SCIENTIFIC SKILLS: ANALYSIS AND SAFETY


A GCSE chemistry revision page on analysis techniques including tests for gases (oxygen, hydrogen, carbon dioxide), tests for ions (chloride, sulfate, carbonate), flame tests, practical steps, observations and explanations to help with common exam questions.
CH22: Describe the difference between a risk and hazard
CORROSIVE

ENVIRONMENTAL HAZARD

FLAMMABLE

HARMFUL

OXIDISING

TOXIC
In any chemical reaction, there will be hazards (things that can cause harm). Those hazards will usually have a hazard symbol, like the ones above. You will be asked how to lower the risk (lower the chance of harm happening).
Normally, the best thing to put is to wear goggles so that you don’t get any chemicals in your eye or to wear gloves so that you don’t get any chemicals on your hands.
There are some other common ways to reduce risk which can be seen in the table below:
CH23: Drawing and interpreting graphs
If you are given some data and asked to plot it into a graph, make sure that the independent variable (left column) is on the x-axis, and the dependent variable (the result) is on the y-axis.
Make sure you space the points out evenly (going in 5’s, 10’s, etc…) and make sure you put axis on your graph, as well as a line of best fit.
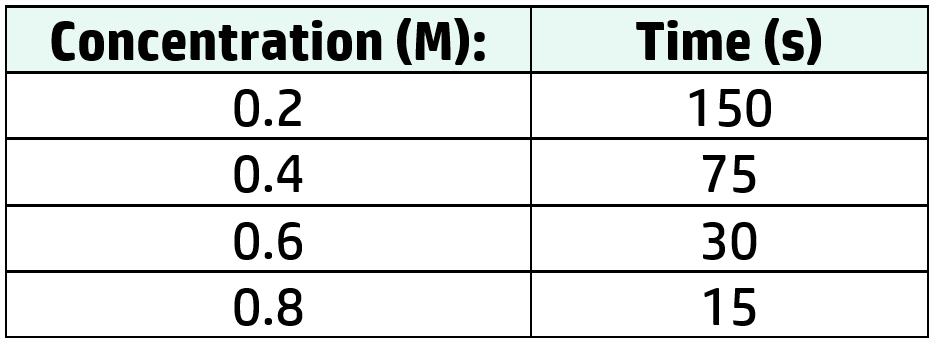
Example: Draw a graph for the following data:
Always make sure your axis cover at least 50% of the graph paper!
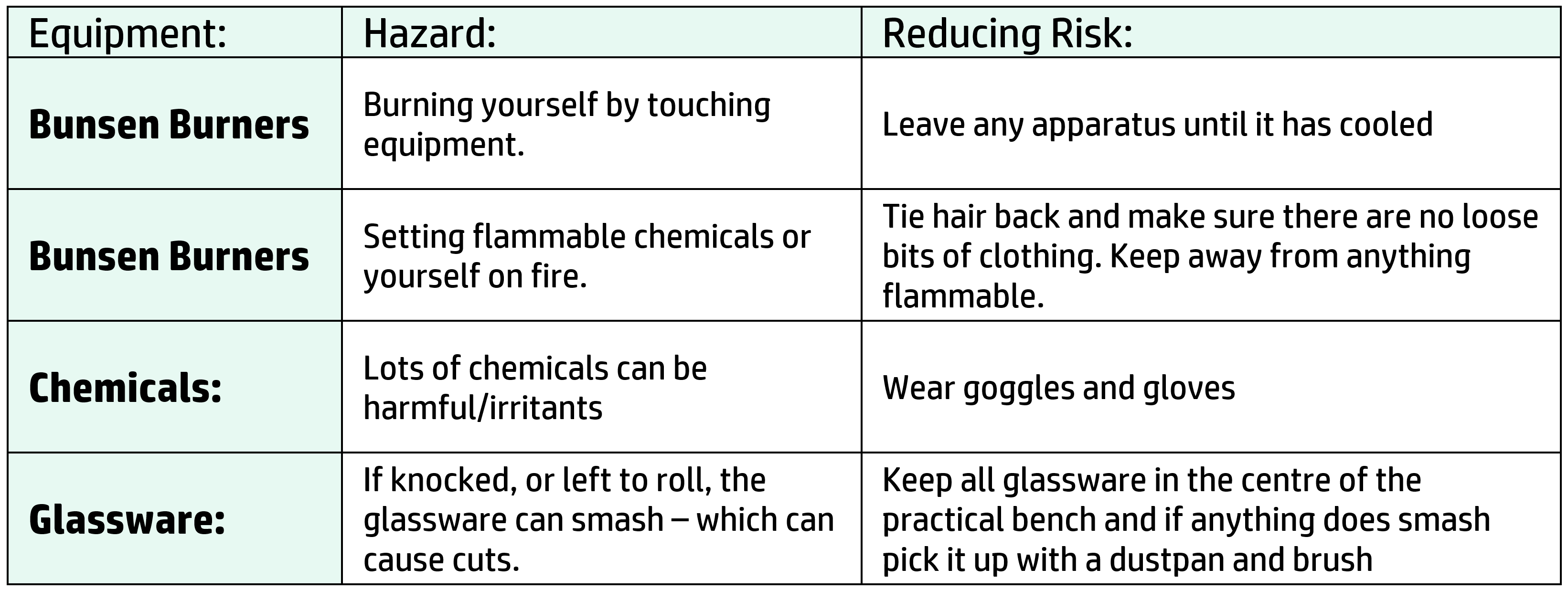
CH24: Analysing data from tables
If you are given a table of data (or a graph) look at the first column – this is your independent variable. Look at what happens to the dependent variable as the first column changes and state this. Use values to back up your conclusion.
Example: Describe the effect of increasing the concentration on the time taken for the reaction to occur.

OK Answer: As concentration increases, time decreases.
Better Answer: As the concentration increases from 0.2M to 0.8M, the time decreases from 150s to 15s.
CH25: Calculate percentages
Example 1: Calculate the percentage of lithium in lithium chloride, LiCl. (Ar: Li = 7, Cl = 35.5)
- Step 1: Work out the Relative Formula Mass of lithium chloride (CH78):
- Li = 7 x 1, Cl = 35.5 x 1; 7 + 35.5 = 42.5
- Step 2: Divide the mass of lithium by the mass of lithium chloride and multiply by 100:
- (7 ÷ 42.5) x 100 = 16.5%
Example 2: In a chemical reaction, phosphorous reacted with oxygen in a gas syringe. The gas syringe was full of 50cm3 of air. At the end of the reaction, the volume of gas had decreased by 21%. Calculate the final volume of air in the syringe.
- Step 1: Work out 21% of 50cm3:
- 50cm3 ÷ 100 = 0.5; 0.5 x 21 = 10.5cm3 - this is the volume of air ‘lost’.
- Step 2: Subtract this from the initial volume to get your final volume:
- 50 – 10.5 = 39.5cm3
CH26: Calculate Means
To calculate the mean, just add up all of the values and divide by the number of values:
Example: Calculate the mean time for the chemical reaction below:

Here we have 4 times and no anomalies, so we add all four values up and divide by 4 giving an answer of 14.5s
If we did have an anomaly (a value that doesn’t fit the pattern), we should remove it before working out the mean:

Here, 19.6 is quite far out from the other three values, so don't include it. Add the three concordant values together (14.2, 13.8 and 14.5) and divide by three because we now are only using three values. This gives an answer of 14.2s.
CH271: Identify variables in an investigation

Independent Variable: The thing you change in a reaction
Dependent Variable: The thing you measure/record in an investigation
Control Variable: The things you keep the same in an investigation
Example: Investigating the effect of mass on the volume of gas produced in a reaction.
Measure 30cm3 of 1 moldm-3ethanoic acid and add it to a conical flask. Measure 0.05g of calcium carbonate and add it to the conical flask. Place the bung into the conical flask and measure the final volume of gas produced on the syringe. Repeat the experiment with 0.10g, 0.15g and 0.20g of calcium carbonate
In the above reaction:
- We have changed the mass of calcium carbonate in each experiment – this is the independent variable.
- We have measured / recorded the volume of gas produced – this is the dependent variable.
- Control variables that should be kept the same are:
- The volume of ethanoic acid
- The concentration of ethanoic acid
- The temperature of ethanoic acid
- The surface area of calcium carbonate
