SCIENTIFIC SKILLS: IONIC EQUATIONS


A GCSE chemistry revision page on ionic equations, explaining how to write full and net ionic equations, showing ion formation in solution, cancelling spectator ions, examples with precipitation and neutralisation reactions, and practice guidance.
CH18: Writing Ionic Equations (H)
To write ionic equations, you must be able to work out the charge of your ions. Once you can do that, it is fairly straight forward.
The key is that you only split up aqueous ionic substances – the rest stays the same.
Worked Example: Write the ionic equation for the following chemical equation:

Cl2 (g) + 2KBr (aq) → 2KCl (aq) + Br2 (l)
Step 1: Split aqueous ionic compounds into ions. Only substances labelled (aq) are split into ions, so we keep Cl2 (g) and Br2 (l) the same.
- Rule 1: Group 1 metals always form +1 ions. So, both KBr and KCl contain K⁺ ions.
- Rule 2: Halogens form −1 ions. Bromine and chlorine are both in Group 7, so KBr contains Br⁻ ions and KCl contains Cl⁻ ions.
Step 2: Put the ions into the equation. Don't forget to include balancing, so we have 2 x K⁺ ions and 2 x the halide ions:
Cl2 (g) + 2K⁺ (aq) + 2Br⁻ (aq) → 2K⁺ + 2Cl⁻ (aq) + Br2 (l)
Step 3: Remove the spectator ions. These are the ions that are the same on both sides - in this case, the 2K⁺ ions:
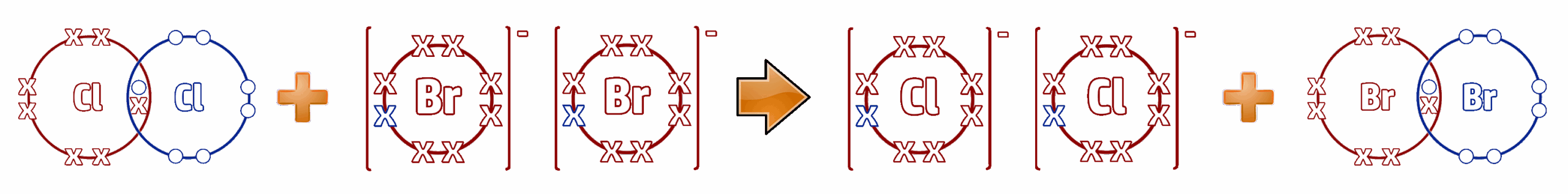
Cl2 (g) + 2Br⁻ (aq) → 2Cl⁻ (aq) + Br2 (l)
CH19: Write ionic equations for neutralisation reactions
Worked Example: Write the ionic equation for the following neutralisation reaction:
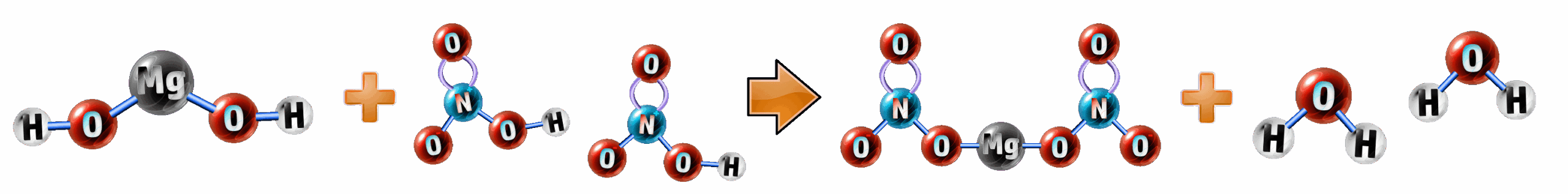
Mg(OH)2 (aq) + 2HNO3 (aq) → Mg(NO3)2 (aq) + 2H2O (l)
Step 1: Split aqueous ionic compounds into ions. Only substances labelled (aq) are split into ions, so we keep H2O (l) the same.
- Rule 1: Group 2 metals always form +2 ions. So, both Mg(OH)2 and Mg(NO3)2 contain Mg2+ ions.
- Rule 2: You need to learn the charge for the following ions: Nitrate ions are NO3-, hydrogen ions are H+ and hydroxide ions are OH-.
Step 2: Put the ions into the equation. Don't forget to include balancing, so we have 2 x H⁺ ions and 2 x NO3- ions:
Mg2+ (aq) + 2OH- (aq) + 2H+ (aq) + NO3- (aq) → Mg2+ (aq) + 2NO3- (aq) + 2H2O (l)
Step 3: Remove the spectator ions. These are the ions that are the same on both sides - in this case, the Mg2+ and NO3- ions. You can also simplify this as all of the numbers are 2:
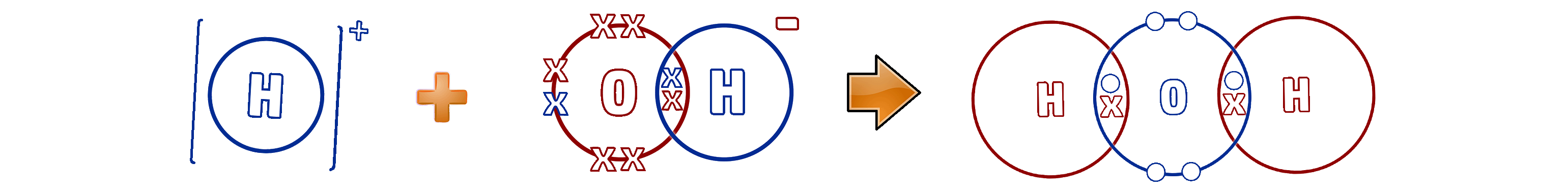
H+ (aq) + OH- (aq) → H2O (l)
CH20: Write ionic equations for precipitation reactions
Worked Example: Write the ionic equation for the formation of copper hydroxide from copper chloride and sodium hydroxide:

CuCl2 (aq) + 2NaOH (aq) → Cu(OH)2 (s) + 2NaCl (aq)
Step 1: Split aqueous ionic compounds into ions. Only substances labelled (aq) are split into ions, so we keep Cu(OH)2 (s) the same.
- Rule 1: Halogens form −1 ions. Chlorine is in Group 7, so CuCl2 and NaCl both contain Cl⁻ ions.
- Rule 2: Copper is a transition metal. The formula tells us the charge. There are 2 chloride ions (Cl⁻) and each chloride has a charge of −1. Therefore, the total negative charge is −2. To balance that, the copper must be a Cu²⁺ ion.
- Rule 3: Sodium is in group 1 so forms a Na+ ion and the hydroxide is an OH⁻ ion.
Step 2: Put the ions into the equation. Don't forget to include balancing, so we have 2 x Cl⁻, 2 x Na+ and 2 x OH- ions:
Cu²⁺ (aq) + 2Cl⁻ (aq) + 2Na+ (aq) + 2OH- (aq) → Cu(OH)2 (s) + 2Na+ (aq) + 2Cl⁻ (aq)
Step 3: Remove the spectator ions. These are the ions that are the same on both sides - in this case, the 2Na⁺ and 2Cl⁻ ions:
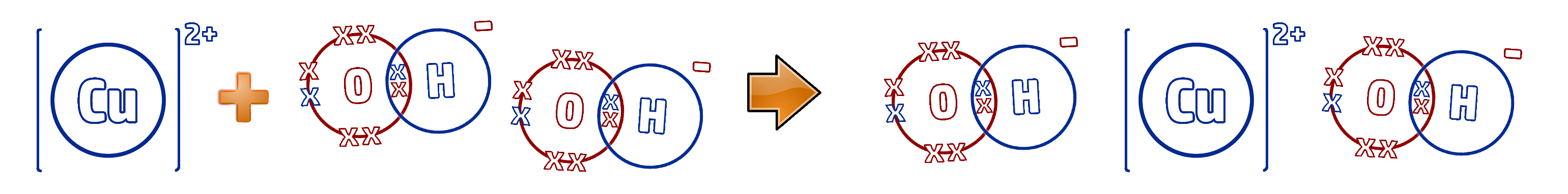
Cu²⁺ (aq) + 2OH- (aq) → Cu(OH)2 (s)
CH21: Write ionic equations for displacement reactions
Worked Example: Write the ionic equation for the following displacement reaction between sodium and calcium chloride:

2Na (s) + CaCl2 (aq) → 2NaCl (aq) + Ca (s)
Step 1: Split aqueous ionic compounds into ions. Only substances labelled (aq) are split into ions, so we keep 2Na (s) and Ca (s) the same.
- Rule 1: Group 1 metals always form +1 ions. So, NaCl contains Na⁺ ions.
- Rule 2: Group 2 metals always form +2 ions. So, CaCl2 contains Ca2+ ions.
- Rule 3: Halogens form −1 ions. Chlorine is in Group 7, so CaCl2 and NaCl both contain Cl⁻ ions.
Step 2: Put the ions into the equation. Don't forget to include balancing, so we have 2 x Na⁺ ions and 2 x the halide ions:
2Na (s) + Ca2+ (aq) + 2Cl- (aq) → 2Na⁺ (aq) + 2Cl⁻ (aq) + Ca (s)
Step 3: Remove the spectator ions. These are the ions that are the same on both sides - in this case, the 2Cl- ions:

2Na (s) + Ca2+ (aq) → 2Na⁺ (aq) + Ca (s)
