USING RESOURCES

An AQA GCSE chemistry revision page on INSERT CONTENT HERE
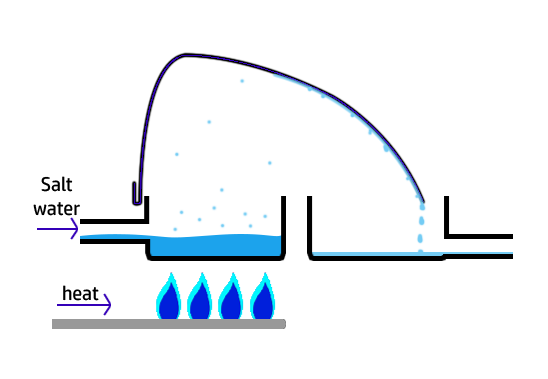
CH106: Describe to make salt water potable (safe to drink)
The word potable means safe to drink. This is how to make saltwater potable:
- Salt water is added to a large container and heated using crude oil.
- The water will evaporate and then hit the lid and cool back down – condensing it back down into water.
This use a lot of energy, so is not used to purify water on a large scale.
CH107: Describe how to make lake water potable (safe to drink)
To make water from lakes, reservoirs, or aquifers (underground rocks that contain water) potable, which means safe to drink, there are a series of steps the water must go through:
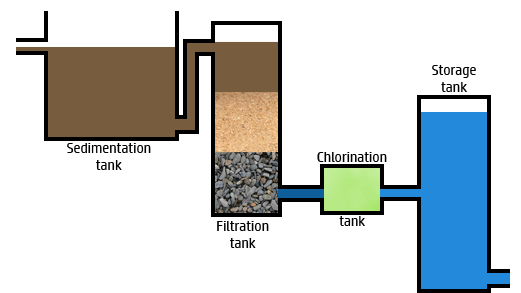
- Step 1: Screening. A sieve is used to remove large impurities, such as twigs and leaves.
- Step 2: Sedimentation. The water is left so that the small particles settle out at the bottom.
- Step 3: Filtration. Beds of sand and gravel are used to remove smaller insoluble particles that remain.
- Step 4: Chlorination: Chlorine is added to sterilise the water – killing any microorganisms present.
CH150: Evaluate Bioleaching and Phytoextraction (H)
There are two other methods of extracting metals which are used to extract rocks with small amounts of metals in – called low grade ores: Bioleaching and Phytoextraction.
Bioleaching:
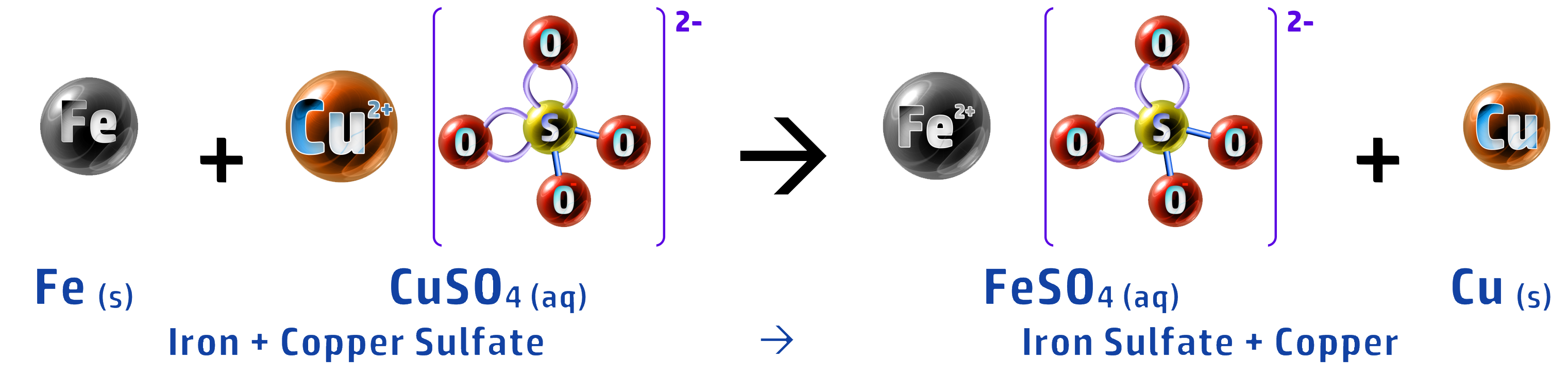
- Bioleaching uses bacteria to break down low-grade ores into a solution called a leachate.
- This solution is acidic and contains the metal ions you want.
- An example of this is collecting copper, from a copper sulfate leachate, using scrap iron.
- Iron is more reactive than copper so displaces it:
| Advantages: | Disadvantages: |
|---|---|
| Does not require higher temperatures. No harmful gases Less damage to landscape than mining Conserves supplies of higher-grade ores | Very slow. Toxic substances and sulphuric acid can be produced – damaging the environment |
Phytoextraction
- Plants absorb mineral ions from the soil.
- The metal ions become concentrated in the plant cells.
- The plants are burnt, leaving behind the metal in the ash.
| Advantages: | Disadvantages: |
|---|---|
| Can extract metals from contaminated soil. No harmful gases Less damage to landscape than mining Conserves supplies of higher-grade ores | Very slow. Can only grow in certain climates |
CH151: Describe the advantages and disadvantages of Recycling
Advantages of recycling:
- It conserves Earth’s natural resources – meaning we won’t run out as fast!
- Less mining of ores is needed. This is good because:
- It doesn’t damage the landscape.
- It doesn’t create noise/dust pollution.
- It can take less energy to recycle than to extract from the ore.
- Metals don’t end up wasted in landfill sites.
Disadvantages of recycling:
- It takes time for people to sort through all of the different plastics / metals.
- It costs money to employ people to sort the plastics / metals.
- Some materials cannot be recycled.

CH152: Life Cycle Assessments
A lifestyle assessment looks at the total environmental cost of a product by looking at each stage of the life of a product:
| Stage: | Description: |
|---|---|
| Choice of material | When metals are extracted from their ores, it needs a lot of energy and can produce a lot of pollution. Raw materials can come from crude oil, which is non-renewable. Crude oil also gives out greenhouse gases when combustion occurs. |
| Manufacture of product | Manufacturing needs a lot of energy. It can cause a lot of pollution. Waste products need disposing of safely – some can be recycled. The water used in lots of manufacturing needs to be safe/unpolluted when put back into the environment |
| Use of product | The products can themselves be harmful – such as: Toxic fumes from paint, Toxic gases from combustion and Fertilisers draining into lakes/rivers causing eutrophication |
| Disposal of product | Lots of products are disposed of in landfills, which takes up space and can pollute the land/water. Products can also be burnt – which can give off toxic gases or greenhouse gases |
Each of these factors need to be considered when manufacturing a product. If there are multiple ways of producing a product, the one that has the least environmental cost will be chosen.
CH153: Evaluating Life Cycle Assessments
Example: A decision needs to be made about whether to produce a cabinet from two different sources:
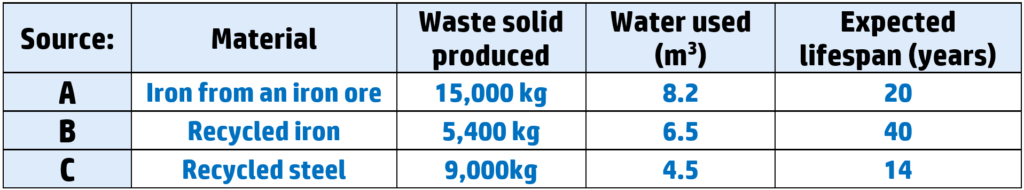
You would not choose A because the iron comes from an ore – which takes more energy to extract than recycling. It also produces the most waste and uses more water.
You would choose B because it is recycled, which uses less energy, and produces the least waste. It also will last the longest and therefore has the least environmental cost.
CH156: How is ammonia produced?
Ammonia is a chemical that is used in the manufacture of fertilisers. Nitrogen (from air) and hydrogen (from natural gas)are reacted together, using high temperature and pressure (see CH157), to form ammonia. This is a reversible reaction (see CH154):
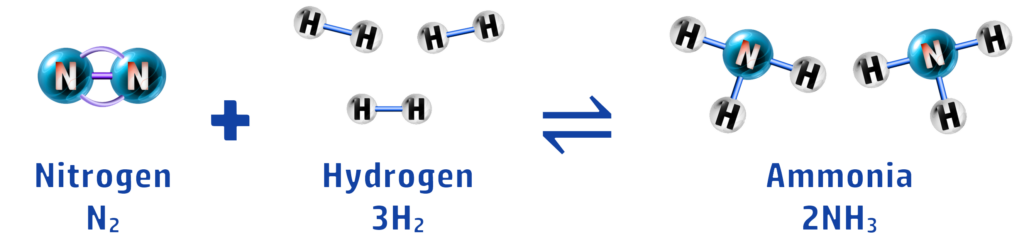
CH157: Recall the conditions for the Haber Process
| Temperature: 450oC | Pressure: 200atm | Catalyst: Iron |
|---|---|---|
| 450oC is the temperature used in the Haber Process. It is a compromise between yield (amount made) and rate of production. (See CH158) | The Haber process is carried out at 200atm. This is a compromise between the cost of the equipment and yield of ammonia (See CH158) | Iron is the catalyst used in the Haber Process. Catalysts speed up the rate of reaction, increasing the yield, but does not change the position of equilibrium (CH158). |
CH160: Explain the difference between corrosion and rusting
Corrosion:
Corrosion is the weakening of a metal over time through rusting, oxidation, or chemical reactions.
The more reactive a metal, the more quickly it corrodes. This is because they lose their electrons faster. Some metals, such as gold, do not corrode at all.
Some metals, such as aluminium, will form a layer of tarnish when they oxidise – stopping oxygen from getting to the surface of the metal.
The statue of liberty is another example of tarnish protecting the metal from corroding further.
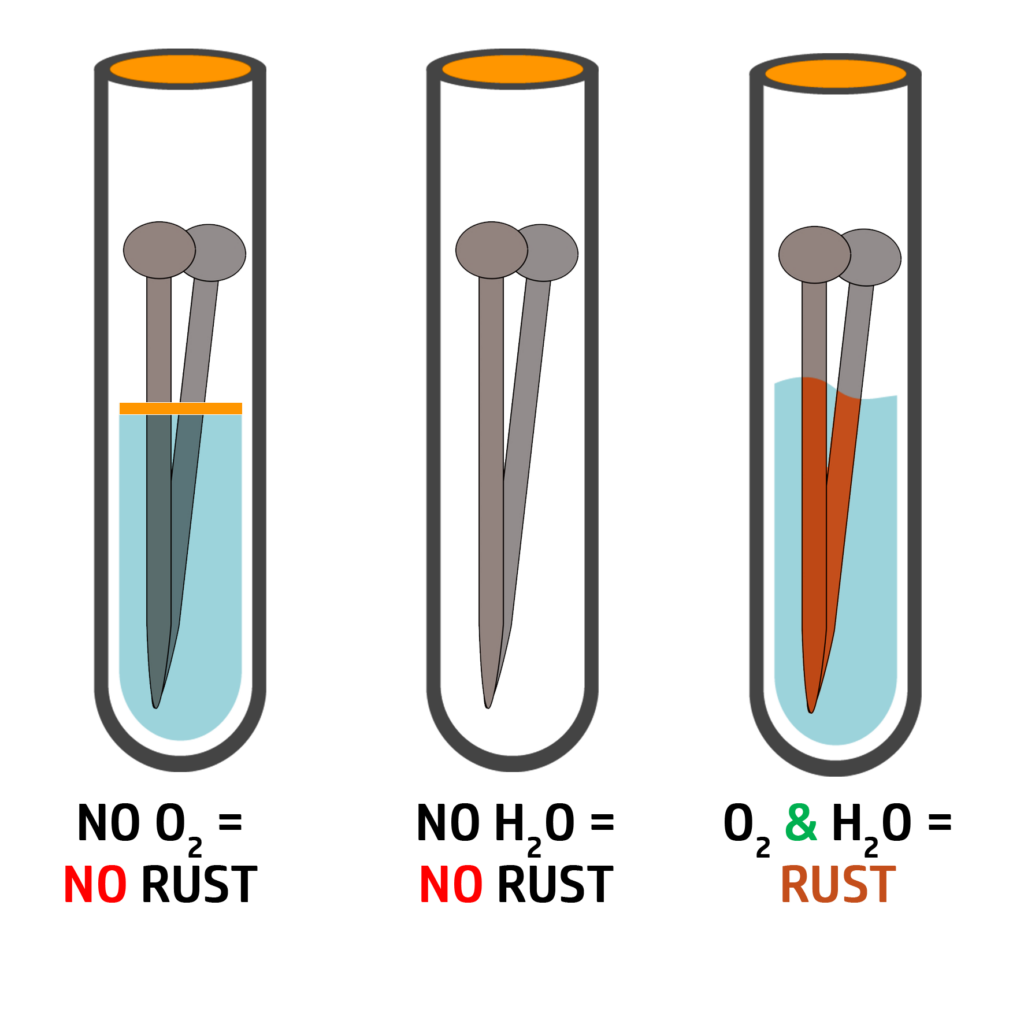
Rusting:
Rusting only occurs in iron. Iron reacting with oxygen is the same as corrosion.
For iron to rust it needs oxygen and water.
As you can see on the right, without oxygen and water, the nail does not rust.
iron + oxygen + water → hydrated iron oxide
CH161: Explain how to prevent corrosion / rusting
There are several ways you can stop rusting and corrosion from occurring:
1. Removing Oxygen: Storing the metal in argon / nitrogen
2. Physical Barriers: Coating the metal with paint, oil or plastic.
3. Sacrificial Protection: Putting a more reactive metal, such as magnesium, over the iron.
The oxygen will then react with the more reactive metal, instead of the iron, stopping the iron from rusting.
The more reactive metal loses its electrons (oxidises) faster.
4. Electroplating: Using electricity to put a thin layer of a less reactive metal on to the one you want to protect (See CH162)
5. Galvanizing is a combination of different rust protections. The iron/steel is coated in a thin layer of zinc. This provides a physical barrier and sacrificial protection.

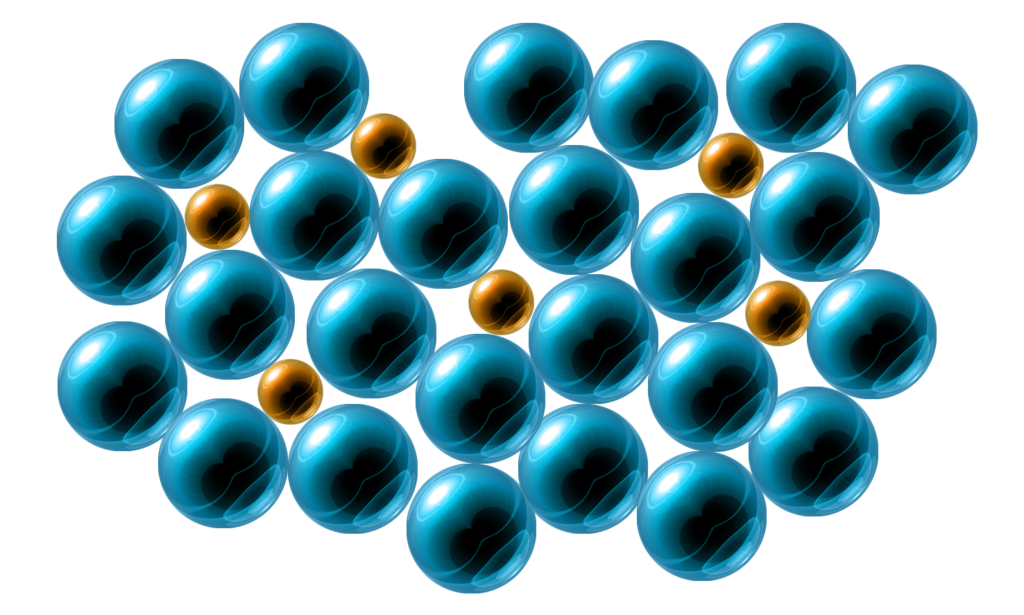
CH163: Explain how alloying makes metals stronger
An alloy is a mixture of a metal and another element – usually another metal.

Pure Metals:
In a pure metal, all the particles are the same size, forming a regular pattern / lattice.
This means that the layers can slide past each other – making them malleable, ductile, and SOFT.
Alloys:
In an alloy, the particles are different sizes.
This means that the layers cannot slide past each other.
This makes alloys much stronger than pure metals.
CH164: Explain the uses and properties of metals and their alloys
| Name: | Metal or Alloy? | Use: | Explanation |
|---|---|---|---|
| Gold | Pure metal | Memory Chips | Excellent conductor of electricity.Soft and expensive. |
| Jewellery Gold | Alloy | Jewellery | Alloy of gold and copper.Stronger than gold but still attractive. |
| Copper | Pure metal | Wires and Pipes | Good conductor of electricity and ductile. Cheaper than gold. |
| Brass | Alloy | Plug Pins | Alloy of copper and zinc.Worse conductor, but stronger than copper. |
| Aluminium | Pure metal | Overhead Cables | Good conductor of electricity.Less dense and cheaper than copper. |
| Magnalium | Alloy | Aircraft Parts | Alloy of magnesium (5%) and aluminium (95%).Stronger than aluminium but still low density. |
CH174: Explain the factors that affect equilibrium
Example: Sulfur trioxide is made by reacting sulfur dioxide with oxygen. It is a reversible reaction. The forward reaction is exothermic.
2SO2 + O2 ⇌ 2SO3
Explain the effects of temperature, concentration, pressure and a catalyst on the position of equilibrium in the formation of sulfur trioxide.
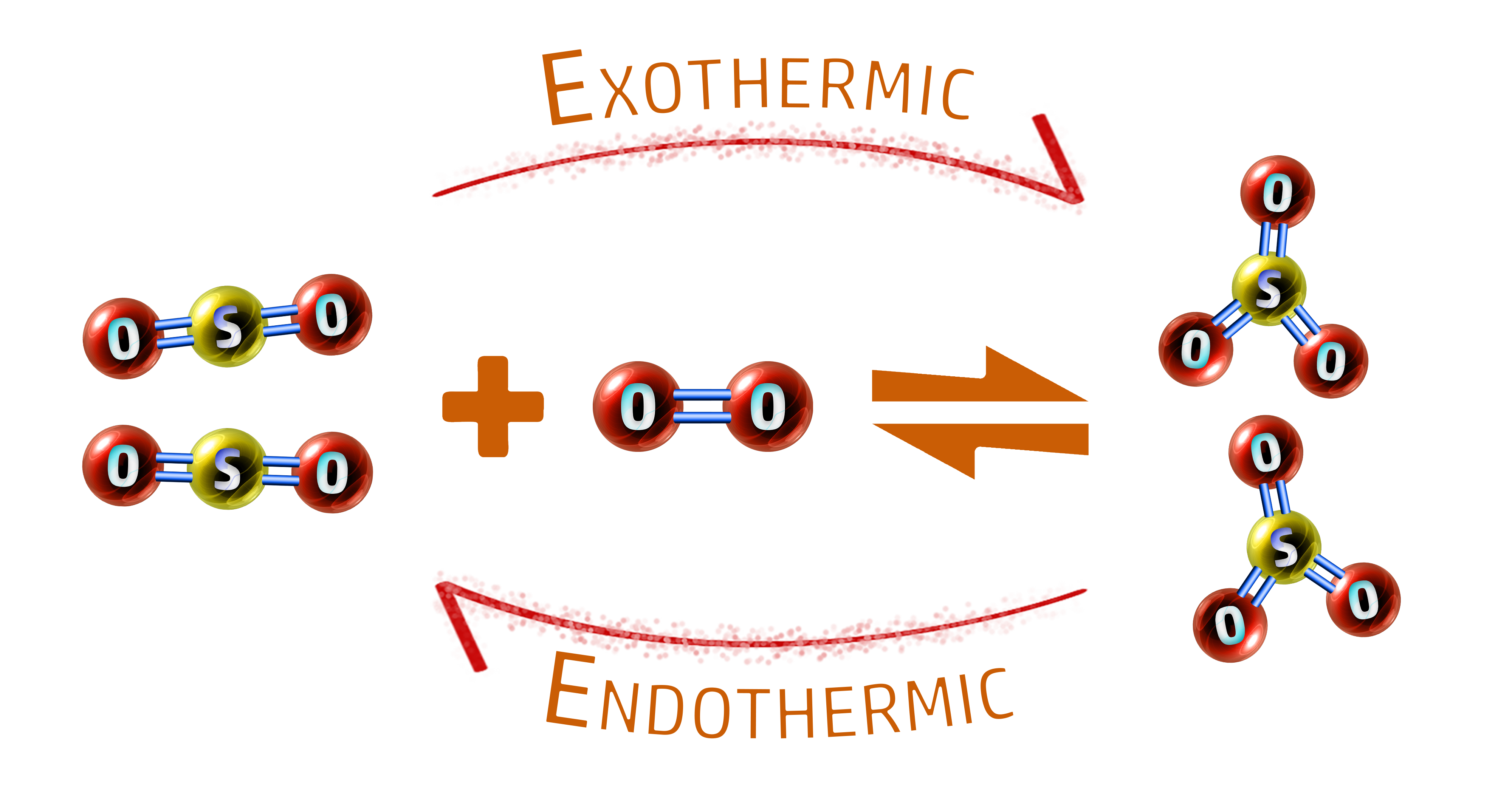
Temperature:
- Increasing the temperature favours the endothermic reaction.
- In the reaction above, the backwards reaction is endothermic so equilibrium will shift to the left.
- This means increasing the temperature will give a lower yield of sulfur trioxide, SO3.
- The yield of the reactants (SO2 & O2) would increase.
- The reason we increase the temperature is because we get that (smaller) yield much faster than at lower temperatures.
Concentration:
- If you increase the concentration of reactants, there will be more frequent collisions.
- This will move the equilibrium to the right and give you a larger yield of ammonia.
Pressure:
- Increasing the pressure favours the side with less molecules.
- In the reaction above, there are three molecules on the left (one O2 molecule and two SO2 molecules) and two molecules on the right (two SO3 molecules).
- Therefore, increasing the pressure will move equilibrium to the right (the side with the least molecules) and increase the yield of sulfur trioxide.
Catalyst:
- Using a catalyst speeds up the rate of attainment of equilibrium, but does not affect the position of equilibrium.
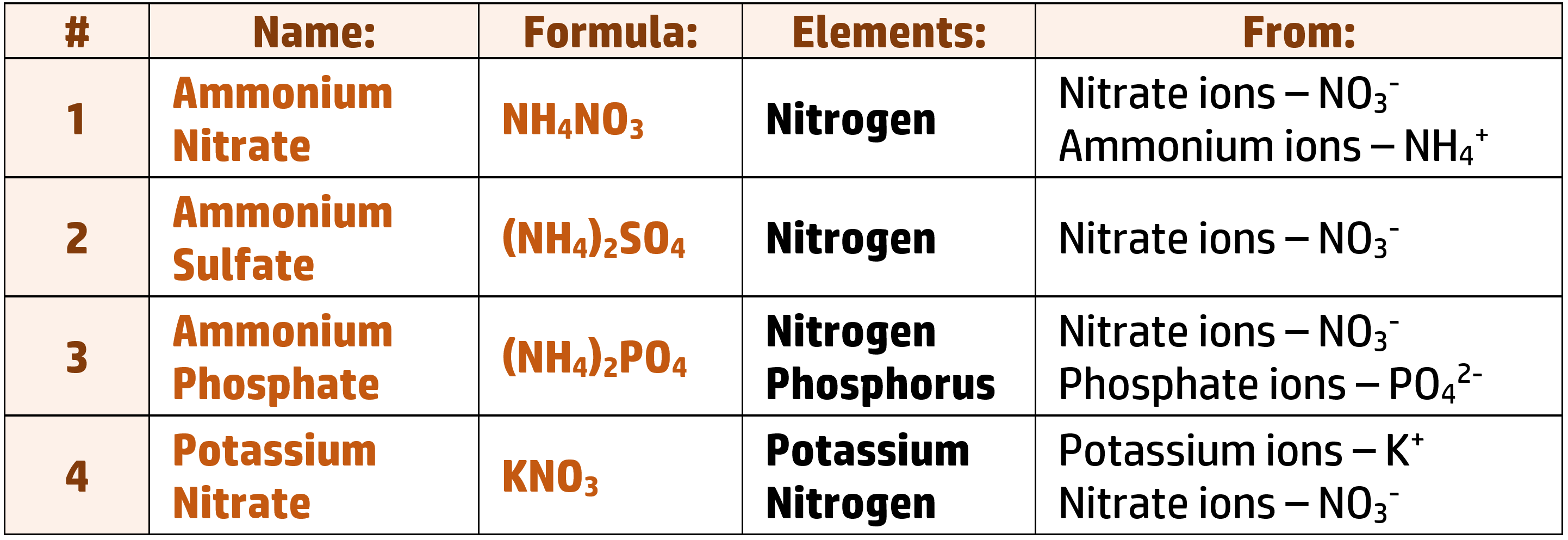
CH175: Describe the importance of fertilisers
When plants grow, they absorb minerals from the soil. Over time, the soil can become mineral deficient which can stop plants from growing – or cause mineral deficiency diseases in the plants. Farmers get round this by using fertilisers.
Fertilisers contain three key minerals: Nitrogen, Phosphorus and Potassium:
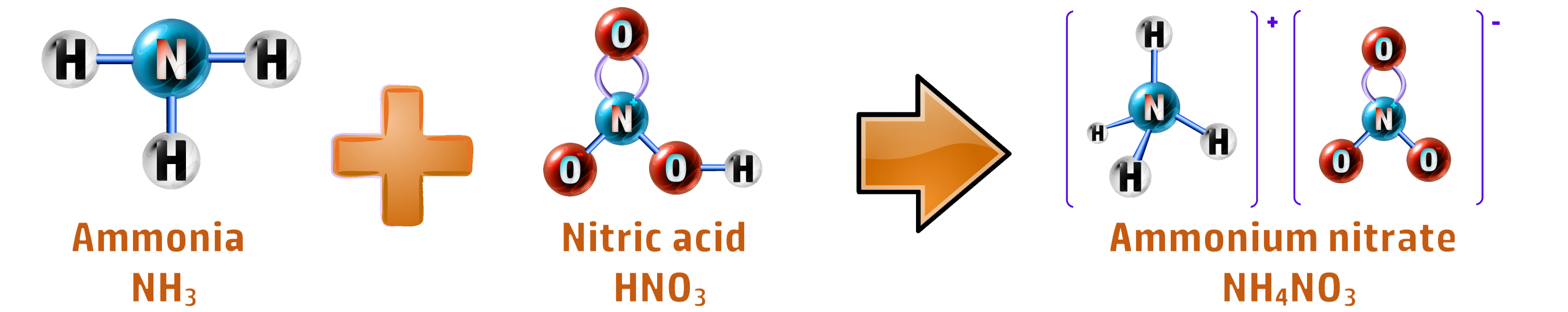
CH176: Describe the formation of ammonium nitrate
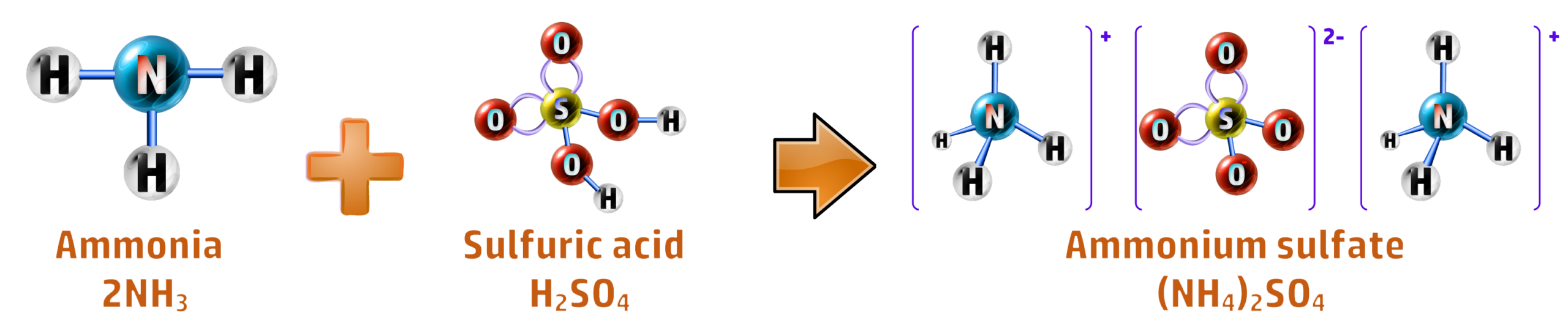
CH177: Describe the steps necessary to produce ammonium sulfate
Ammonium sulfate can be produced both in the Science labs (batch production) and on a large scale (continuous production).
Batch Production: In the lab, it is as simple as a titration between ammonia solution and sulfuric acid, followed by crystallisation:
Batch processes are much slower than large scale continuous processes, so are not good for making large amounts of the fertiliser.
Continuous Processes: In industry, when producing on a larger scale, there are several steps:
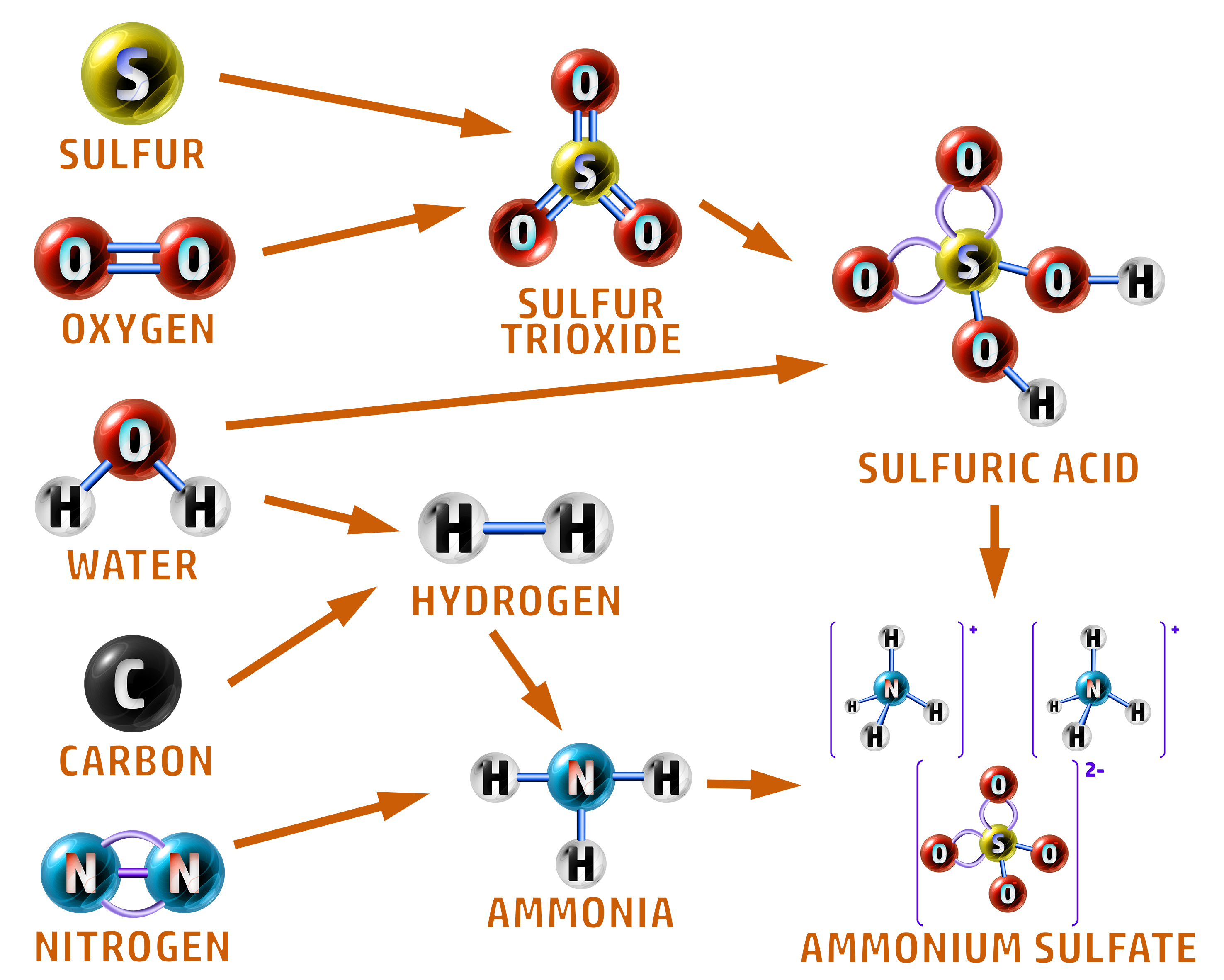
The raw materials are all added continuously:
- Sulfur and oxygen react to form sulfur trioxide.
- Water and sulfur trioxide then form sulfuric acid.
- Water and carbon are used to make hydrogen.
- Hydrogen and nitrogen react to form ammonia (Haber Process).
- Ammonia and sulfuric acid react to form ammonium sulfate.
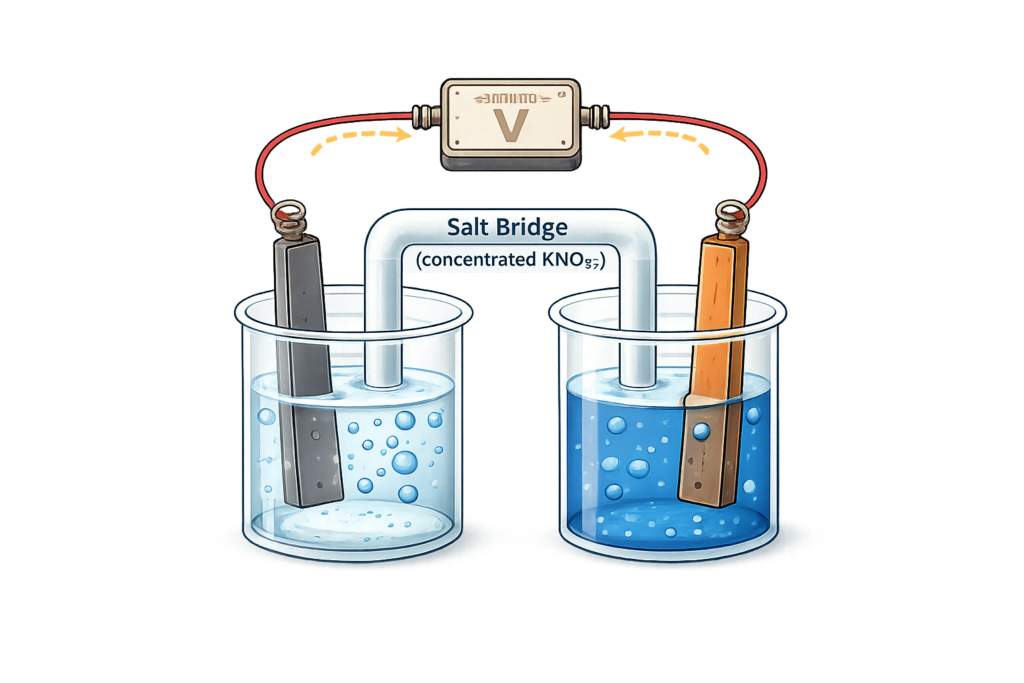
CH178: Describe what a chemical cell is
Chemical cells – such as everyday batteries – produce a voltage when a chemical reaction occurs.
The voltage will start high, then start to decrease until it lowers to zero. At this point, the device will stop working.
They contain two parts:
- Two different metals with solutions of their salts
- A salt bridge to move the ions between the two parts of the cell.
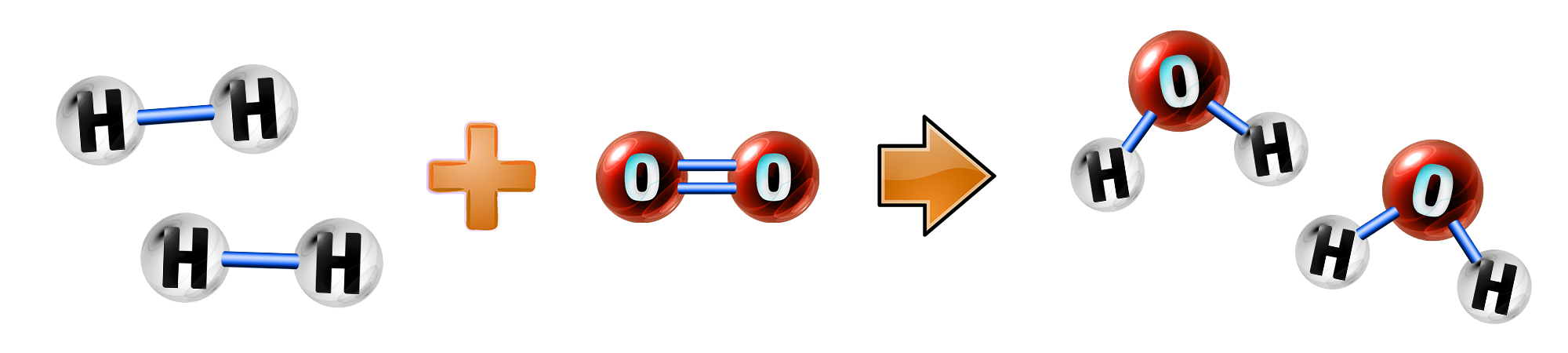
CH179: Explain how a hydrogen fuel cell works
A fuel cell is made up of hydrogen (from fuels) and oxygen (from air)
During the reaction, hydrogen loses electrons (oxidation) to form hydrogen ions:
2H2 → 4H+ + 4e–
The hydrogen ions then react with oxygen, gain electrons (reduction) and form water:
4H+ + 4e– + O2 → 2H2O
CH180: Evaluate the advantages of using a fuel cell instead of fossil fuels
| Advantages of Fuel Cells: | Disadvantages of Fuel Cells: |
|---|---|
| More efficient than using petrol | Hydrogen is a gas – so it takes more space to store |
| No moving parts means less energy lost due to friction | Hydrogen is explosive – so it is harder to store |
| Less reaction steps means less heat lost | Hydrogen is produced from hydrocarbons or electrolysis – both of which use fossil fuels and give off carbon dioxide. |
| No pollutants such as CO2, NO2, SO2 and CO |
