HYDROCARBONS AND COMBUSTION

A GCSE chemistry revision page on hydrocarbons covering alkanes and alkenes, combustion reactions, cracking to produce smaller alkanes and alkenes, and how these reactions relate to fuel properties and chemical behaviour with examples and explanations.
CH211: Describe what a hydrocarbon is
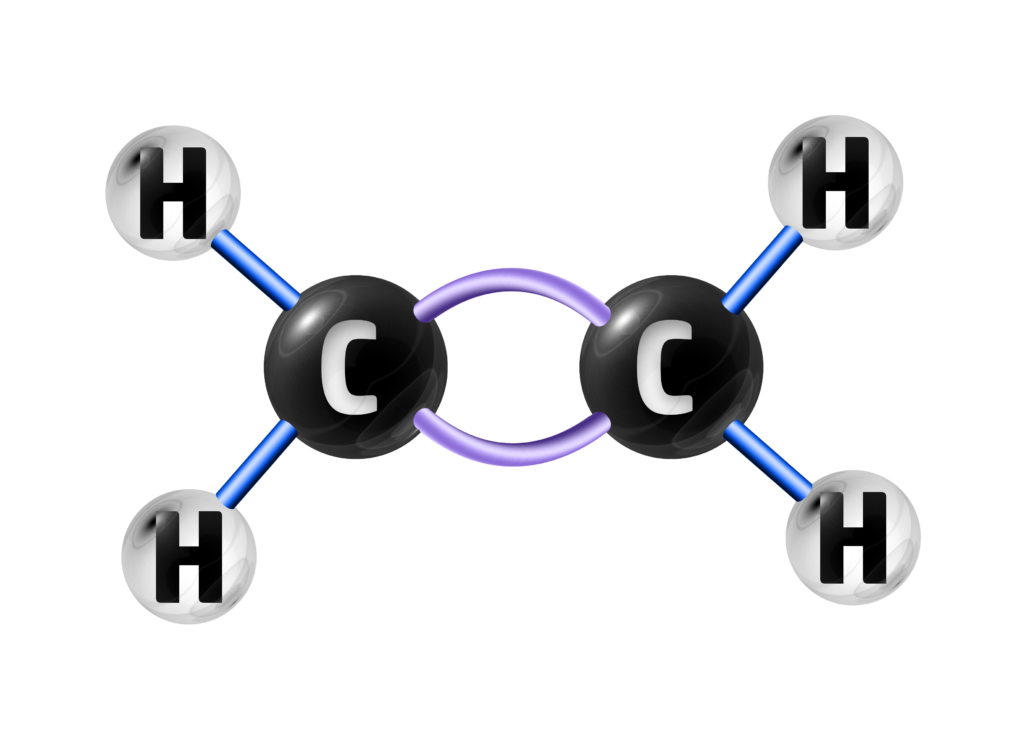
A hydrocarbon is a compound containing hydrogen and carbon atoms only.
In the three chemicals below, A and C are hydrocarbons because they ONLY contain hydrogen and carbon.
B is not a hydrocarbon because it also contains oxygen.
A
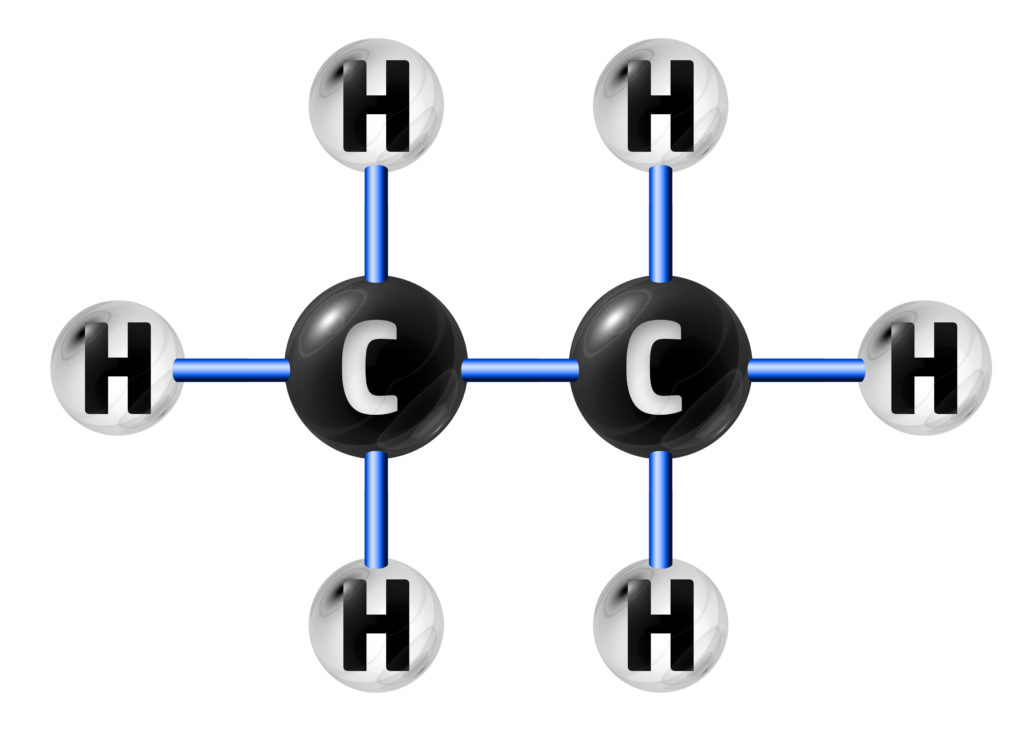
HYDROCARBON
B
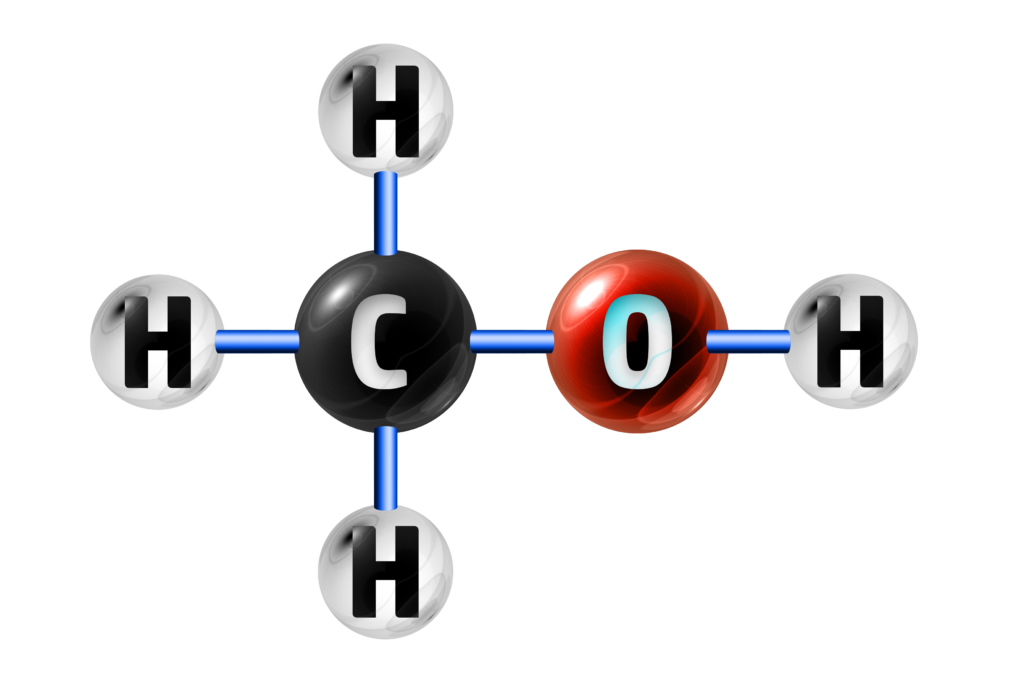
NOT A HYDROCARBON
C
HYDROCARBON
CH212: Describe what crude oil is made up of
Crude oil is a mixture of hydrocarbons (See CH211).
Carbon atoms have a valency of 4, meaning they can make 4 covalent bonds.
This high valency leads to many hydrocarbons being formed, ranging from 1 carbon to hundreds of them – which can be in the form of chains and rings.
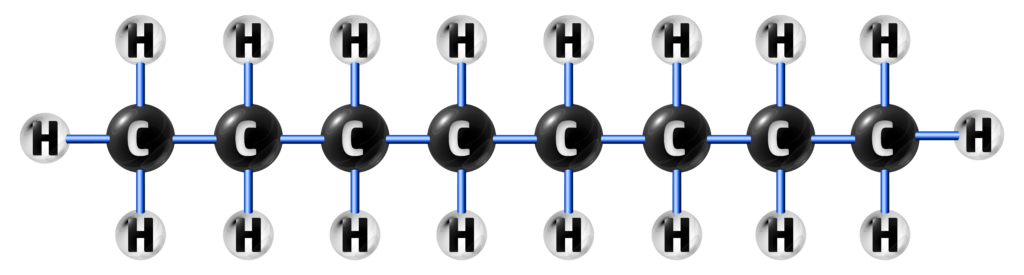
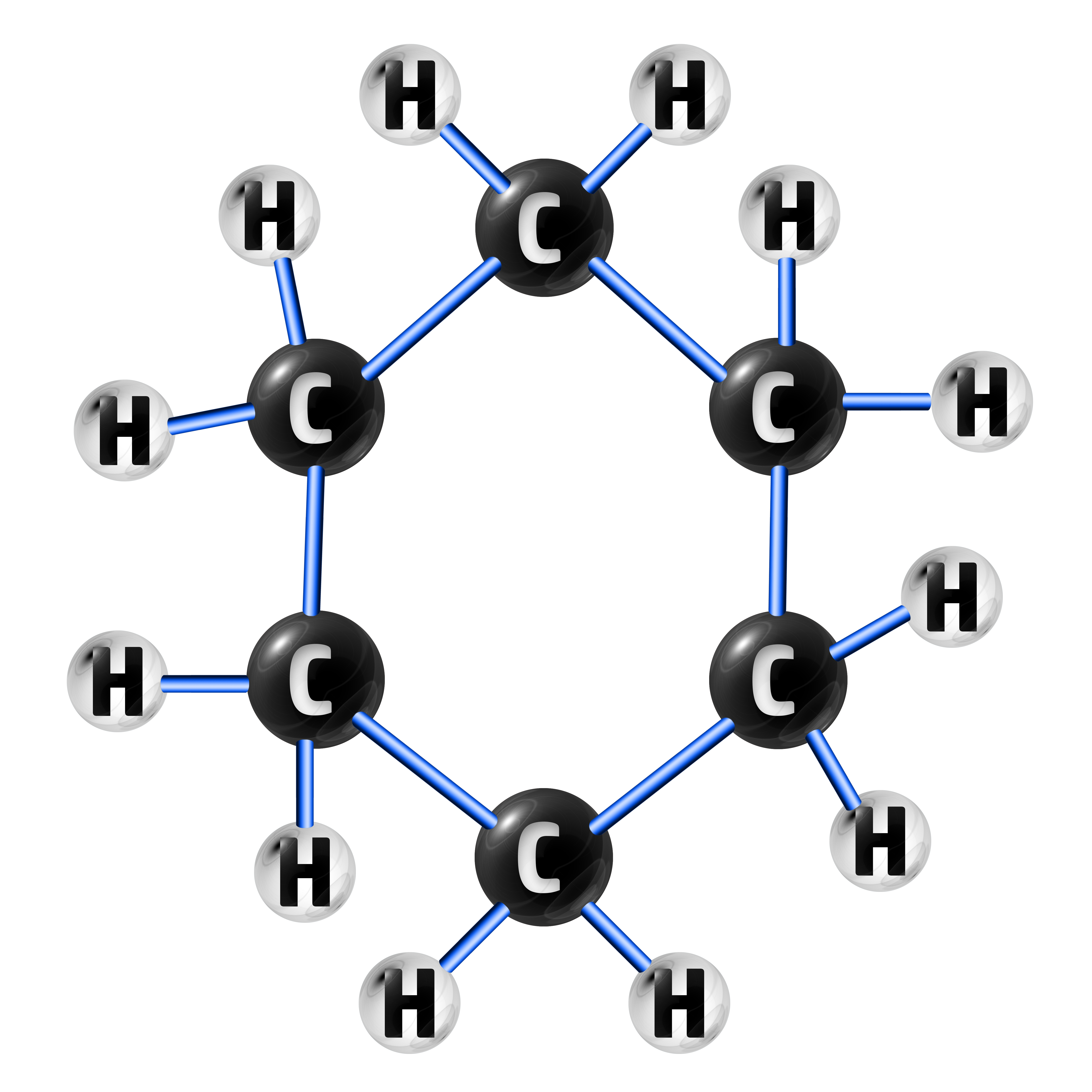
CH213: Describe what a homologous series is
A homologous series is any group of chemicals that have:
- similar chemical properties,
- a gradual change in physical properties (boiling points and melting points),
- the same general formula,
- Each member of the series goes up by a CH2 each time.
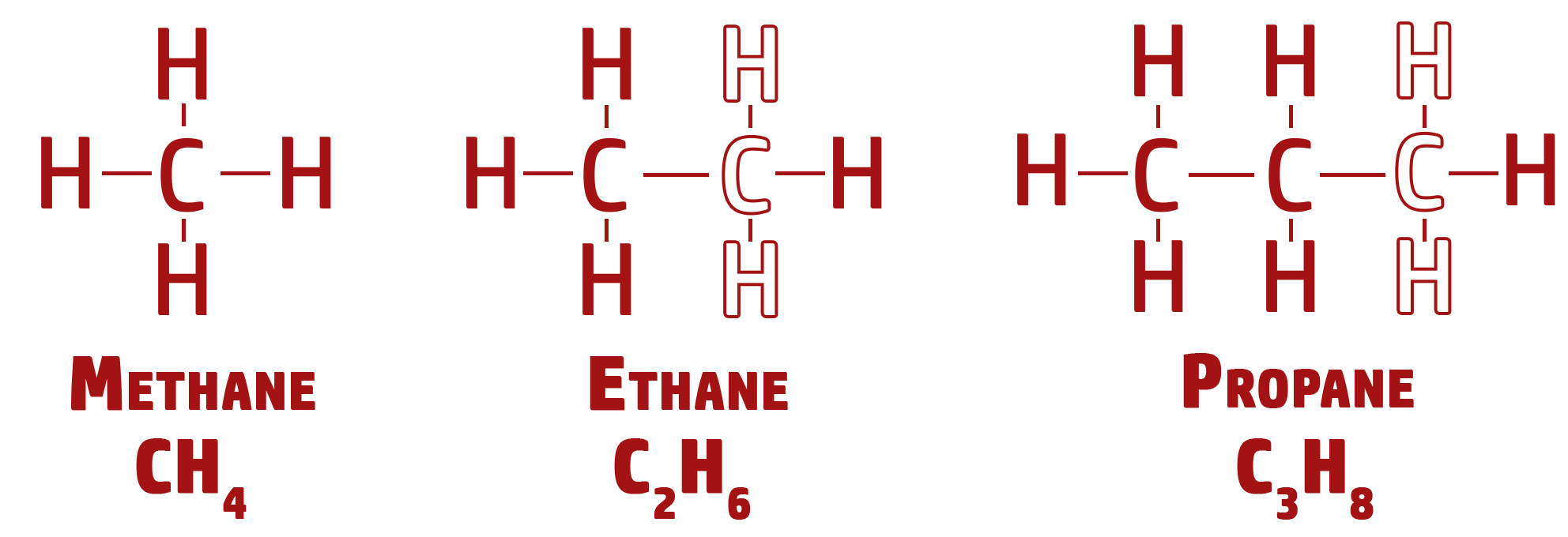
These are called homologues series because they have the same general formula which in this case is CnH2n+2.
- For example, when n=2, the molecule is C2H(2x2)+2 = C2H6.
- This formula works by adding in a Carbon and 2 Hydrogens (CH2) each time.
CH214: Describe what a finite resource is
Crude oil is a finite resource which is the term given to something that is either not being made any more or it is being made very slowly.
This makes crude oil non-renewable (cannot be replaced when it is used up).

CH215: Explain how crude oil is separated into fractions
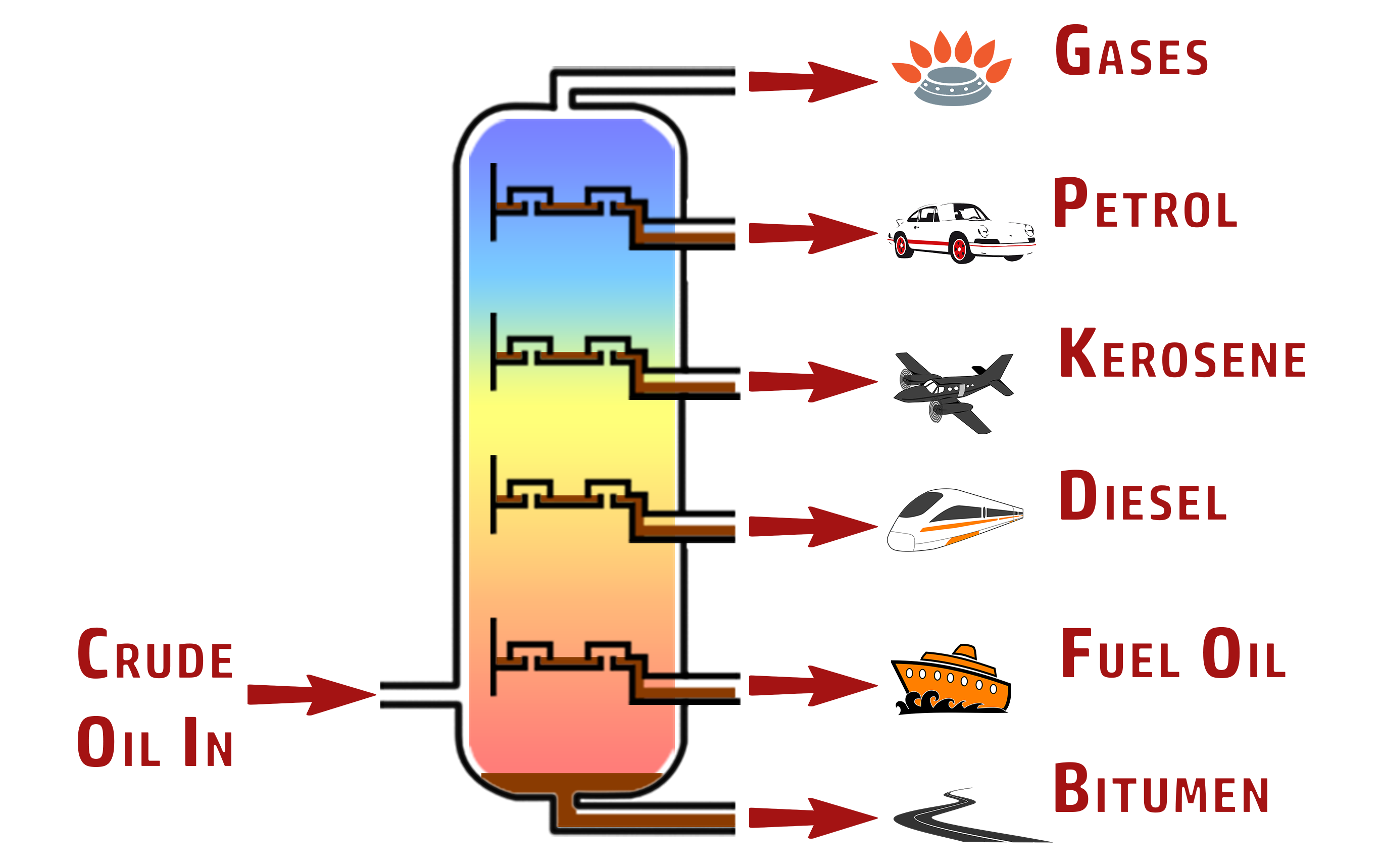
Crude oil contains many different fuels, with different uses. We cannot just put crude oil into our cars, for example – it needs to be separated into the different fractions.
This is done using fractional distillation – which means separating into their fractions using their boiling points – in the following way:
- The crude oil is vapourised and added to the column
- The fractionating column is hottest at the bottom and coolest at the top.
- The vapours rise throughout the column until they fall below their boiling point – they then condense.
- The liquid falls into the tray and is collected.
The rest of the hydrocarbons will move up the column, and the process continues until you are left with just the gases at the top.
CH216: Identify the names and uses of the fractions of crude oil
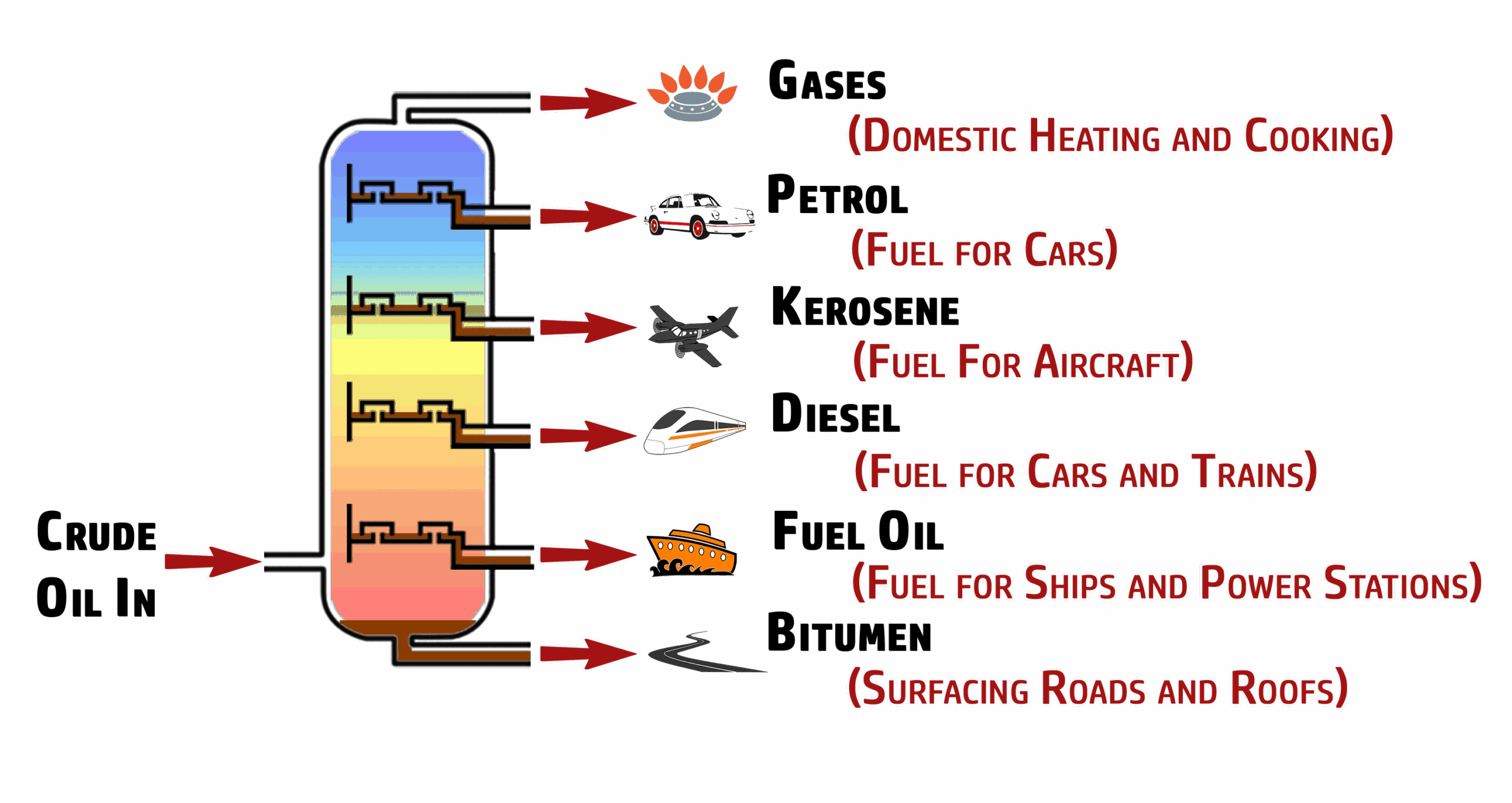
CH217: Compare the properties of the fractions of crude oil
The gases at the top of the column are the easiest to ignite, have the lowest boiling points, have the smallest chain lengths (less carbon atoms) and are the least viscous (flow more easily).
The bitumen at the bottom of the column is the hardest to ignite, has the highest boiling points, has the largest chain lengths (more carbon atoms) and are the most viscous (thick and doesn’t flow easily)
CH218: Describe the process of catalytic cracking
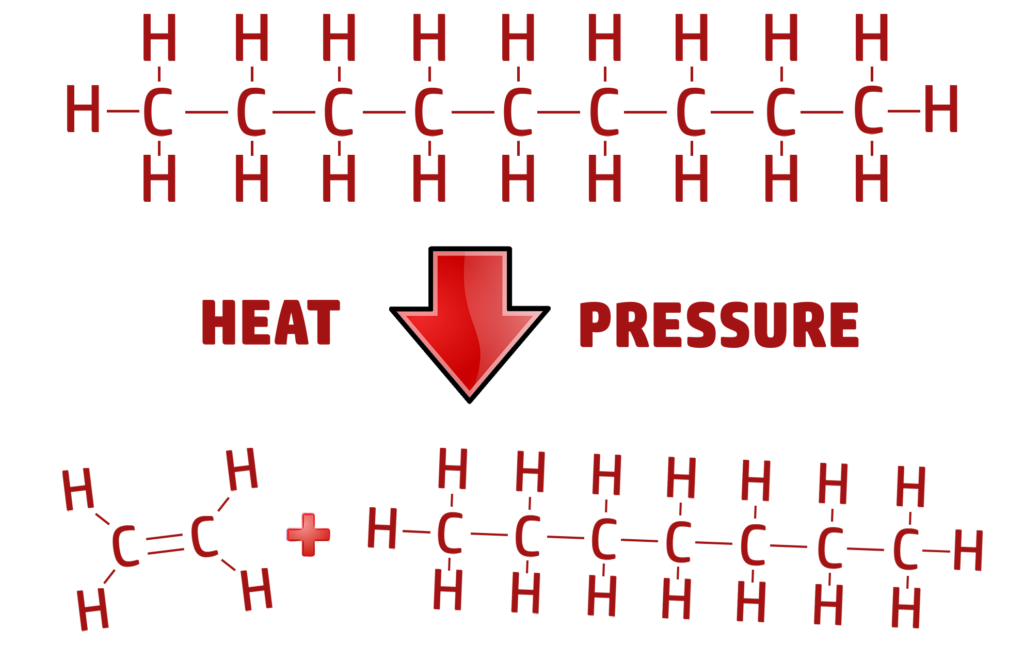
When a large-chained hydrocarbon (alkane) is put under heat and pressure with a catalyst, they are broken down into short chained hydrocarbons called alkanes and alkenes.
CH219: Explain the importance of catalytic cracking
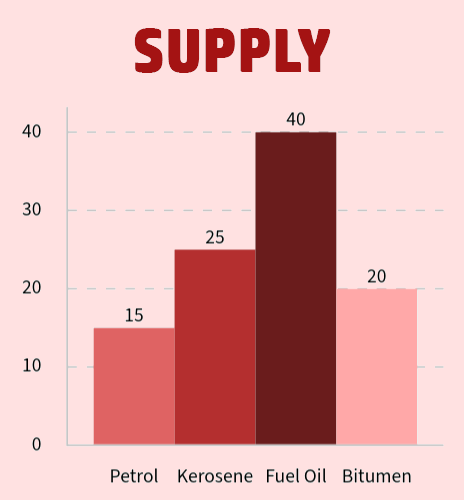
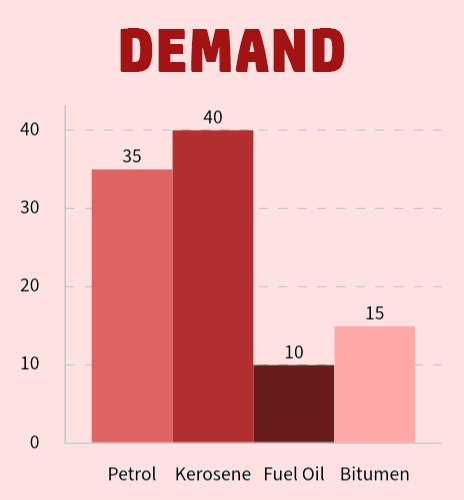
On the right, you can see that we have a large demand for petrol and kerosene, but the supply for each is not enough.
- We use cracking to break down the long changed hydrocarbons such as fuel oil (supply: 40%; demand: 10%) that we have too much of...
- ...into shorter chained hydrocarbons like petrol that we don't have enough of (supply 15%; demand 35%).
CH220: Describe what complete combustion is
Complete combustion is where a fuel reacts with lots of oxygen, giving out lots of energy as well as producing carbon dioxide and water.
Writing word and balanced chemical equations for this is easy. Just remember that oxygen is O2, carbon dioxide is CO2 and water is H2O and you’re good to go.
Example: Write the word equation for the complete combustion of methane.
Methane + Oxygen → Carbon Dioxide + Water
Example 2: Write the balanced equation for the complete combustion of methane., CH4.

CH4+ 2O2 → CO2 + 2H2O
CH221: Describe what incomplete combustion is
Incomplete combustion occurs when there is not enough oxygen. Water is still formed, but instead of just carbon dioxide, carbon monoxide and carbon (soot) are produced as well.
Possible products of incomplete combustion:

Soot
C
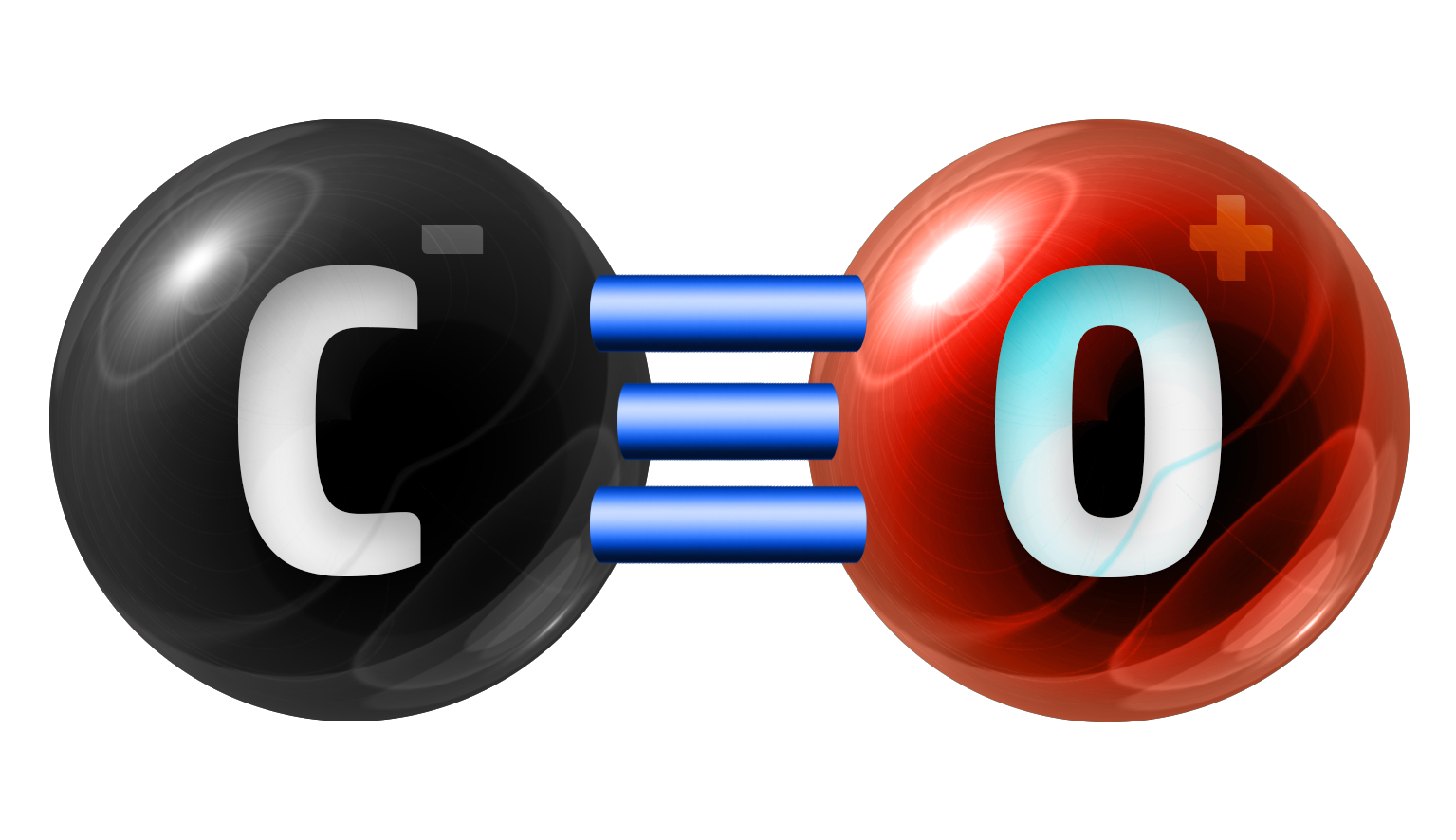
Carbon Monoxide
CO

Carbon Dioxide
CO2
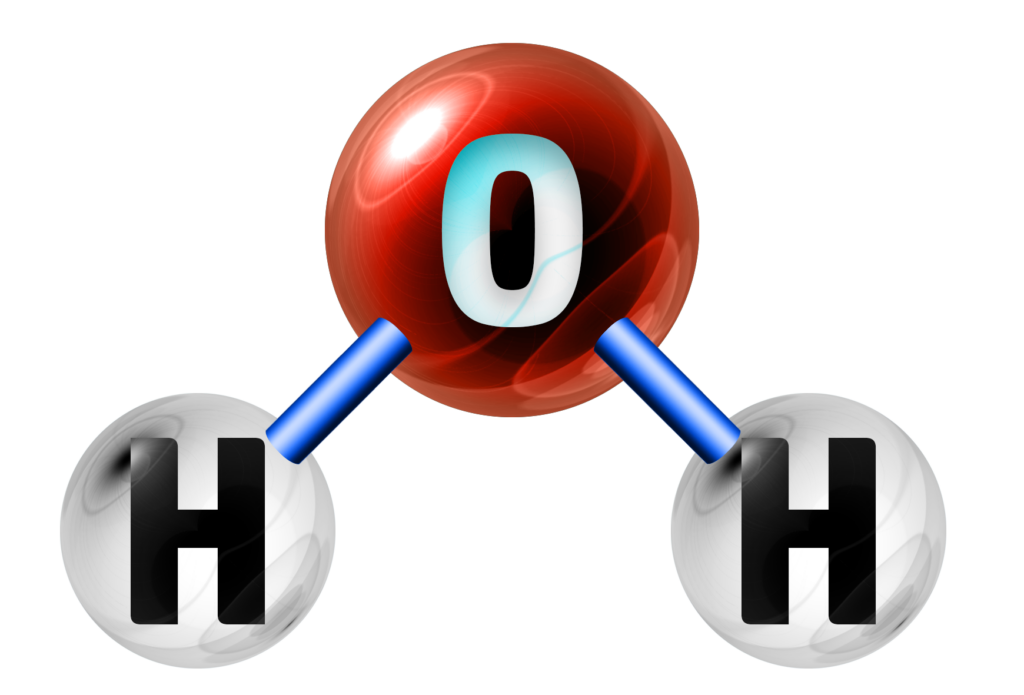
Water
H2O
CH222: Explain the dangers of carbon monoxide

Carbon monoxide is a toxic gas – which means it is poisonous. This is because:
- It binds to the haemoglobin in the red blood cells, taking up space needed by the oxygen.
- This reduces the amount of oxygen reaching the cells and lowers the amount of respiration occurring.
- This makes people sleepy and can cause them to become unconscious – and can lead to death.
CH223: Describe the problems of soot

Soot blocks pipes carrying away waste gases from a boiler.
It can cause breathing problems if it collects in the lungs.
It can make buildings dirty.
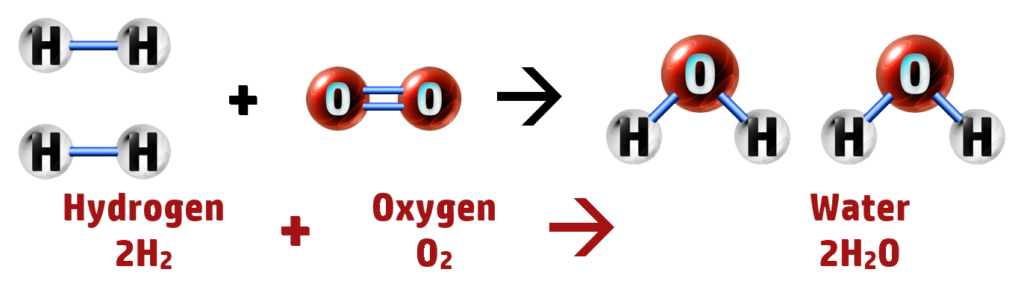
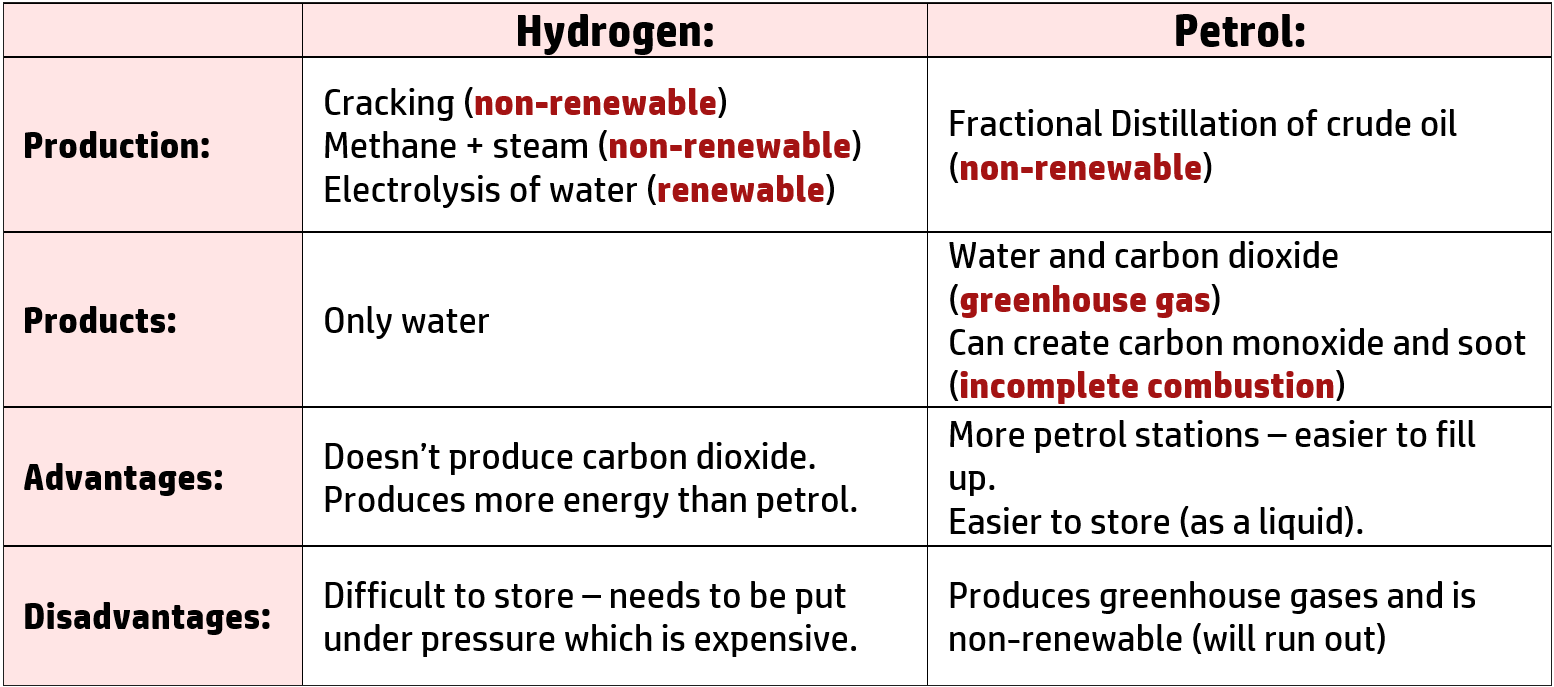
CH224: Evaluate using hydrogen as a fuel instead of petrol
Hydrogen can be used as a fuel instead of fossil fuels. It reacts with oxygen to form water: