SCIENTIFIC SKILLS: BALANCING EQUATIONS


A GCSE chemistry revision page on balancing equations, explaining how to balance chemical equations step-by-step, including examples with molecules, ions and reactions such as combustion and displacement.
CH1: Recall the formulae of simple molecules
Diatomic Elements:
There are several diatomic elements (elements that go around in pairs) you need to remember for the GCSE.
A great way to remember them is the saying: Have No Fear Of Ice Cold Beer!
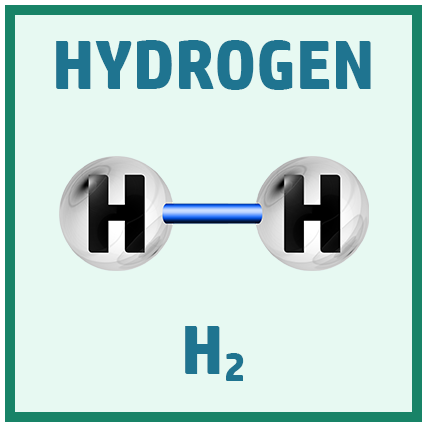

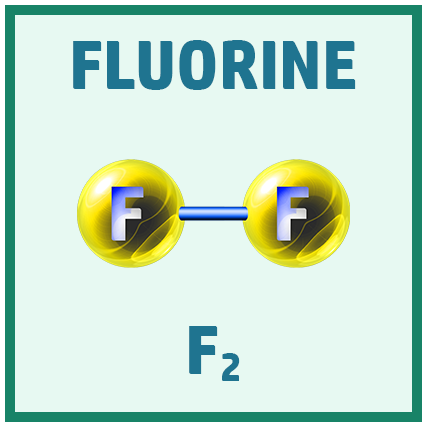

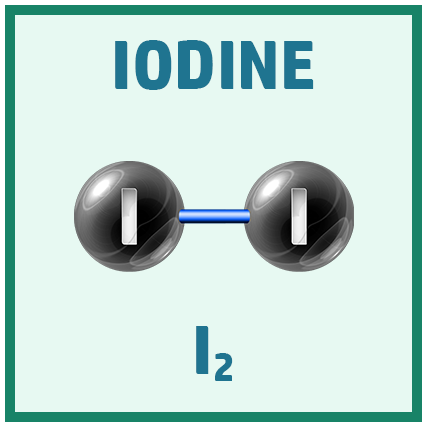
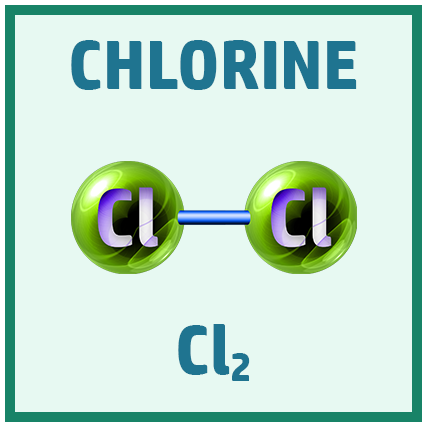

Acids:
There are three main acids that you should know to help with the chemical change topic:
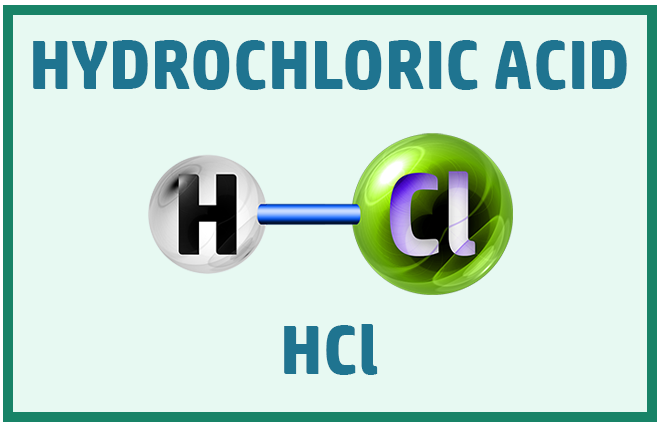
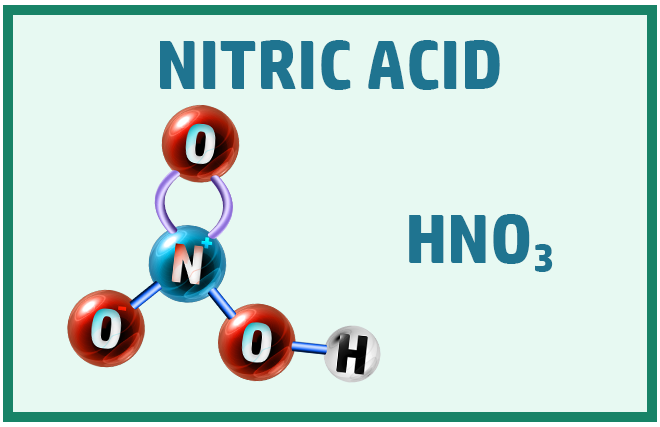
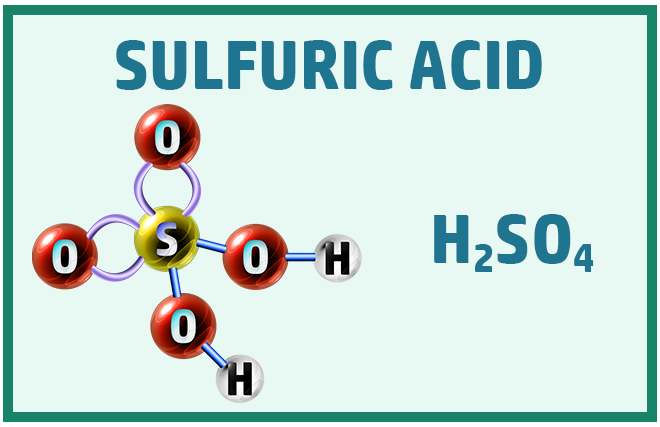
Covalent Substances:
Other common covalent molecules you should know are:
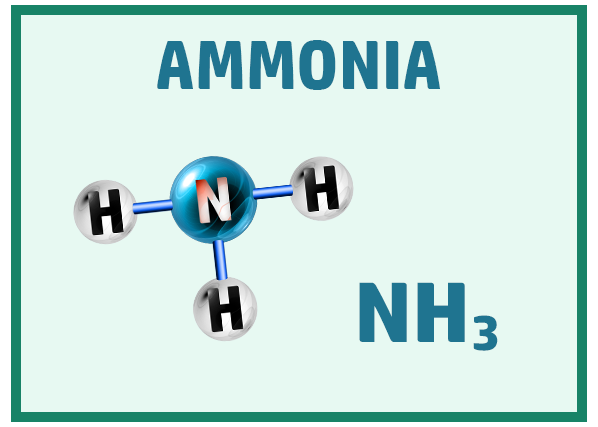
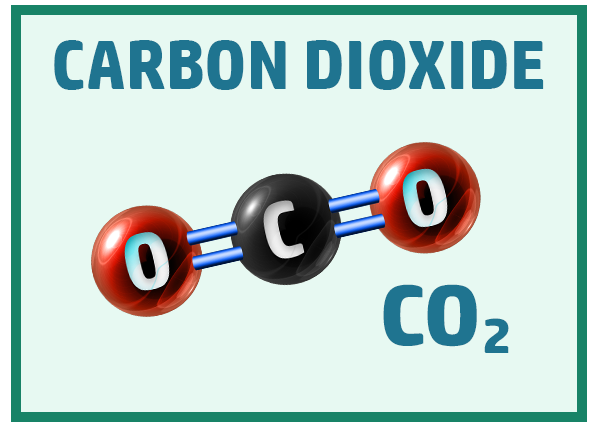

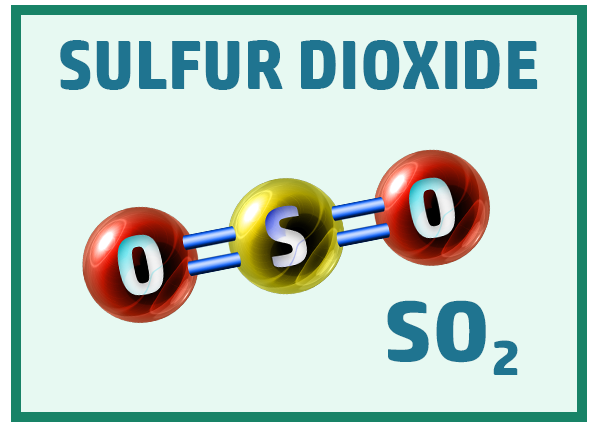
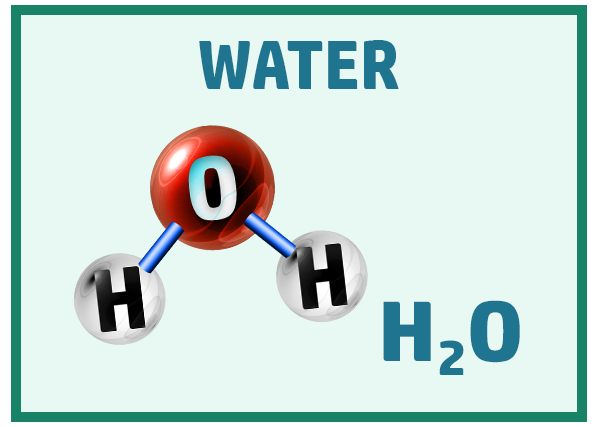
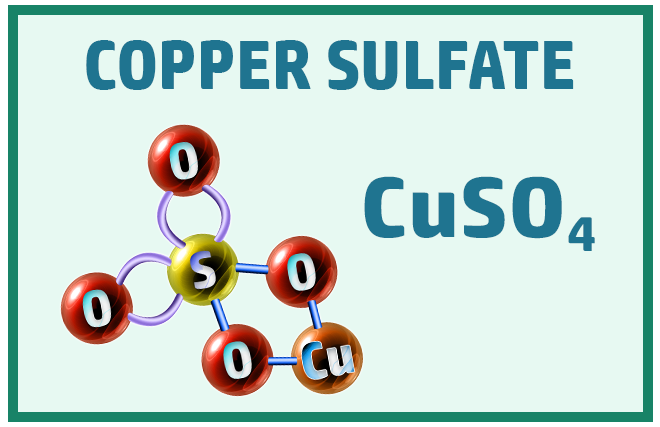
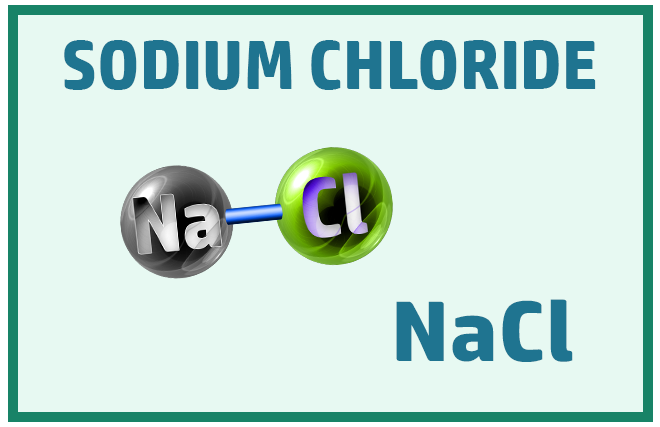
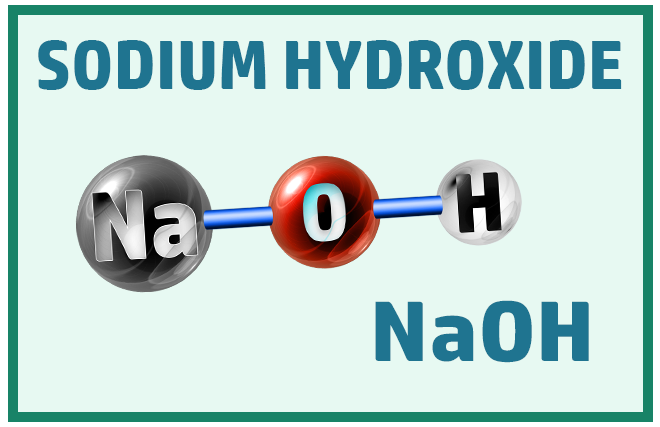
Ionic Substances:
Other common ionic molecules you should know are:
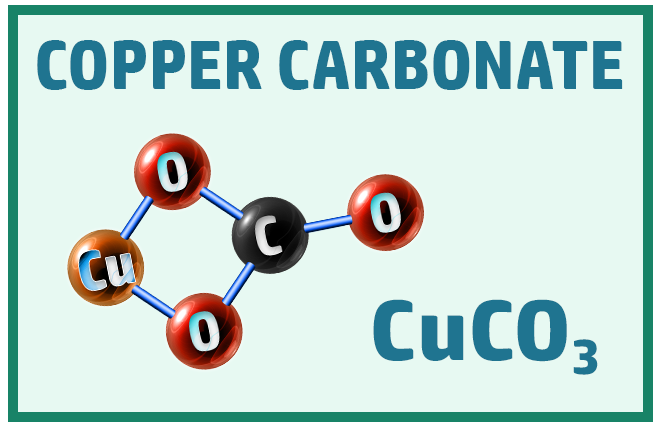
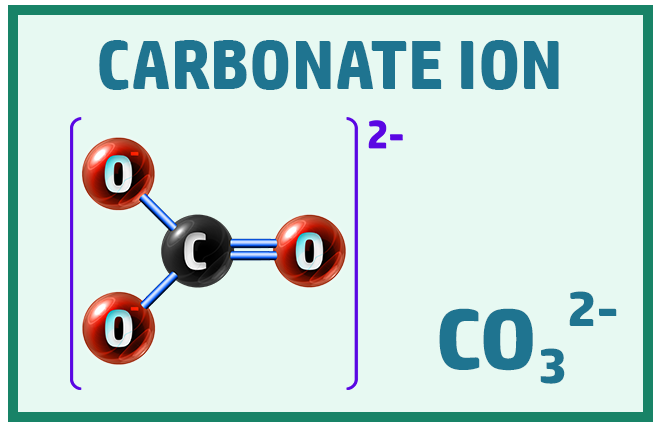
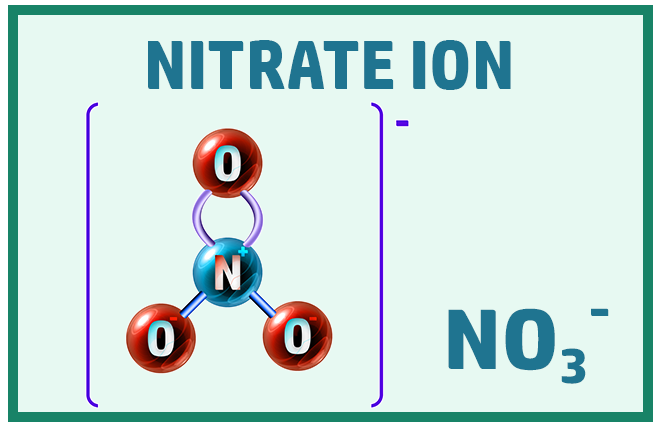
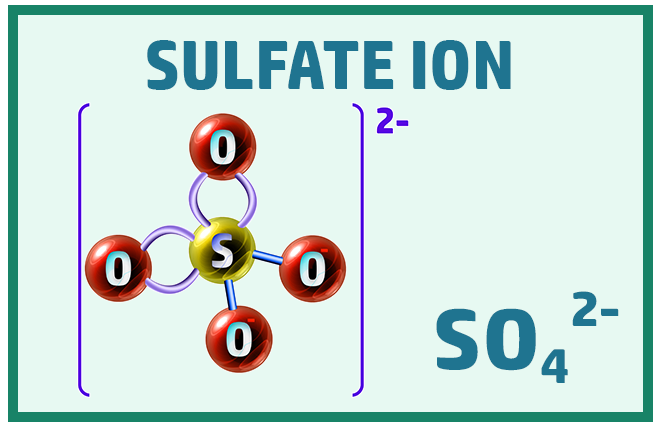
Polyatomic Ions:
Finally, there are some ions that will really help when working out the formula of ionic compounds:

CH9: Balance simple chemical equations
Balancing is all about making sure the number of each element is the same on both sides of the arrow.
The main rules:
- You can only put a number at the beginning of a substance.
- That number multiplies the whole substance.
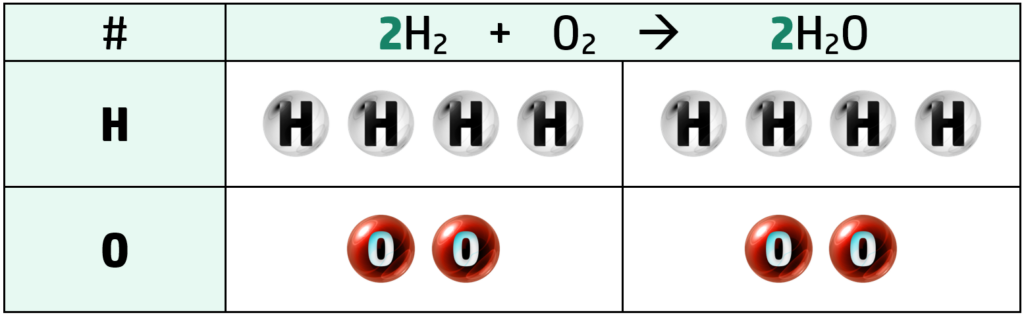
Example: Write the balanced equation for the reaction between hydrogen, H2, and oxygen, O2 to form water, H2O.
Step 1: Write the unbalanced equation and count up the number of each element on either side of the arrow.
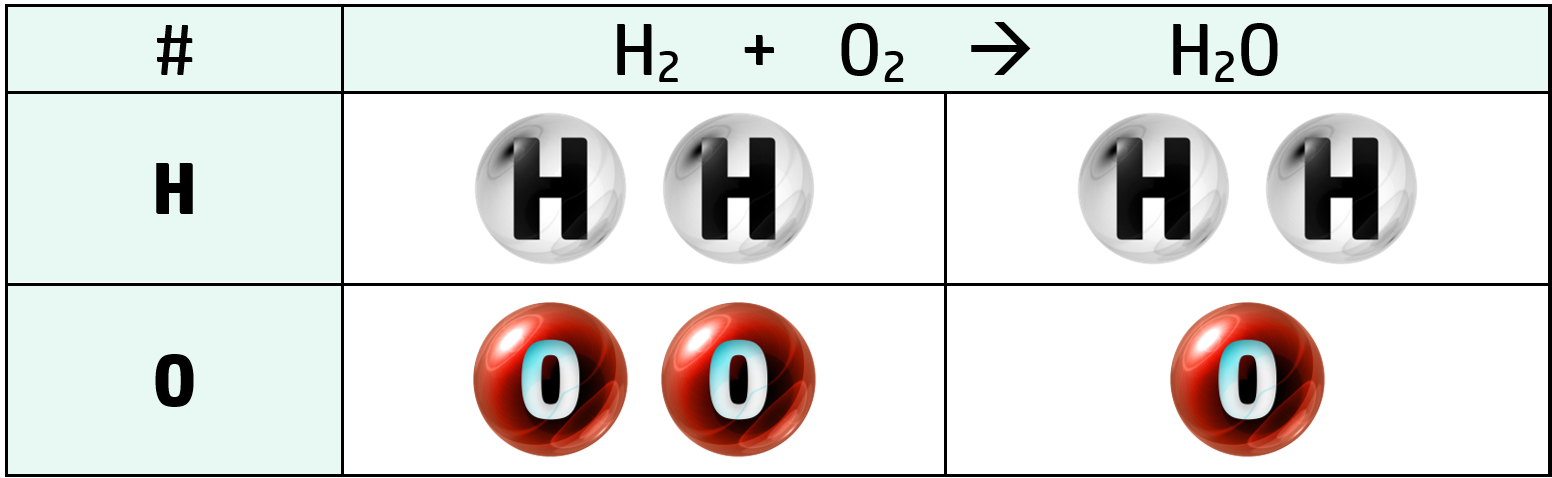
Step 2: The only thing here that is unbalanced is the oxygen in H2O, so put a 2 in front of H2O. This doubles the whole H2O molecule
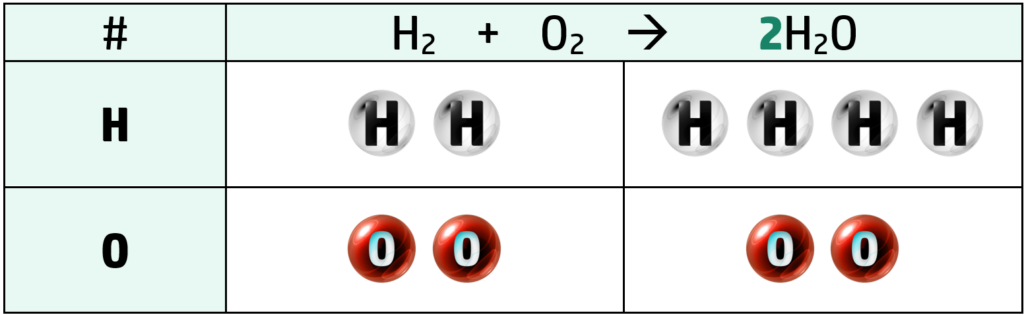
Step 3: Continue this pattern until balanced. Here, we only need to put a 2 before the H2 on the left of the arrow and it is balanced.
Other tips and tricks:
- Always start with the most complicated substance first. Especially if it contains odd / unbalanced numbers. Start by doubling this to make everything even.
- The little number applies to the one element (capital letter) before it. If there is no number, you have one of that element. Example: CO2 contains 1 x carbon and 2 x oxygen.
- If you have a bracket, multiply everything inside the bracket by that number. Example in Mg(NO3)2 – this is telling us we have two lots of NO3 – so we have 2 x Nitrogen and 6 x Oxygen.
- You don’t just have to multiply by 2. If you need to, you can multiply by any number. Example: 2C2H6 + 7O2 → 4CO2 + 6H2O
CH10: Add state symbols to chemical equations
There are 4 state symbols you need to know:




Key things to remember:
- If you have a metal, it will always be a solid, s (except mercury!)
- If you have anything dissolved in water it is aqueous, aq.
- All acids are aqueous, aq.
- Bubbling? You have a gas, g.
Example: A spatula of lithium carbonate powder, Li2CO3, is reacted with nitric acid, HNO3. The products are a solution of lithium nitrate, LiNO3, bubbles of carbon dioxide, CO2, and water, H2O. Complete the state symbols for the balanced equation below:
Li2CO3 ( ) + 2HNO3 ( ) → 2LiNO3 ( ) + CO2 ( ) + H2O ( )
- We are told we are adding a spatula of lithium carbonate powder - powders are solids, so we put (s).
- We have a solution of lithium nitrate, so this is aqueous. All acids are aqueous as well, so we can put (aq) to show that these are dissolved in water .
- We are also told that we have bubbles of carbon dioxide - so this is a gas - so put (g).
- Water is the main example of a liquid that you will see, so put (l).
Therefore, our state symbols should look like this:
Li2CO3 (s) + 2HNO3 (aq) → 2LiNO3 (aq) + CO2 (g) + H2O (l)
CH11: Write balanced equations when given the names of reactants and products (H)
Example: Write the balanced chemical equation for the reaction between calcium carbonate and hydrochloric acid to form calcium chloride, carbon dioxide and water.
Step 1: You need to remember the formula of the basic chemicals – see CH1. From the above reaction, you should already know that hydrochloric acid is HCl, carbon dioxide is CO2 and water is H2O, so we have the following:
Calcium Carbonate + HCl → Calcium chloride + CO2 + H2O
Step 2: You need to work out the formula for calcium carbonate and calcium chloride (CH54):
- Calcium Carbonate: Calcium is in group 2, so loses 2 electrons and has a valency of Ca2. Carbonate ions have a formula of CO32-. The valency is the same, so the formula will be CaCO3.
- Calcium Chloride: Calcium is in group 2, so loses 2 electrons and has a valency of Ca2. Chlorine is in group 7, so gains 1 electron and has a valency of Cl1. To work out the formula, swap the valency and put them subscript: CaCl2.
Step 3: Put this into the equation to have your unbalanced equation:
CaCO3 + HCl → CaCl2 + CO2 + H2O
Step 4: Balance the equation (see CH9 for help):
Here, everything is balanced except for the hydrogen and the chlorine - so we need to put a 2 before the HCl:

CaCO3 + 2HCl → CaCl2 + CO2 + H2O
CH12: Balance equations for reactions between metals and oxygen
Example: Write the balanced equation for the reaction between aluminium and oxygen.
When metals react with oxygen, metal oxides are formed (see CH3), so our word equation looks like this:
Aluminium + Oxygen → Aluminium Oxide
Step 1: Reactants: The metal is just the symbol from the periodic table (Al) and oxygen is O2 (CH1). This can be substituted into the word equation:
Al + O2 → Aluminium Oxide
Step 2: Products: To do this, work out the valency (CH54) for aluminium and oxygen. Aluminium wants to lose three electrons so has a valency of Al3. Oxygen wants to gain two electrons, so has a valency of O2. Swap the valency and we have a formula of Al2O3, which can be added to the equation:
Al + O2 → Al2O3
Step 3: Balance the equation (CH9): Start by doubling the aluminium oxide as this is the most complex substance and has an odd number in it. This gives us 4 aluminium's and 6 x oxygen’s, so 4Al and 3O2 balances the equation:

4Al + 3O2 → 2Al2O3
CH13: Balance equations for reactions between metals and water
Example: When lithium reacts with water, lithium hydroxide and hydrogen are formed. Write the balanced equation for the reaction.
Lithium + Water → Lithium Hydroxide + Hydrogen
Step 1: Put in the things you know: The metal is just the symbol from the periodic table (Li), water is H2O and hydrogen is diatomic, so has the formula H2 (see CH1):
Li + H2O → Lithium Hydroxide + H2
Step 2: Formula for lithium hydroxide. Lithium has a valency (CH54) of Li1 as hydrogen wants to lose one electron. A hydroxide ion has the charge of OH-, so also has a valency of 1. Therefore, as both have the same valency, we have one of each, and a formula of LiOH:
Li + H2O → LiOH + H2
Step 3: Balance the equation (CH9). Start with the most complicated molecule and double it - in this case, we double the LiOH as it has odd, unbalanced numbers in it. This gives us 2 x lithium, 4 hydrogen’s and 2 oxygen’s. So doubling everything on the left of the arrow will balance our equation:

2Li + 2H2O → 2LiOH + H2
CH14: Balance equations for displacement reactions
Example: When calcium chloride reacts with sodium hydroxide, a displacement reaction occurs and calcium hydroxide and sodium chloride form. Write the balanced equation for the reaction.
calcium chloride + sodium hydroxide → calcium hydroxide + sodium chloride
Step 1: Work out the valency (CH54): Sodium has a valency of Na1 (it loses one electron); calcium has a valency of Ca2 (it loses two electrons); chlorine has a valency of Cl1 (gains one electron); the hydroxide has a valency of OH1 (charge is OH-). Therefore, the formulae are:
- Calcium chloride: Ca2 + Cl1 → CaCl2
- Sodium hydroxide: Na1 + OH1 → NaOH
- Calcium hydroxide: Ca2 + OH1 → Ca(OH)2
- Sodium chloride: Na1 + Cl1 → NaCl
CaCl2 + NaOH → Ca(OH)2 + NaCl
Step 2: Balance the equation (CH9): Calcium hydroxide has two OH groups, so start by doubling the NaOH on the left. Then we have two sodium's on the left, so double the NaCl on the right and then we are balanced:

CaCl2 + 2NaOH → Ca(OH)2 + 2NaCl
CH15: Balance equations for complete combustion
Example: Write the balanced equation for the complete combustion of methane, CH4.
Complete combustion always involves a fuel reacting with oxygen and always produces carbon dioxide and water:
methane + oxygen → carbon dioxide + water
Step 1: Write the unbalanced equation: Methane has been given in the question and oxygen; carbon dioxide and water are all common covalent substances that you need to learn (CH1)
CH4 + O2 → CO2 + H2O
Step 2: Balance the equation (CH9): Always leave oxygen to the end when balancing combustion equations. So, here, we have 4 hydrogens on the left and only 2 on the right of the arrow – double the H2O. Now all that is left is the oxygen – we have 4 on the right, so a 2 before the O2 completely balances the equation:

CH4 + 2O2 → CO2 + 2H2O
CH16: Balance equations for neutralisation reactions
Example: When nitric acid and magnesium hydroxide react together, magnesium nitrate and water are formed. Write the balanced equation for the reaction.
magnesium hydroxide + nitric acid → magnesium nitrate + water
Step 1: Substitute in the ones you know (CH1): You should know nitric acid water, so add these into the equation:
magnesium hydroxide + HNO3 → magnesium nitrate + H2O
Step 1: Formula for magnesium hydroxide and magnesium nitrate: Magnesium has a valency (CH54) of Mg2 as it wants to lose 2 electrons. Hydroxide ions have the formula OH- and nitrate ions have the formula NO3-. With this, you can work out the formula (not forgetting brackets for polyatomic ions!):
- Magnesium hydroxide: Mg2 + OH1 → Mg(OH)2
- Magnesium nitrate: Mg2+ NO31→ Mg(NO3)2
Mg(OH)2 + HNO3 → Mg(NO3)2 + H2O
Step 3: Balance the equation (CH9): You have two nitrate ions on the right, so put a 2 before the nitric acid. Then all that is left is to put a 2 before the water on the right and it is balanced:
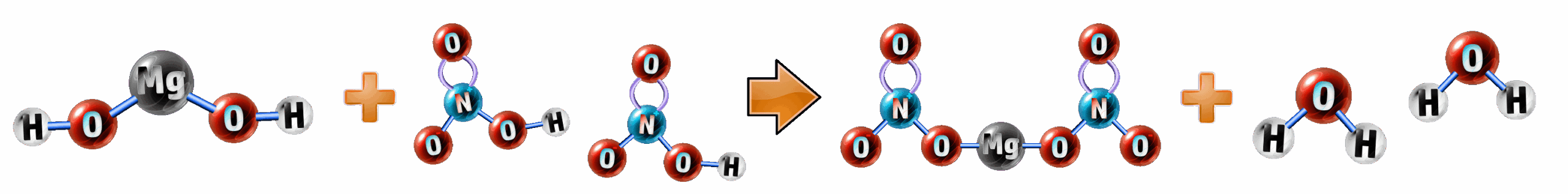
Mg(OH)2 + 2HNO3 → Mg(NO3)2 + 2H2O
CH17: Balance equations for reduction reactions
Example: Iron oxide, FeO, can be reduced by heating with carbon. The products are iron and carbon dioxide. Write the balanced equation for the reaction.
Iron oxide + carbon → Iron + carbon dioxide
Step 1: The unbalanced equation: You have been given the formula of iron oxide: FeO. Iron and carbon are both elements, so will have the formulae ‘Fe’ and ‘C’ respectively. Carbon dioxide is CO2, which you are expected to know (CH1).
FeO + C → Fe + CO2
Step 2: Balance the equation (CH9): We have two oxygens on the right of the arrow, so need to double the FeO – put a 2 before it. All that is left is to double the Fe on the right and it is balanced:

2FeO + C → 2Fe + CO2
