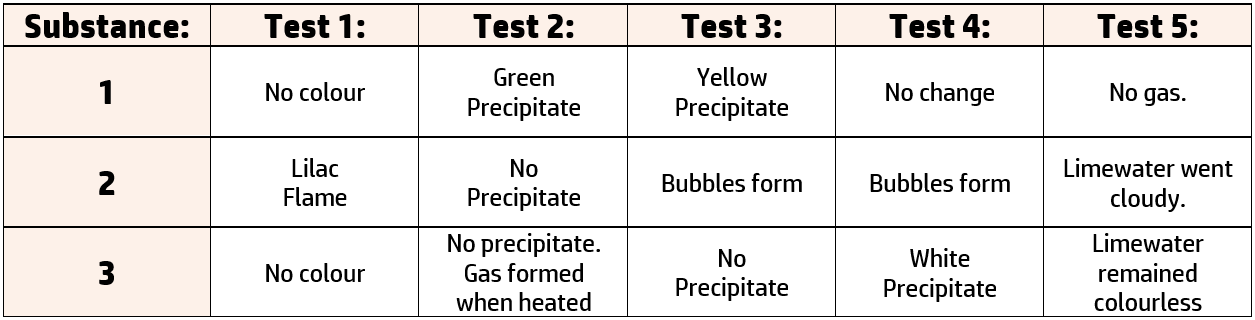
You are asked to name the substances using the results from these tests. To do this you need to know the tests:
Substance 1: Iron (II) chloride
Test 2 produced a green precipitate when sodium hydroxide was added. A green precipitate (CH239) tells us our cation is iron (II).
Test 3 produced a yellow precipitate when silver nitrate was added. This is the test for halides (CH241), and iodide ions are the only halides that turn yellow, so we have an iodide.
Name: Iron (II) iodide, FeI2

IRON (II)

IODIDE
Substance 2: Potassium carbonate
Test 1 produced a lilac flame (CH238), so straight away we know this is potassium.
A gas was formed – this could either be ammonia or carbon dioxide. Test five (CH241) shows that limewater went cloudy, so it must be a carbonate.
Name: Potassium carbonate, K2CO3

POTASSIUM

CARBONATE
Substance 3: Ammonium* sulfate
Test 1 and 2 did not produce a flame or a precipitate, but a gas was formed. Limewater didn’t go cloudy, so it is not a carbonate. This leaves us with ammonium – but this is not a confirmatory test. We would need to add the gas to damp red litmus paper (CH240) for a confirmatory test.
Test 4 produced a white precipitate. Lots of test produce white precipitates, so we have to be sure of the test. Here, barium chloride has been added, so we know this is the test for sulfates (CH241).
Name: Ammonium (?) sulfate, (NH4)2SO4
AMMONIUM (?)

SULFATE
CH243: Explain the advantages of instrumental analysis
It is better to use machines, such as flame photometry (see CH244), instead of flame tests because:
- They are more sensitive. (They can detect much smaller amounts)
- They are more accurate. (They give values closer to the true values)
- They give results faster.
CH244: Explain why flame photometry is more accurate than flame tests
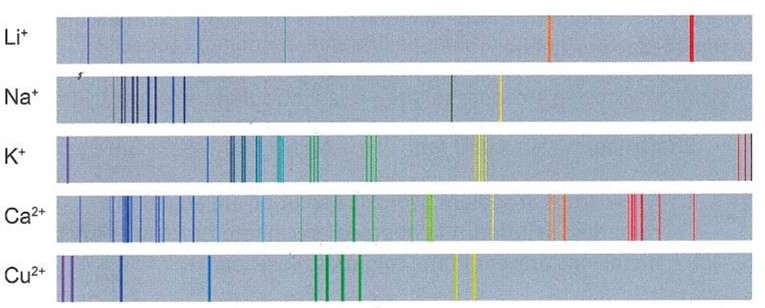
Flame photometers measure light intensity of flame colours and determines the concentration of a metal ion in solution.
This then produces an emission spectrum, such as the ones on the right. Each ion produces a unique spectrum, making them more accurate than flame tests.
CH264: Describe what a nanoparticle is
Nanoparticles are chemicals that range from 1 to 100 nanometres (nm) in size.1nm is the same as 0.000,000,001m (1x10-9m)
You may be asked to compare the size of a nanoparticle to the size of another particle.
Example: A nanoparticle of titanium dioxide has a diameter of 240nm. A titanium atom has a diameter of 0.15nm. Calculate how many times bigger the nanoparticle is compared to a titanium atom.
To answer this question, you need to divide the diameter of the nanoparticle by the diameter of the titanium atom: 240nm / 0.15nm = 1600 times bigger.
They may also ask you to convert between nanometres and metres.
Example: A nanoparticle of titanium dioxide has a size of 240nm. Calculate the size of the particle in nanometres.
To answer this question, you need to remember that 1nm is 1x10-9m.
So 240nm is 240x10-9m. This can be simplified → 24x10-8m → 2.4x10-7m.
CH265: Calculate the surface area-to-volume ratio of a nanoparticle
Example: A cube shaped nanoparticle has sides of 20nm. Calculate its surface area-to-volume ratio.
Step 1: Work out the area of one side of the cube.
- Area = Height x Width = 20nm x 20nm = 400nm2
Step 2: Work out the total surface area of the cube.
- Surface Area (of a cube) = Area x 6 = 400nm2 x 6 = 2400nm2
Step 3: Work out the total volume of the cube.
- Volume = Height x Width x Depth = 20nm x 20nm x 20nm = 8000nm3
Step 3: Work out the surface area-to-volume ratio of the cube.
- Surface area-to-volume ratio = surface area ÷ volume = 2400nm2 ÷ 8000nm3 = 0.3 : 1
CH266: Explain why nanoparticles can be used in sunscreens and as catalysts

Sunscreen
When used as a sunscreen, the particles are extremely small, making the titanium dioxide transparent. This means that it doesn’t leave white marks.
The titanium dioxide in sunscreen absorbs ultraviolet rays. This helps to prevent skin cancer (which ultraviolet rays can cause).
It is also a nanoparticulate, giving it different properties to when it is used in bulk.
Catalysts
Nanoparticles have a large surface area-to-volume ratio, which makes them useful as catalysts.
They can be used as catalysts in detergent and self-cleaning windows because they help to catalyse the breakdown of dirt, keeping the clothes / windows clean.

CH267: Explain the concerns over nanoparticles
There are a few concerns about using nanoparticles:
- The small size allows them to be breathed in / absorbed into body.
- Once in the body, they may catalyse harmful reactions, harming our health.
- Nanoparticles are a new technology, so we don’t know the long term impact of using them.
CH268: Explain the formation and properties of ceramics
Ceramics are hard non-metal compounds (giant covalent) that are unaffected by heat.
Ceramics have the following physical properties:
- Chemically Unreactive
- Hard/Stiff
- Brittle (Easily shattered)
- Poor conductors of heat / electricity.

Glass Ceramics:
Glass is made by melting sand and allowing it to cool/solidify.
They don’t form a regular pattern and don’t form crystals, making them transparent.
Clay Ceramics:
Clay ceramics are made by clay being heated to high temperatures, forming crystals.
This makes them opaque.

CH269: Explain how composite materials are useful
Composite materials are mixtures of more than one material, combined together to give it different properties.
Most composites have a reinforcement part and a matrix part.
- Matrix: The substance that binds the reinforcement material together
- Reinforcement: The substance bound together by the matrix.
| Material: | Description: | Reinforcement: | Matrix: | Properties: |
|---|---|---|---|---|
| Concrete | Sand, Cement, Aggregate and water are mixed. When it sets, chemical reactions occur and bond the components together | Sand and aggregate | Cement | Strong Good insulator |
| Fibreglass | Thin glass fibres and a polymer resin are reacted together | Glass fibres (Brittle, low density and high tensile strength) | Polymer Resin (Hardwearing but not strong) | Lightweight Strong Tough |
| Reinforced Steel | Concrete can be compressed, but when used in beams it can crack. Steel is used to reinforce the concrete. | Steel (High tensile strength and compressive strength) | Concrete (Dense, strong, and cheap) | Strong in tension & compression Cheaper than just steel |
| Laminates | Wood is stronger along the grain than across it. Laminates consist of layers of wood at right angles to each other. | Cellulose fibres | Lignin Polymer | High tensile strength |
CH270: Suggest uses for different materials based on their physical and chemical properties
In the exam, you will be asked to suggest which material to use for a certain use. To do this, you must know the main properties of each of the following materials:
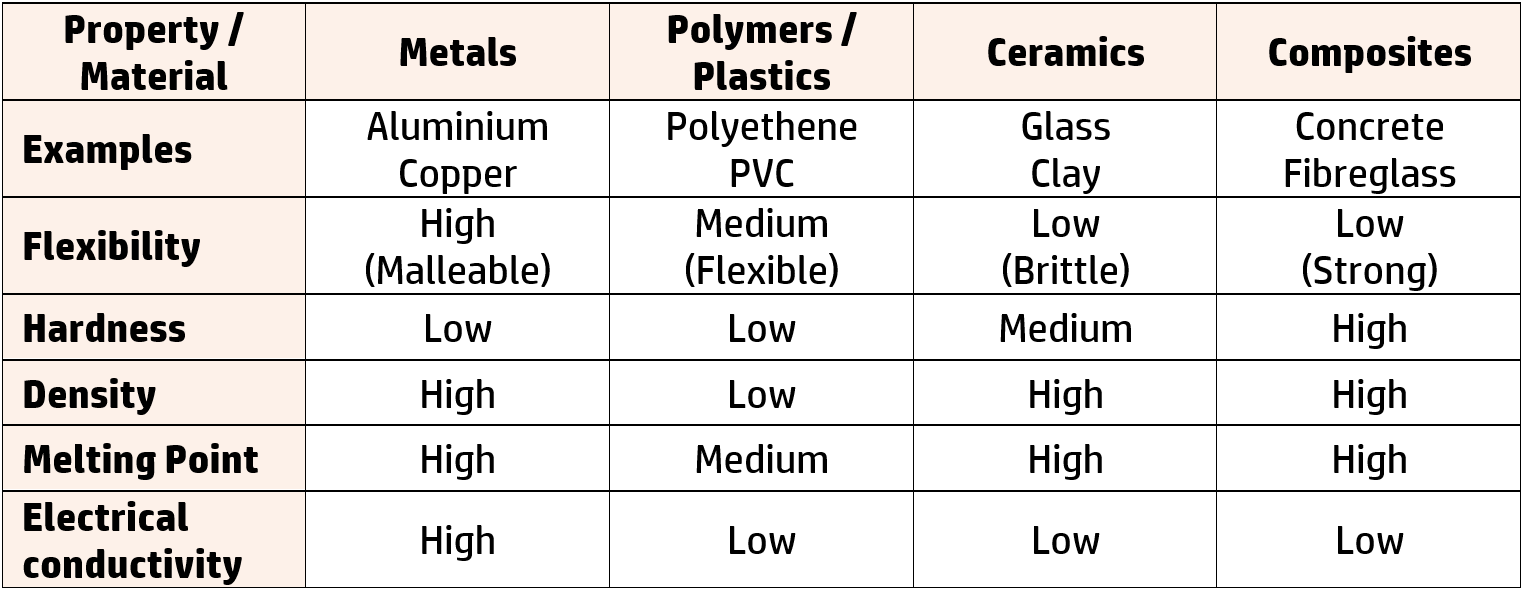
In materials questions, you are usually given a table of properties and asked to decide which material is more suitable for a particular use.
Example: For example, if choosing between glass and polymers for bottles, you should identify the properties that differ and select those that are beneficial.
The most useful properties for a bottle are flexibility and low density, so you would pick those properties, select a material and link the use to the properties:
- Polymers are more flexible than glass (1) meaning it won’t break/shatter (1).
- Polymers are less dense than glass (1) meaning it is easier to transport lots of them (1).
