BONDING AND THE PROPERTIES OF MATTER

An AQA GCSE chemistry revision page showing INSERT CONTENT HERE.
CH48: Describe what an ionic bond is
All elements want a full outer shell. Metals do this by losing electrons and non-metals do this by gaining electrons.
An ionic bond is the transfer of electrons from a metal to a non-metal. An ionic bond is the strong electrostatic attraction between the cation and anion

CH49: Describe what an ion is
An ion is any atom that has lost or gained electrons to become charged.
A metal will always lose electrons to become a positively charged cation (cations are paw-sitive!). A non-metal will always gain electrons to become an anion (ANegative ION)

CH50: Calculate the protons, neutrons and electrons for an ION
Calculating the number of protons and neutrons for an ion is exactly the same as for a normal atom.
For an ion, you must work out how many electrons an atom needs to gain/lose to get a full outer shell and take that away/add that to the number of electrons the atom would have.

Clue: It will always be the same as the closest noble gas.
CH51: Describe the formation of ions in groups 1, 2, 6 and 7
Metals want to lose electrons to get a full outer shell. They always form positive cations.
Non-metals want to gain electrons to get a full outer shell. They always form negative anions.
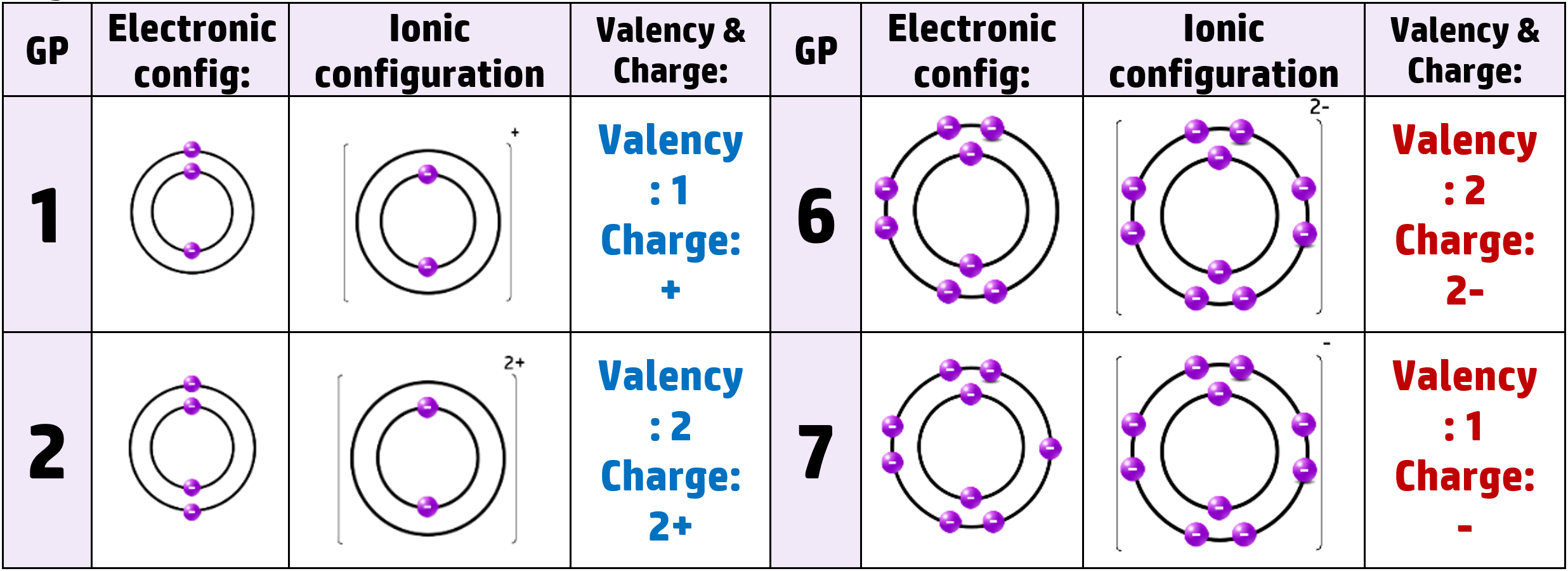
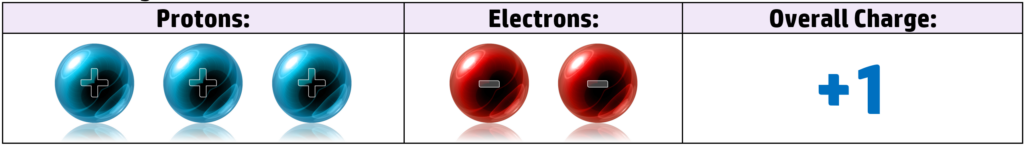
Lithium, for example, has 3 protons and 3 electrons. It has the electronic configuration of 2.1 – meaning it has 1 electron in the outer shell. When it loses that electron to get a full outer shell, it now has 3 positive protons and 2 negative electrons, giving it an overall charge of +1
Similarly, oxygen (in group 6) gains 2 negative electrons and becomes an O2- ion.
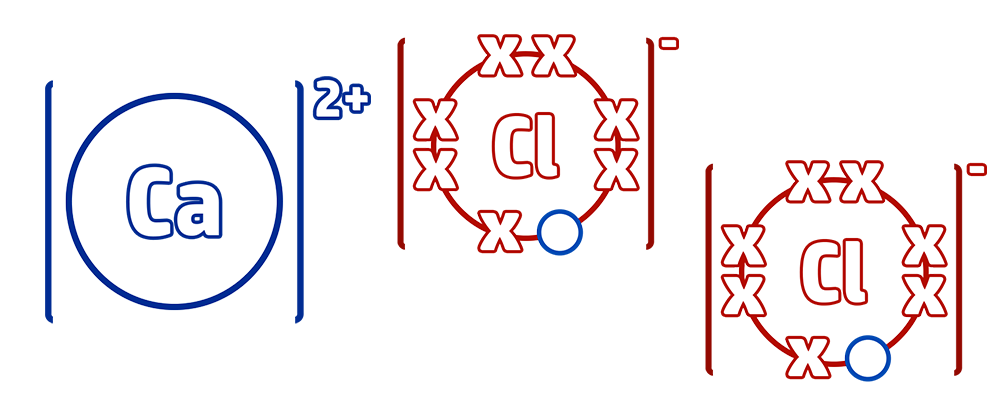
CH52: Use dot and cross diagrams to draw ionic bonding
Step 1: When drawing ionic bonding start by drawing out the outer shell – which is the group the element is in. Calcium is in group 2 and chlorine is in group 7.

Step 2: Move as many electrons as possible from the metal to the non-metal by drawing arrows. Make sure that you don’t go below 0 for your metal, or above 8 for your non-metal.

Step 3: Continue this until both ions have a full outer shell. In this case, calcium still had 1 electron that it needed to lose, but calcium had a full outer shell – therefore add another calcium and continue to transfer.

Step 4: Redraw your new ions with their full outer shells. Add a bracket around both and add the charge (CH51) Calcium chloride has the formula CaCl2
CH53: Describe the difference between ‘-ide’ and ‘-ate’
If you have a compound that ends in ‘-ide’, it will be made of a metal and a non-metal ONLY.
If you have a compound that ends in ‘-ate’, it will be made up of a metal, a non-metal and OXYGEN.
For example:
- Sodium sulphide is only made up of sodium and sulfur and has the formula Na2S.
- Sodium sulfate is made up of sodium, sulfur and oxygen and has the formula Na2SO4.
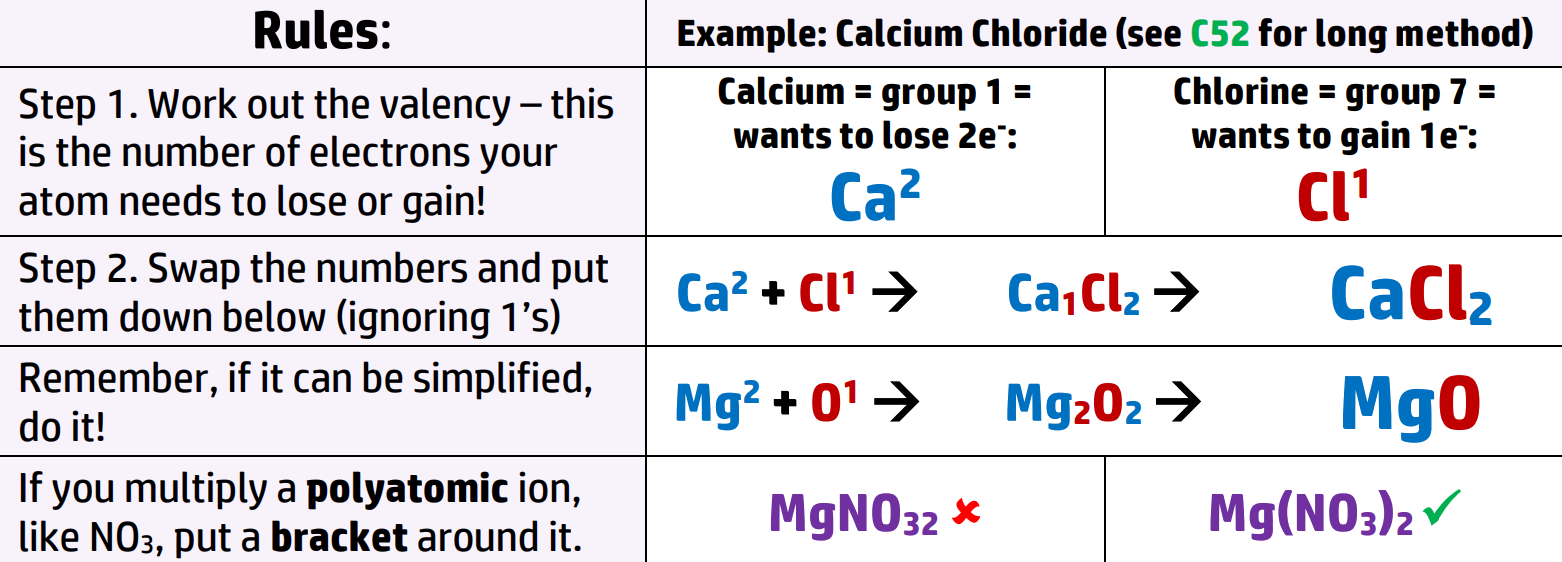
CH54: Work out the formula for ionic compounds
CH55: Describe what an ionic lattice is
A lattice is a regular arrangement of cations and anions held together by a strong electrostatic force of attraction.

CH57: Explain why ionic substances can only conduct when molten
Ionic Compounds cannot conduct when SOLID:
- When solid, there is a strong force of attraction between the ions.
- The ions are not free to move and cannot carry a charge.

Ionic Compounds can conduct when LIQUID/MOLTEN/AQUEOUS:
- When molten (melted) or dissolved, the ions are free to move.
- The ions can now move and conduct electricity.
CH58: Describe what a covalent bond is
A covalent bond is a shared pair of electrons between non-metals.
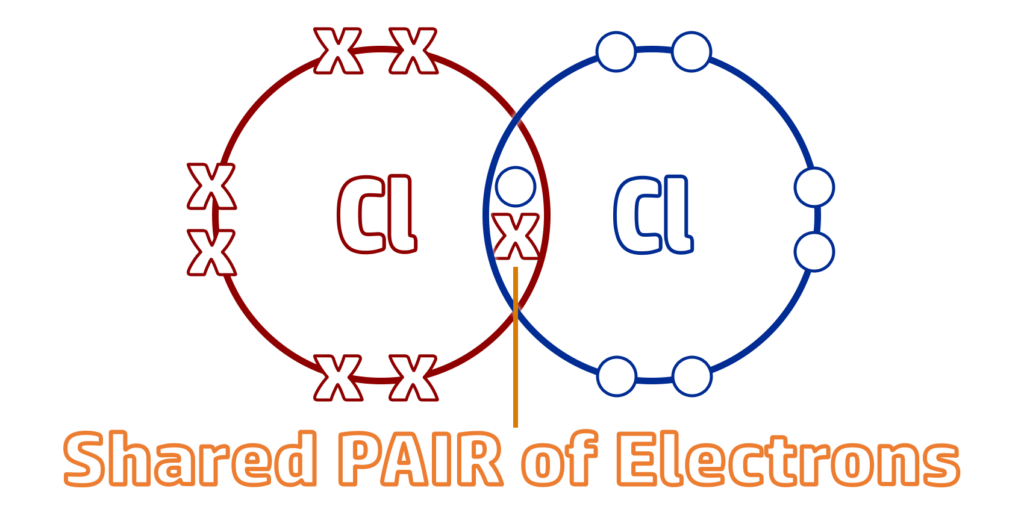
CH59: Describe what a molecule is
A molecule is a substance that involves covalent bonding and only contains a few atoms. Carbon dioxide, CO2, is an example of a molecule.

CH60: Identify the size of a molecule
Molecules are tiny, usually around 1-100nm small.
To convert nm into metres, multiply the size in nm by 1×10-9. To convert metres into nm, multiply the size in m by 1×109
CH61: Draw dot and cross diagrams for covalent substances
Step 1: Work out the valency – this is the number of electrons an atom needs to lose or gain to get a full outer shell.
On the right, chlorine (in group 7) has 7 electrons in its outer shell. It wants to gain one electron, so has a valency of 1: Cl1
Oxygen (in group 6) has 6 electrons in its outer shell. It wants to gain two electrons, so has a valency of 2: O2


Step 2: The valency is the number of bonds an atom can make. So oxygen can make two bonds and chlorine can make 1.
This means that we will need two chlorines to react with one oxygen, giving us the formula OCl2

Step 3: Redraw the stick diagram, but with a circle overlap for every ‘stick’:
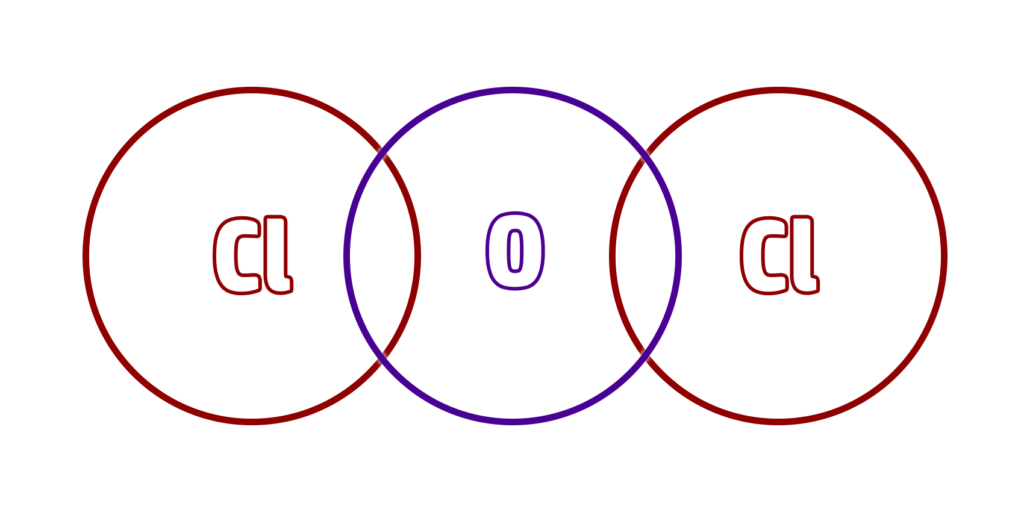
Step 4: Put a shared pair of electrons in the overlaps (a dot and cross).
This shared pair of electrons is your covalent bond.
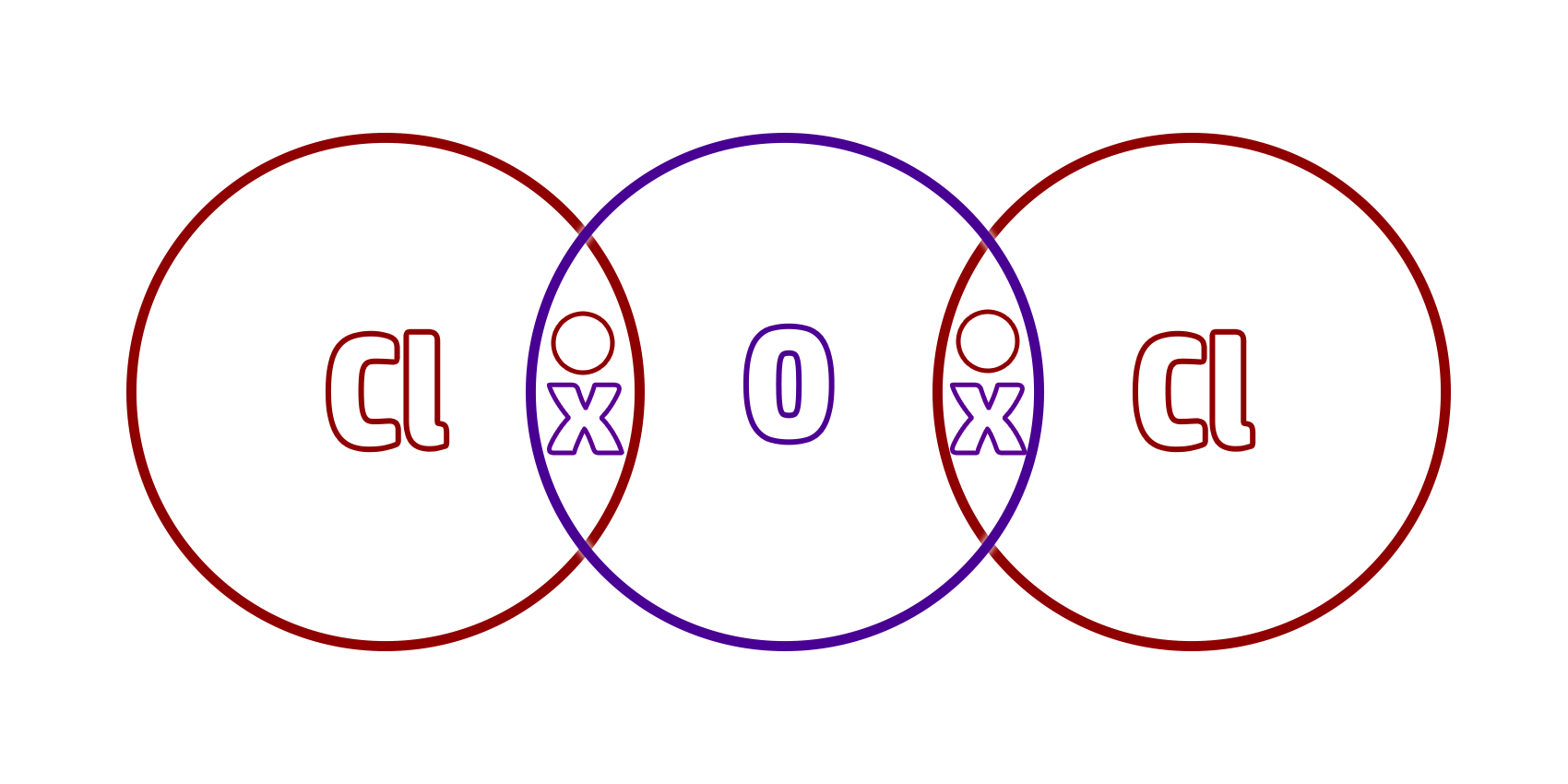
Step 5: Make sure that each atom has a full outer shell (everything should have 8 electrons except for hydrogen which should only have 2)
Watch the video on the right for more examples.
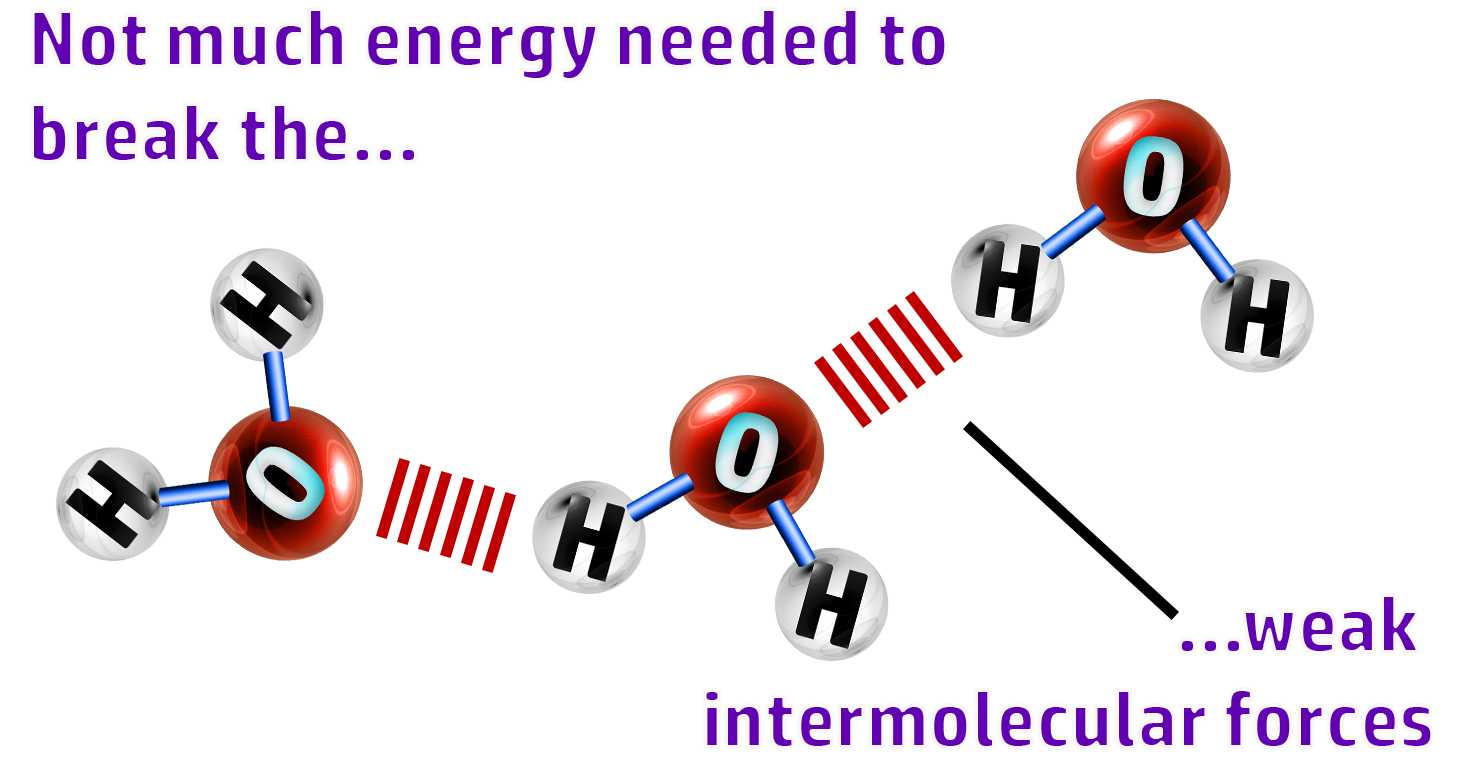
CH62: Explain why simple molecular substances have low melting points
They have low melting point and boiling points
Not much energy is needed to break the weak intermolecular forces (forces between the molecules)
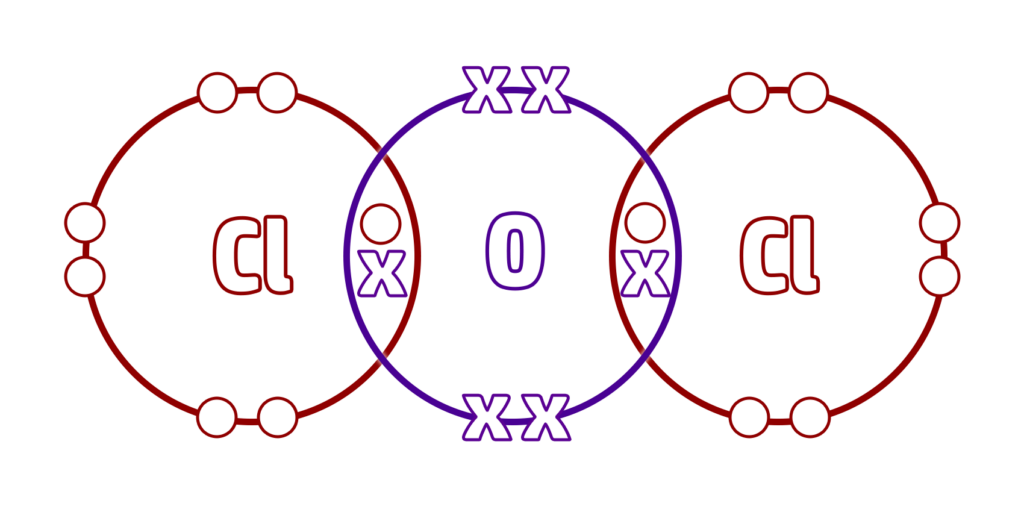
CH63: Explain why simple covalent substances do not conduct electricity
They cannot conduct electricity.
The electrons are not delocalised (free), so cannot move/flow.
This means they cannot carry a charge.

CH64: Describe what a giant covalent substance is
Giant covalent substances are bonded the same way as seen in simple covalent molecules. They still have shared pairs of electrons and involve only non-metals.
The only difference is the number of atoms and bonds involved – there are a lot more.
This gives giant covalent substances a high melting point because lots of energy is needed to break the strong covalent bonds.
They also normally don’t conduct electricity (there are some exceptions, such as graphite, graphene and C60) because the electrons are not delocalised (free), so cannot move/flow.
Diamond and Graphite are both examples of allotropes of carbon – a substance made up of the same element but with a different chemical structure
CH65: Describe the structure of diamond and graphite
Diamond
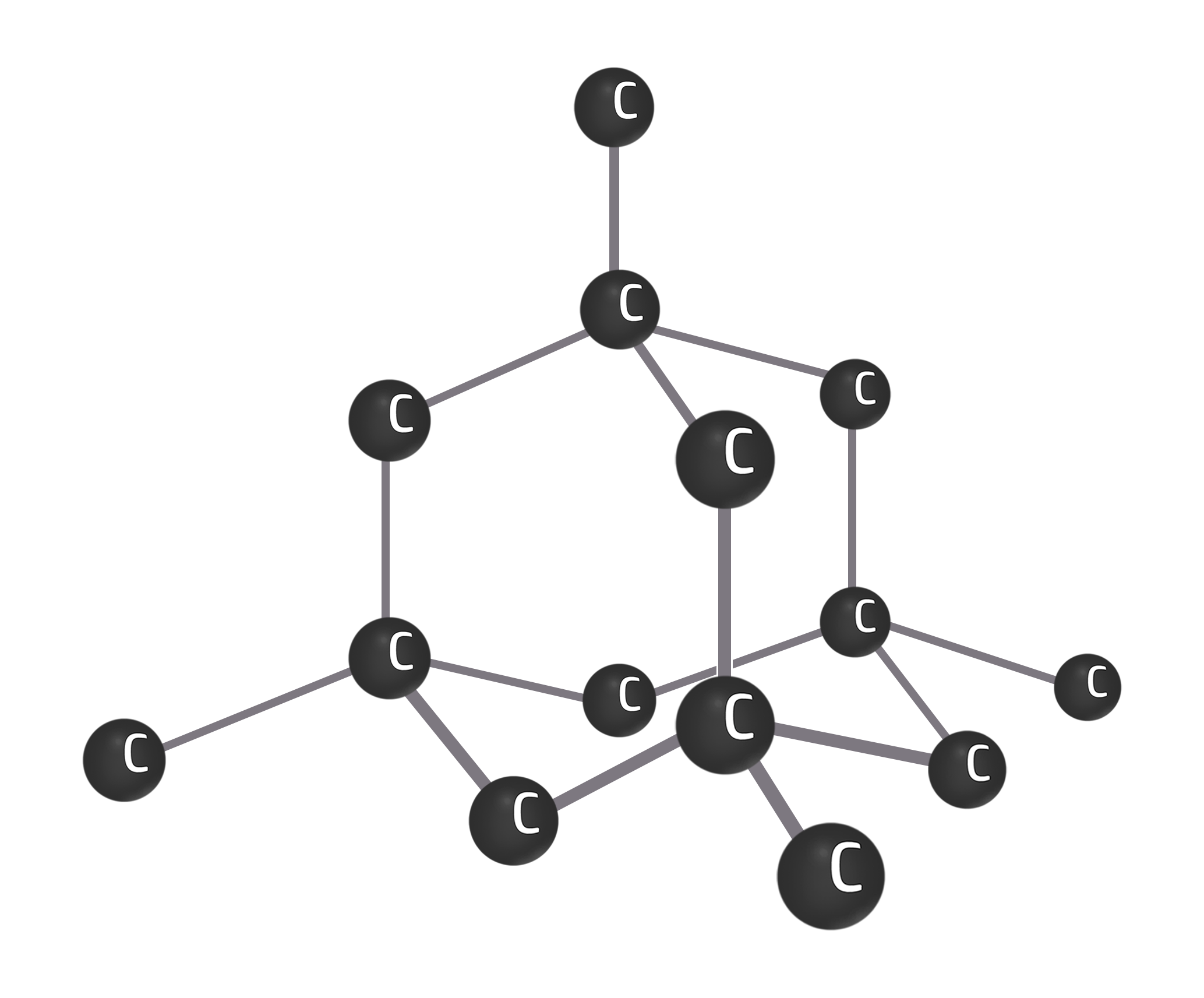
Diamond is an allotrope of carbon (same element, different structure) where every carbon has 4 strong covalent bonds.
Graphite
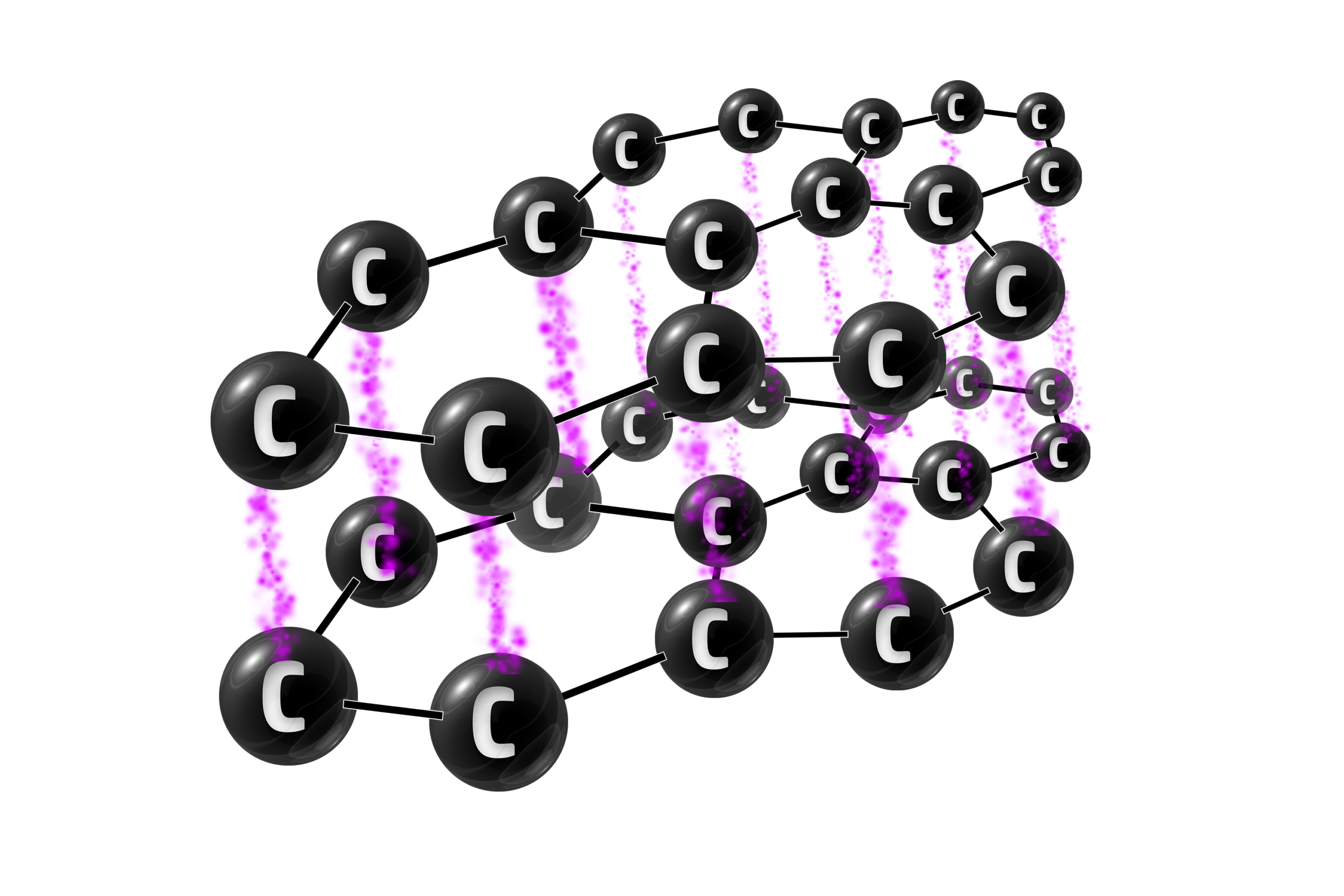
Graphite is an allotrope of carbon (same element, different structure) where every carbon has 3 strong covalent bonds. This gives it thin layers of carbon atoms.
CH66: Explain the uses of diamond and graphite
Diamond
Diamond has 4 strong covalent bonds between each carbon atom. This makes diamond strong.
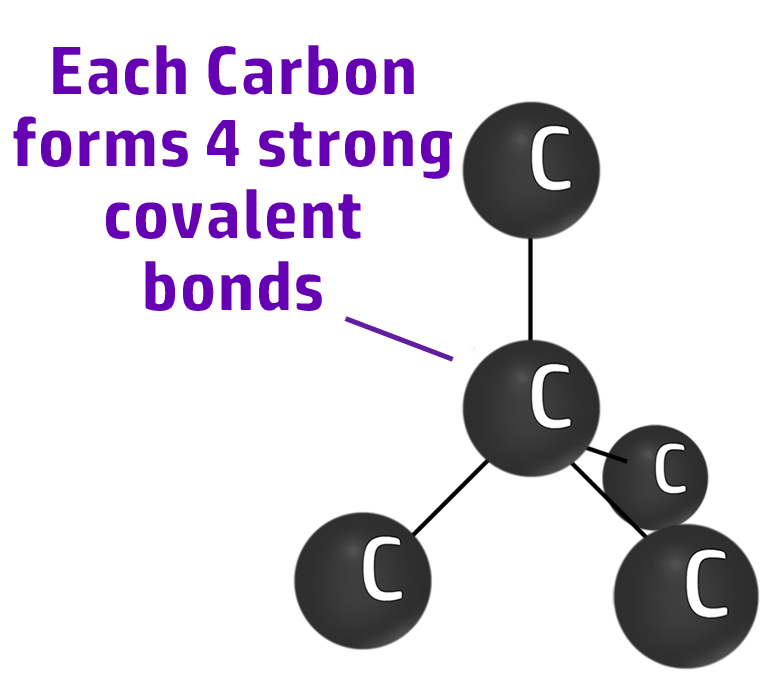
Diamond can be used in cutting tools because it takes a lot of energy to break the strong covalent bonds.
Graphite
Graphite has 3 strong covalent bonds between each carbon atom.
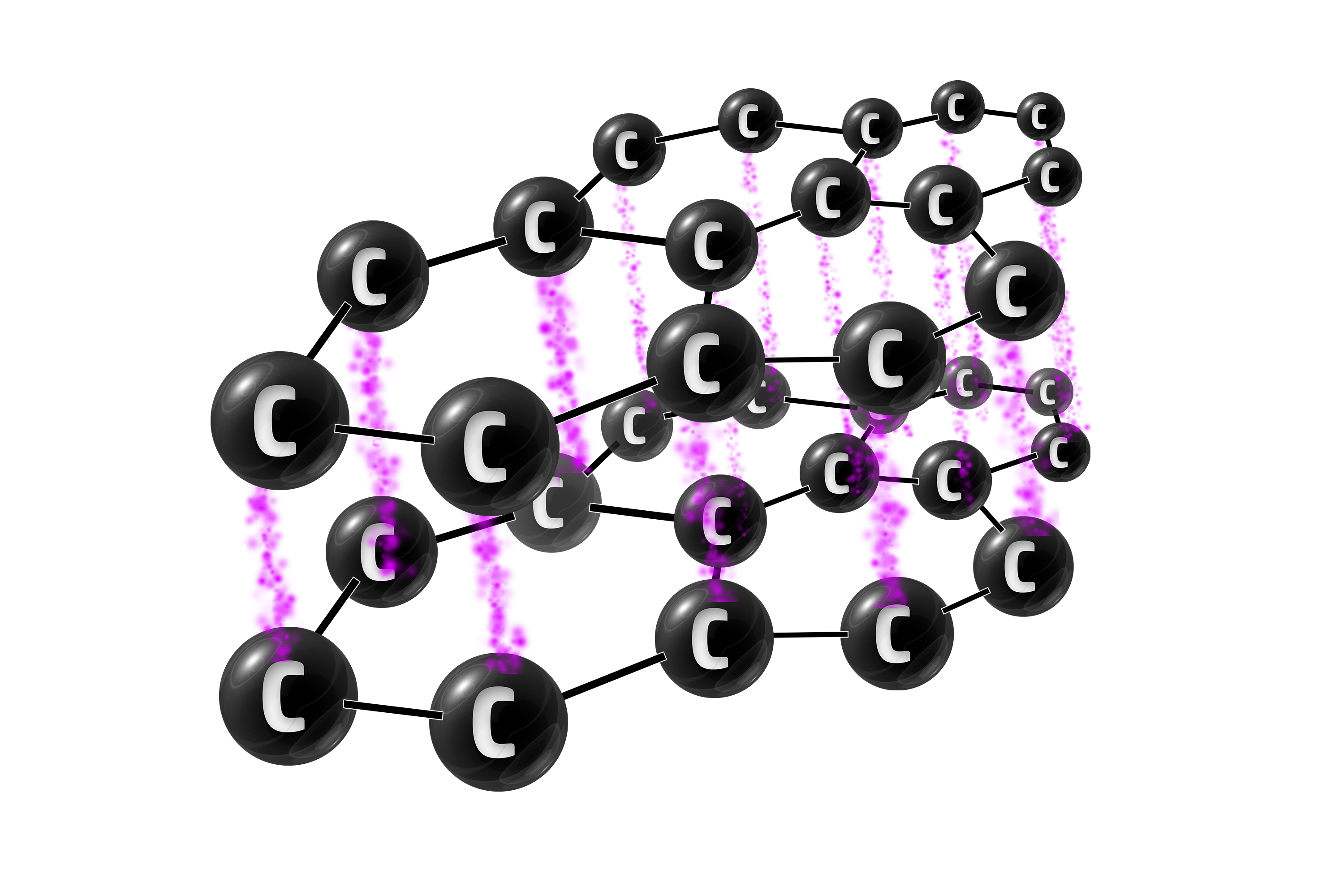
Graphite can be used in electrodes because the delocalised electrons are free to move – allowing it to conduct electricity.
Graphite is also used as a lubricant in cars because it has layers which slide past each other – reducing friction.
CH67: Describe the properties of graphene, fullerenes and nanotubes
Graphene

Graphene is one layer of graphite.
It is light (1 atom thick) and strong (lots of strong covalent bonds). It also conducts electricity because the delocalised electrons are free to move.
Buckyballs
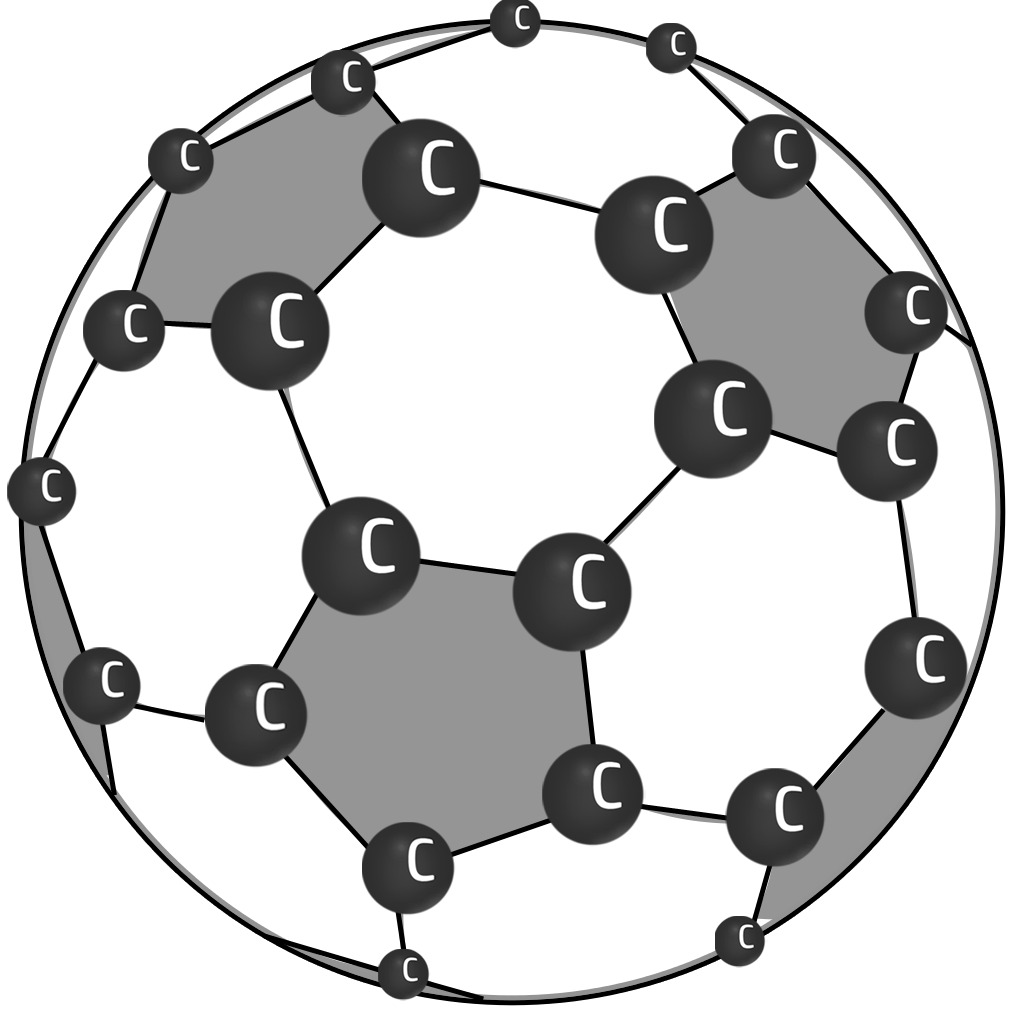
Simple covalent spheres of carbon atoms.
They conduct electricity because the delocalised electrons are free to move. They have low melting points because not much energy is needed to break the weak intermolecular forces.
Nanotubes
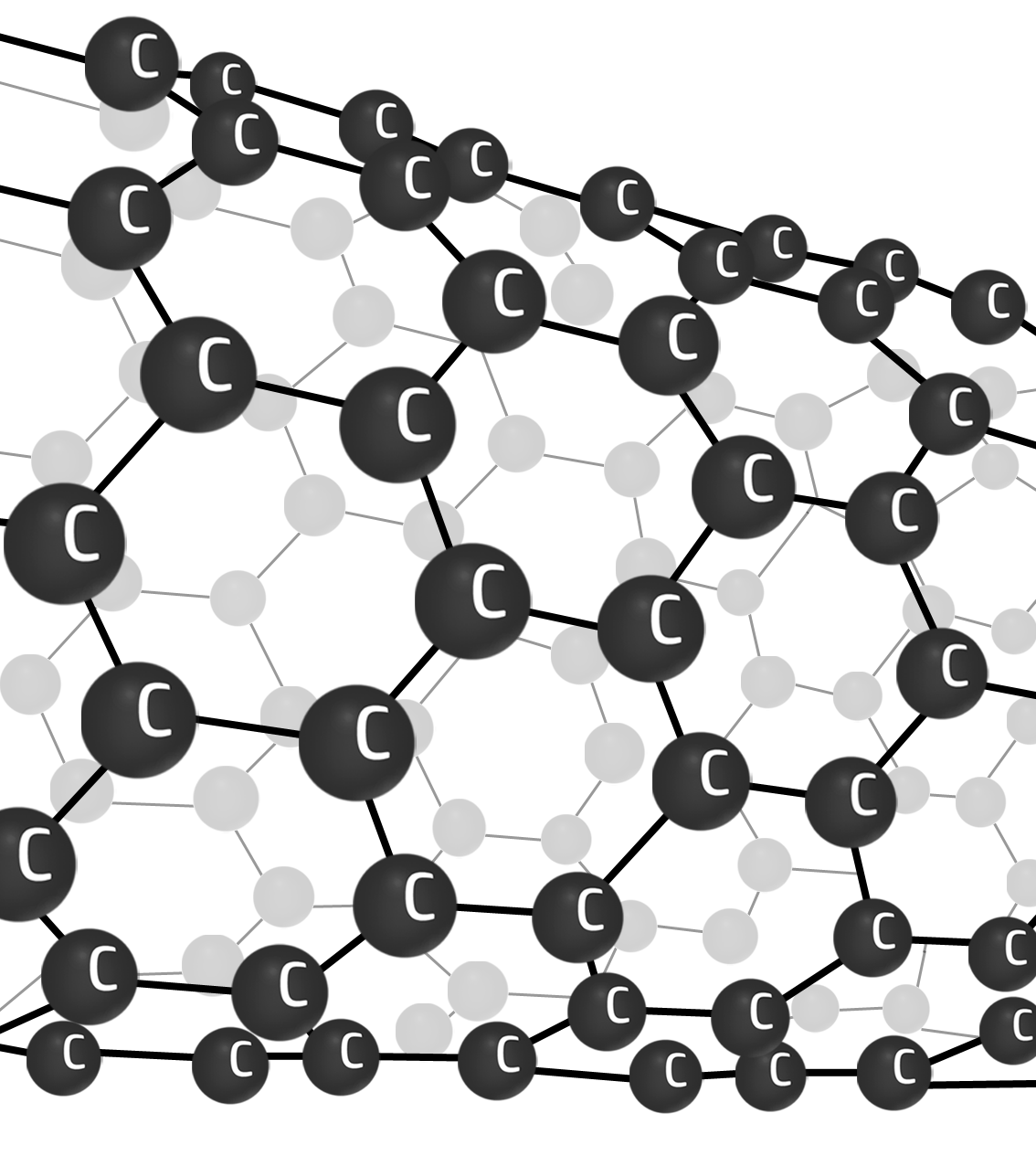
A ‘rolled up’ sheet of graphene.
Nanotubes have high tensile strength (can’t be stretched). They are strong (lots of strong covalent bonds) and can conduct electricity (delocalised electrons flow).
CH68: Explain why metals have high melting points
Lots of energy needed to break the strong electrostatic attraction between cation and delocalised electrons.
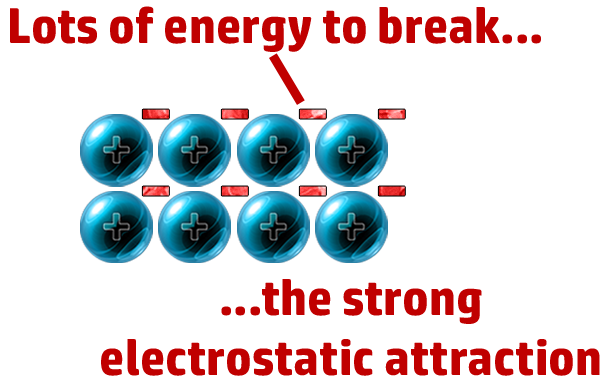
CH69: Explain why metals conduct electricity
Delocalised electrons can flow/move and carry a charge

CH70: Explain why metals are malleable
The layers can slide past each other, but it doesn’t break the strong electrostatic attraction between cation and delocalised electron.
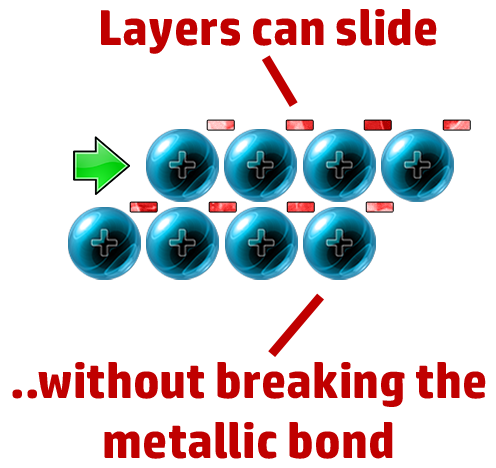
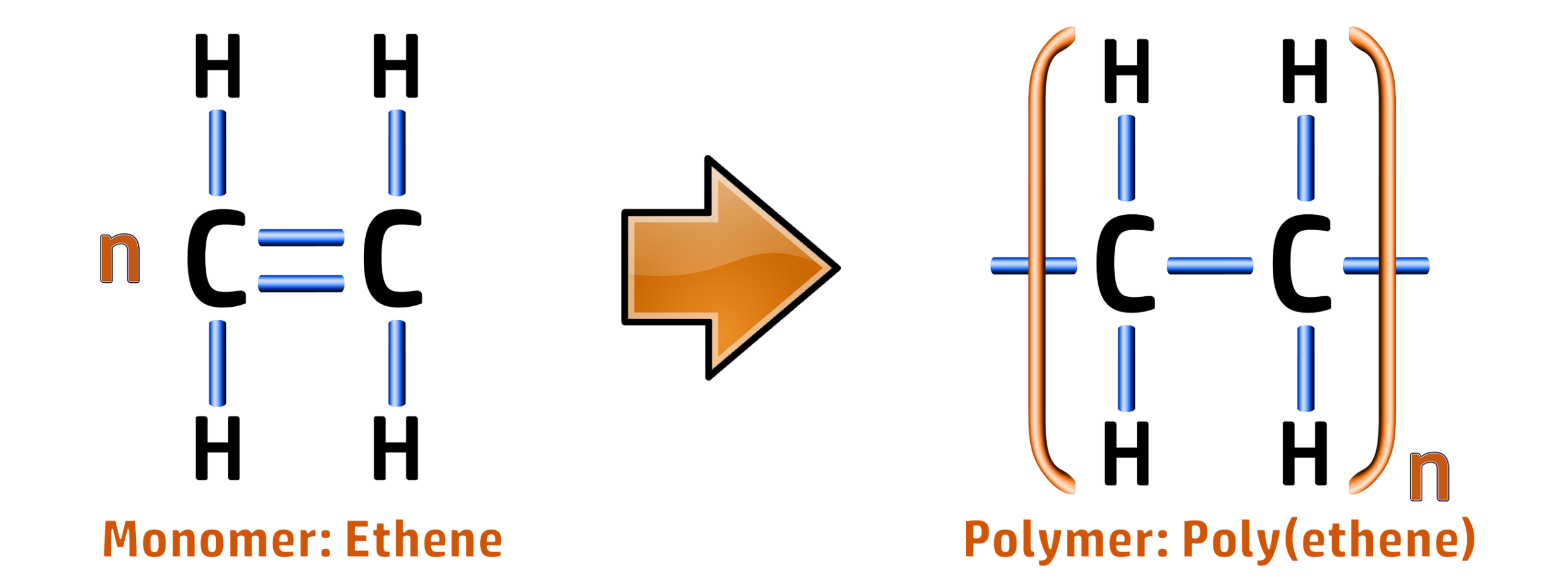
CH72: Explain what a polymer is
A polymer is a long chain of smaller molecules called monomers.
These are examples of simple covalent compounds and have low melting points and cannot conduct electricity.
An example of this is ethene – which has the formula C2H4:
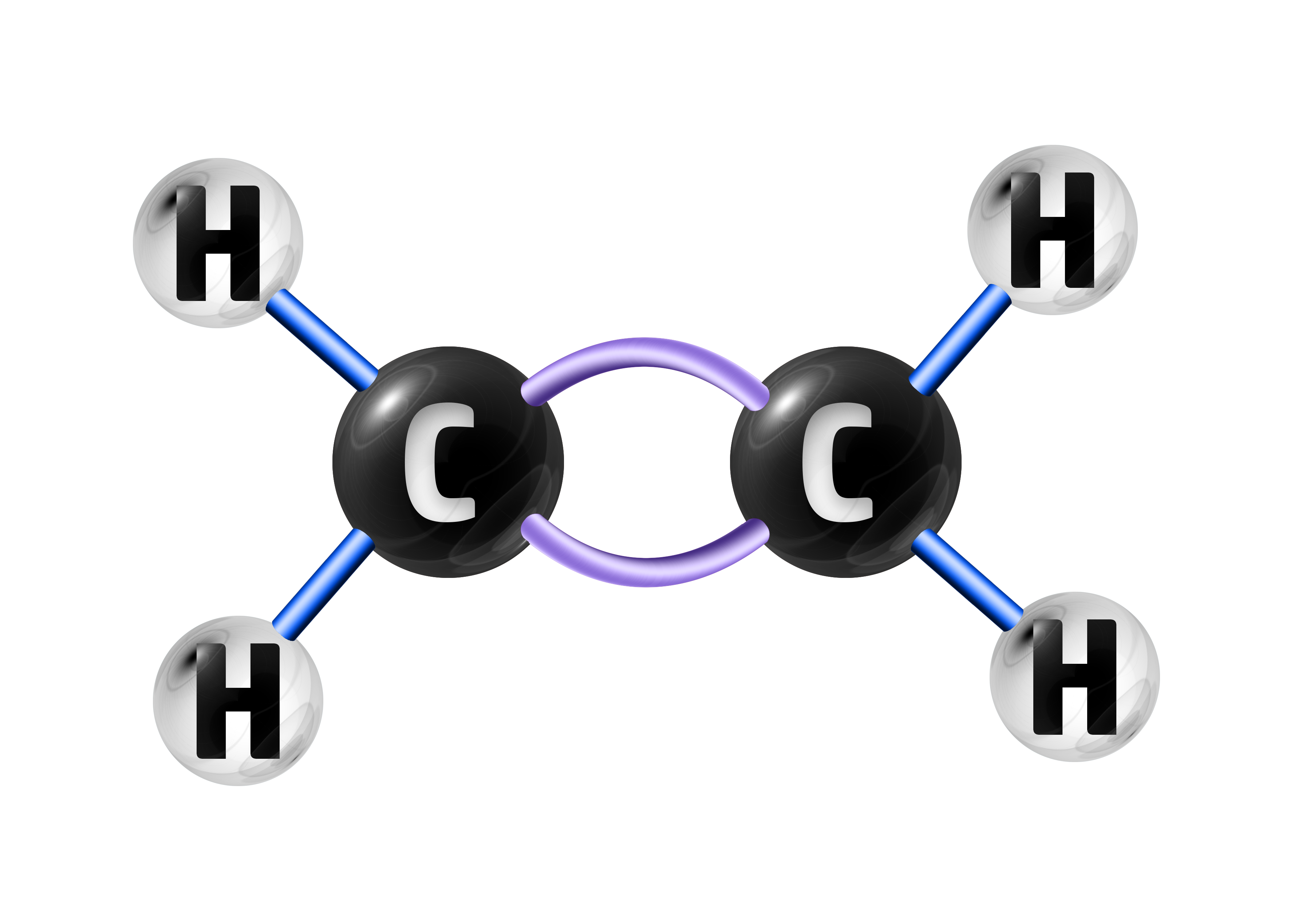
When the double bond is broken, many ethene molecules will join together to make poly(ethene):
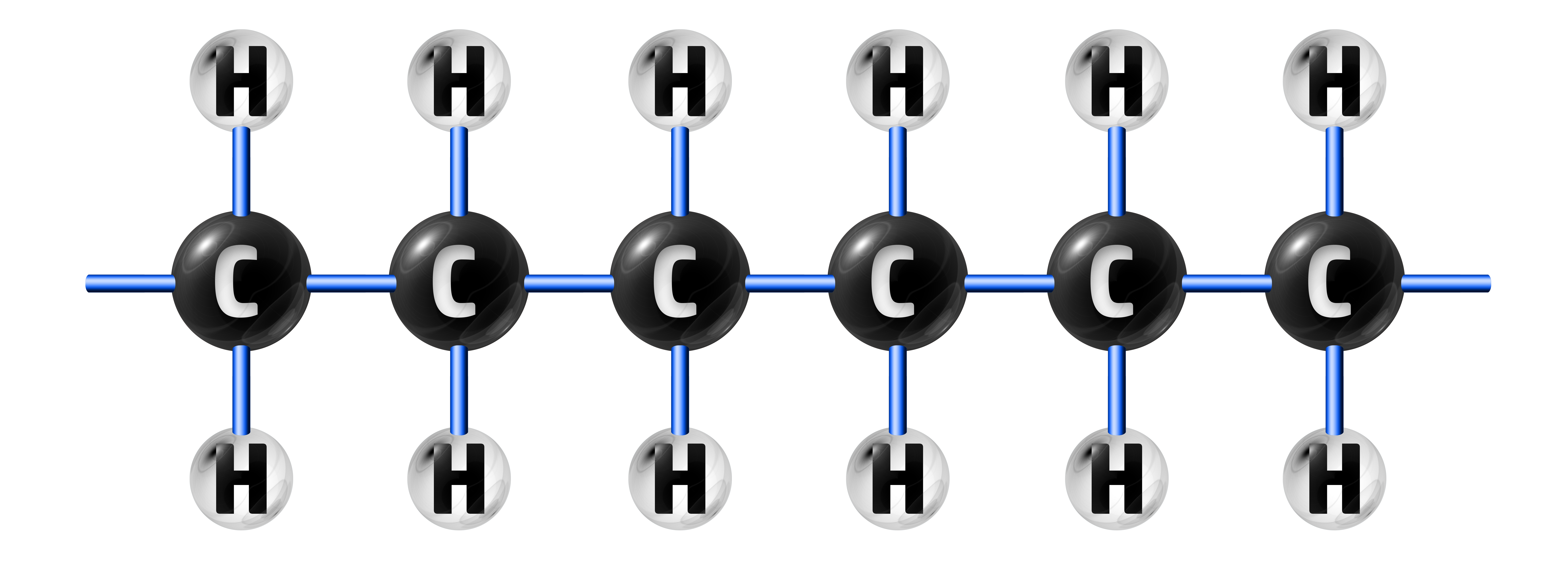
CH163: Explain how alloying makes metals stronger
An alloy is a mixture of a metal and another element – usually another metal.

Pure Metals:
In a pure metal, all the particles are the same size, forming a regular pattern / lattice.
This means that the layers can slide past each other – making them malleable, ductile, and SOFT.
Alloys:
In an alloy, the particles are different sizes.
This means that the layers cannot slide past each other.
This makes alloys much stronger than pure metals.
CH164: Explain the uses and properties of metals and their alloys
| Name: | Metal or Alloy? | Use: | Explanation |
|---|---|---|---|
| Gold | Pure metal | Memory Chips | Excellent conductor of electricity.Soft and expensive. |
| Jewellery Gold | Alloy | Jewellery | Alloy of gold and copper.Stronger than gold but still attractive. |
| Copper | Pure metal | Wires and Pipes | Good conductor of electricity and ductile. Cheaper than gold. |
| Brass | Alloy | Plug Pins | Alloy of copper and zinc.Worse conductor, but stronger than copper. |
| Aluminium | Pure metal | Overhead Cables | Good conductor of electricity.Less dense and cheaper than copper. |
| Magnalium | Alloy | Aircraft Parts | Alloy of magnesium (5%) and aluminium (95%).Stronger than aluminium but still low density. |
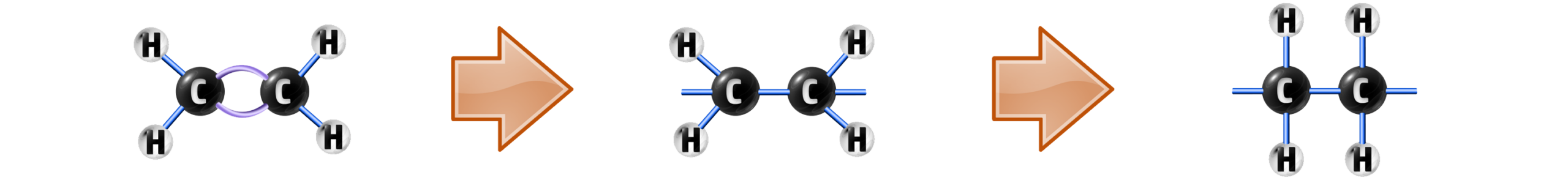
CH254: Explain how polymers form during addition polymerisation
Polymers (See CH72) can be formed in two main ways. Addition Polymerisation and Condensation polymerisation (see CH263).
The addition polymerisation of ethene occurs when a monomer (small molecule) has its double bond broken…
…and then many monomers are added together forming polyethene:
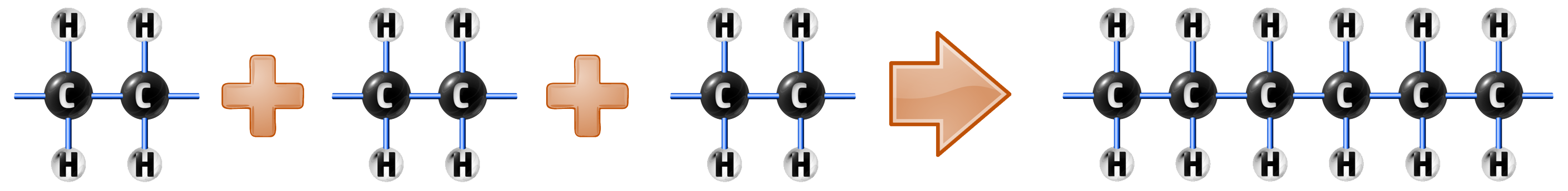
When writing out polymers, you can write them like this:
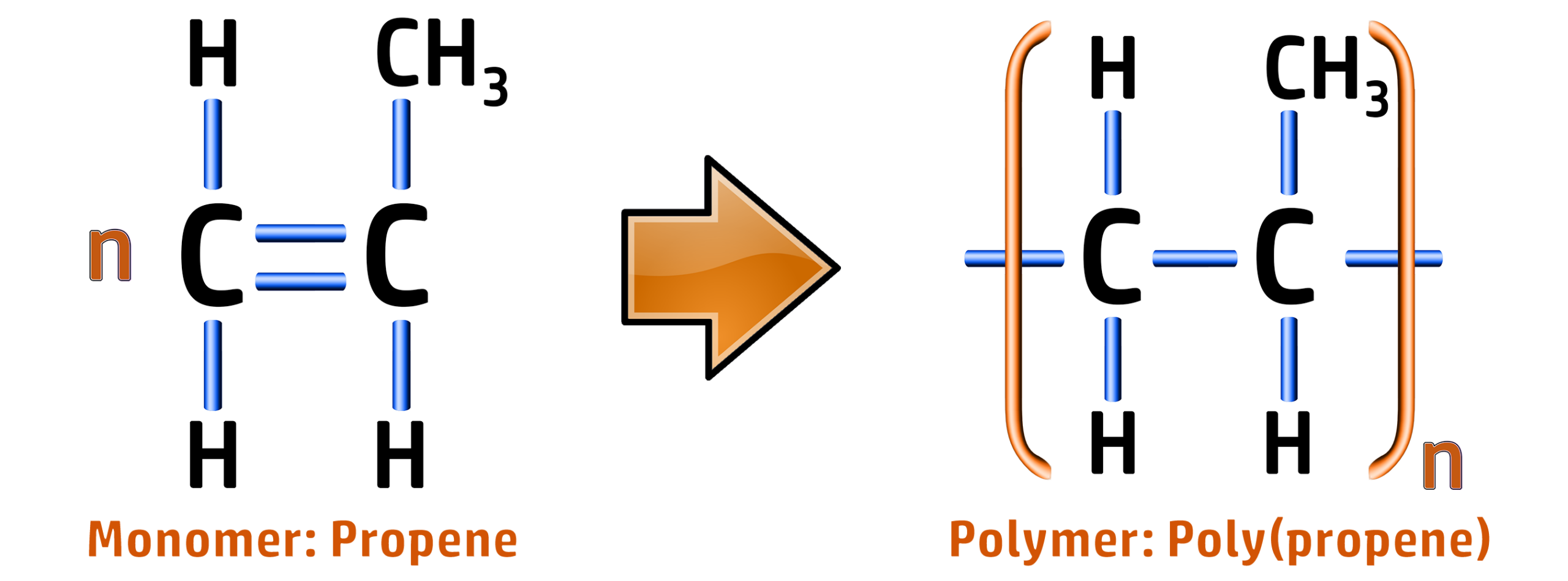
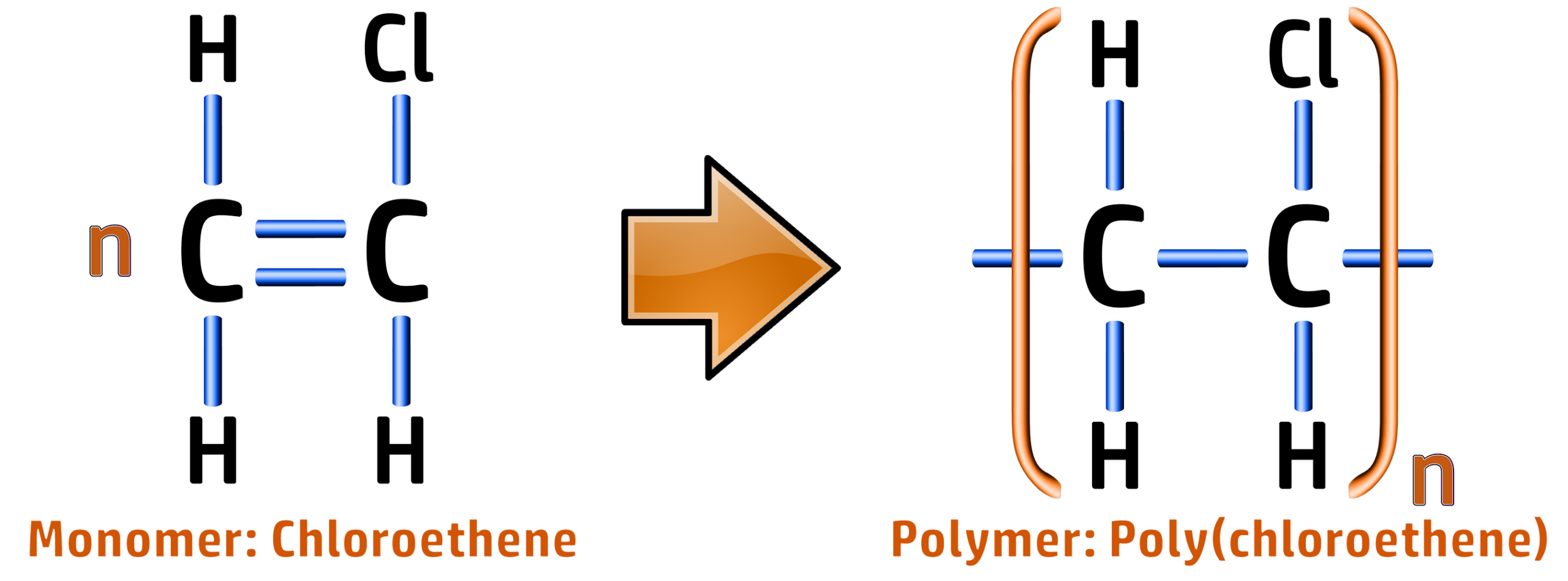
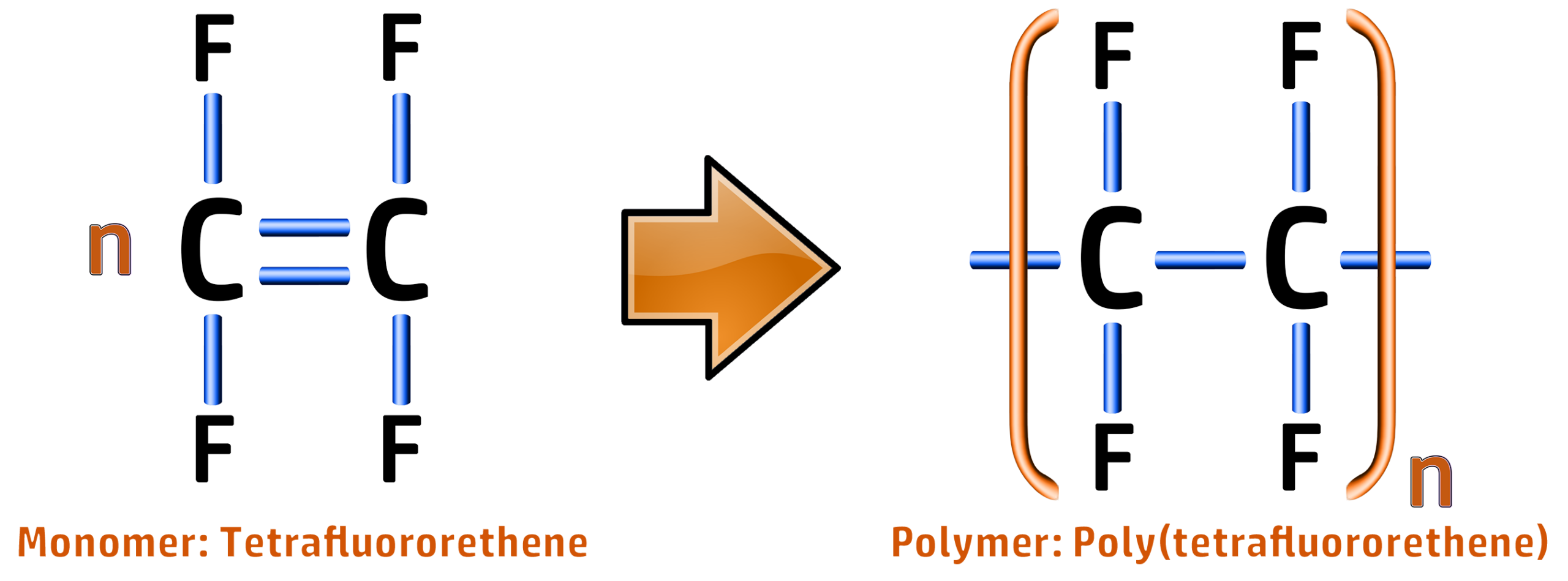
CH255: Draw repeating units for polypropene, PVC and Teflon
Poly(propene):
Poly(chloroethene) / PVC:
Poly(tetrafluoroethene) / Teflon::
CH256: Draw monomers from polymer chains
When given a polymer and asked to draw the monomer for it, all you need to do is look at the first two carbons.
Example: Draw the monomer for the following polymer of poly(bromoethene) below:
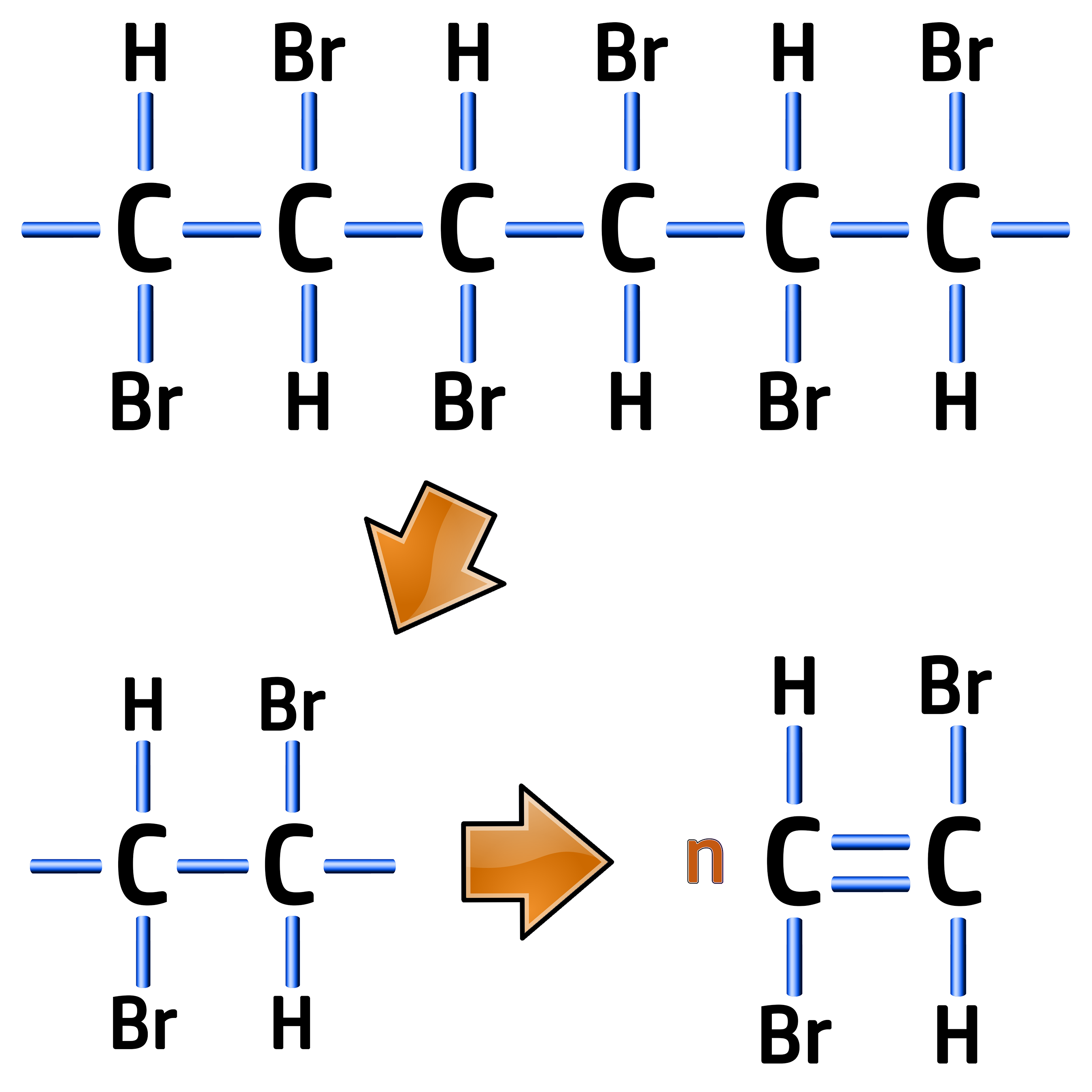
The repeating unit for an addition polymer is always two carbons wide, so you need to replicate the first two carbons making sure the elements above and below them are kept in the same order.
All you need to do is redraw the first two carbons with a double bond between them, like on the left.
CH257: Describe the uses of different polymers and link to their uses

Polymer: Poly(ethene) / Polythene
Formula: [C2H4]n
Type: Addition Polymer
Use: Plastic bags, bottles and clingfilm.
Reason: Flexible, cheap, good insulator.

Polymer: Poly(propene) / Polypropylene
Formula: [C3H6]n
Type: Addition Polymer
Use: Buckets, Crates, Bowls, Ropes and Carpets.
Reason: Flexible, does not shatter.

Polymer: Poly(chloroethene) / PVC
Formula: [C2H3Cl]n
Type: Addition Polymer
Use: Window frames, gutters, pipes, Wire insulation.
Reason: Tough, good insulator for wires.

Polymer: Poly(tetrafluoroethene) / Teflon
Formula: [C2F4]n
Type: Addition Polymer
Use: Non-stick coating for frying pans.
Reason: Tough, slippery.

Polymer: Poly(ethylene terephthalate) / PET
Formula: [C10H8O4]n
Type: Condensation Polymer
Use: Plastic furniture, carpets, fleece, bottles.
Reason: Strong, lightweight, shatter resistant.
CH264: Describe what a nanoparticle is
Nanoparticles are chemicals that range from 1 to 100 nanometres (nm) in size.1nm is the same as 0.000,000,001m (1×10-9m)
You may be asked to compare the size of a nanoparticle to the size of another particle.
Example: A nanoparticle of titanium dioxide has a diameter of 240nm. A titanium atom has a diameter of 0.15nm. Calculate how many times bigger the nanoparticle is compared to a titanium atom.
To answer this question, you need to divide the diameter of the nanoparticle by the diameter of the titanium atom: 240nm / 0.15nm = 1600 times bigger.
They may also ask you to convert between nanometres and metres.
Example: A nanoparticle of titanium dioxide has a size of 240nm. Calculate the size of the particle in nanometres.
To answer this question, you need to remember that 1nm is 1×10-9m.
So 240nm is 240×10-9m. This can be simplified → 24×10-8m → 2.4×10-7m.
CH265: Calculate the surface area-to-volume ratio of a nanoparticle
Example: A cube shaped nanoparticle has sides of 20nm. Calculate its surface area-to-volume ratio.
Step 1: Work out the area of one side of the cube.
- Area = Height x Width = 20nm x 20nm = 400nm2
Step 2: Work out the total surface area of the cube.
- Surface Area (of a cube) = Area x 6 = 400nm2 x 6 = 2400nm2
Step 3: Work out the total volume of the cube.
- Volume = Height x Width x Depth = 20nm x 20nm x 20nm = 8000nm3
Step 3: Work out the surface area-to-volume ratio of the cube.
- Surface area-to-volume ratio = surface area ÷ volume = 2400nm2 ÷ 8000nm3 = 0.3 : 1
CH266: Explain why nanoparticles can be used in sunscreens and as catalysts

Sunscreen
When used as a sunscreen, the particles are extremely small, making the titanium dioxide transparent. This means that it doesn’t leave white marks.
The titanium dioxide in sunscreen absorbs ultraviolet rays. This helps to prevent skin cancer (which ultraviolet rays can cause).
It is also a nanoparticulate, giving it different properties to when it is used in bulk.
Catalysts
Nanoparticles have a large surface area-to-volume ratio, which makes them useful as catalysts.
They can be used as catalysts in detergent and self-cleaning windows because they help to catalyse the breakdown of dirt, keeping the clothes / windows clean.

CH267: Explain the concerns over nanoparticles
There are a few concerns about using nanoparticles:
- The small size allows them to be breathed in / absorbed into body.
- Once in the body, they may catalyse harmful reactions, harming our health.
- Nanoparticles are a new technology, so we don’t know the long term impact of using them.

