THE RATE AND EXTENT OF CHEMICAL CHANGE

An AQA GCSE chemistry revision page on INSERT CONTENT HERE
CH154: What are Reversible Reactions?
A reversible reaction is any reaction that can go in both the forward direction and backwards direction.
- It has a different symbol: ‘⇌’ instead of ‘→’
- This can be at the same time, or separately.
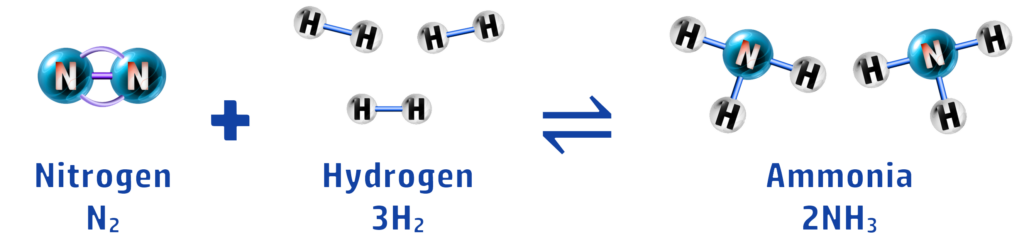
In this reaction, you can see that nitrogen and hydrogen react to form ammonia, and ammonia decomposes to form nitrogen and ammonia. To make it easier to write, we use the ⇌ symbol.
CH155: Explaining Dynamic Equilibrium
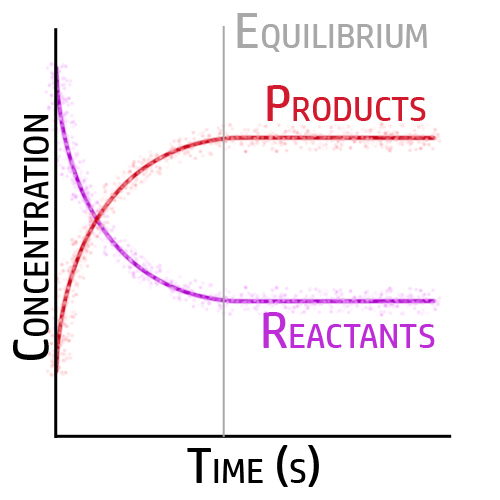
When the forward reaction and backward reaction are occurring at the same rate, and the concentrations do not change – it is at equilibrium.
When the forward and backwards reactions are occurring at the same time and same rate, it is at dynamic equilibrium.
This means that:
- The forward reaction and backward reaction are occurring at the same rate/speed.
- The concentration does not change.
- The forward reaction is occurring.
- The backwards reaction is occurring.
This doesn’t mean that there is 50% of products and 50% of reactants though…
- There may be a higher percentage of products, 80% for example, but it will stay at 80% whilst at equilibrium.
- Similarly, if there is 60% of reactants and it is at dynamic equilibrium, it will stay at 60%.
Remember, for something to be at dynamic equilibrium, it must be in a closed system – this means that no products and reactants should be allowed to escape!
CH156: How is ammonia produced?
Ammonia is a chemical that is used in the manufacture of fertilisers. Nitrogen (from air) and hydrogen (from natural gas)are reacted together, using high temperature and pressure (see CH157), to form ammonia. This is a reversible reaction (see CH154):

CH157: Recall the conditions for the Haber Process
| Temperature: 450oC | Pressure: 200atm | Catalyst: Iron |
|---|---|---|
| 450oC is the temperature used in the Haber Process. It is a compromise between yield (amount made) and rate of production. (See CH158) | The Haber process is carried out at 200atm. This is a compromise between the cost of the equipment and yield of ammonia (See CH158) | Iron is the catalyst used in the Haber Process. Catalysts speed up the rate of reaction, increasing the yield, but does not change the position of equilibrium (CH158). |
CH158: Explain the effect of temperature and pressure on the Haber Process

Temperature
If you increase the temperature, you will always get a higher yield for the endothermic reaction.
In the Haber process, the backwards reaction is endothermic. This means that by increasing the temperature, you will have a lower yield of ammonia (and more nitrogen/hydrogen).
450oC is a compromise between yield and rate.
Pressure / Concentration
If you increase the pressure / concentration, the particles become closer – meaning there are more collisions.
This favours the side with the least particles – the Haber Process has a ratio of 4:2 in favour of ammonia – giving a higher yield of ammonia.
Increasing the pressure is very expensive, however.
200atm is a compromise between yield and cost.
CH197: Core Practical: Investigating Rates of Reaction
Part 1: Measuring the production of a gas (in the reaction between hydrochloric acid and marble chips)
Hydrochloric acid + calcium carbonate → calcium chloride + carbon dioxide + water
2HCl (aq) + CaCO3 (s)→ CaCl2 (aq) + CO2 (g) + H2O (l)
Key Steps: Mix → Bung → Timer → Gas → Repeat
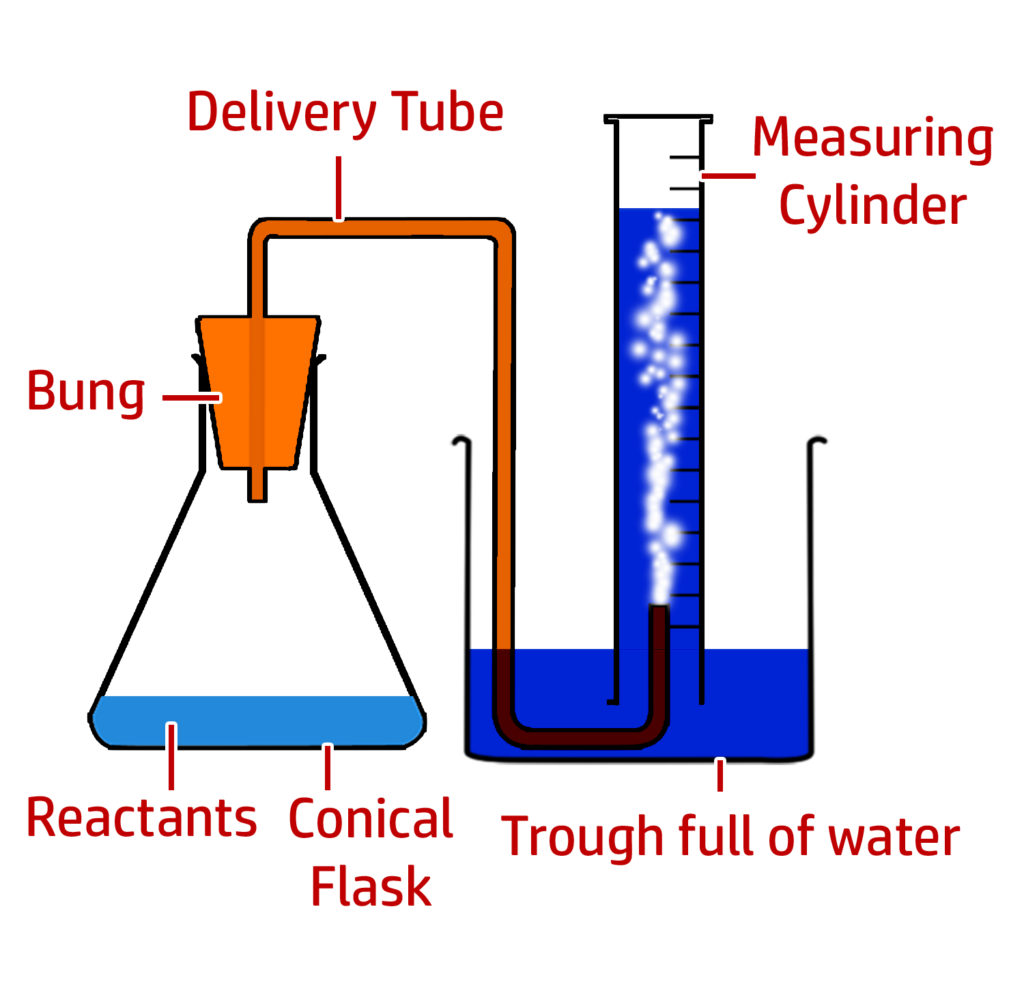
Method:
- Mix the hydrochloric acid and marble chip.
- Add the bung to the conical flask and start the timer.
- Stop the timer when 50cm3 of carbon dioxide gas has been produced.
- Repeat for different {concentrations / temperatures / surface areas}
Variables:
- Independent Variable: Change the concentration, surface area OR temperature (the question will give you this info).
- Dependent Variable: Measure the time it takes to produce 50cm3 of gas.
- Control Variable: Keep the volume of acid and mass of marble chip the same (as well as the {concentration/surface area/temperature} that wasn’t changed earlier.
Results:
Increasing the {temperature / concentration / surface area} will mean a faster reaction. This will mean that carbon dioxide is produced quicker so the {water is displaced quicker / gas syringe fills up quicker}.
Part 2: Investigating the disappearing cross:
Hydrochloric acid + sodium thiosulfate → sodium chloride + sulfur dioxide + sulfur + water
2HCl (aq) + Na2S2O3 (aq)→ 2NaCl (aq) + SO2 (g) + S (s) + H2O (l)
Key Steps: Cross → Mix → Timer → Disappear→ Repeat
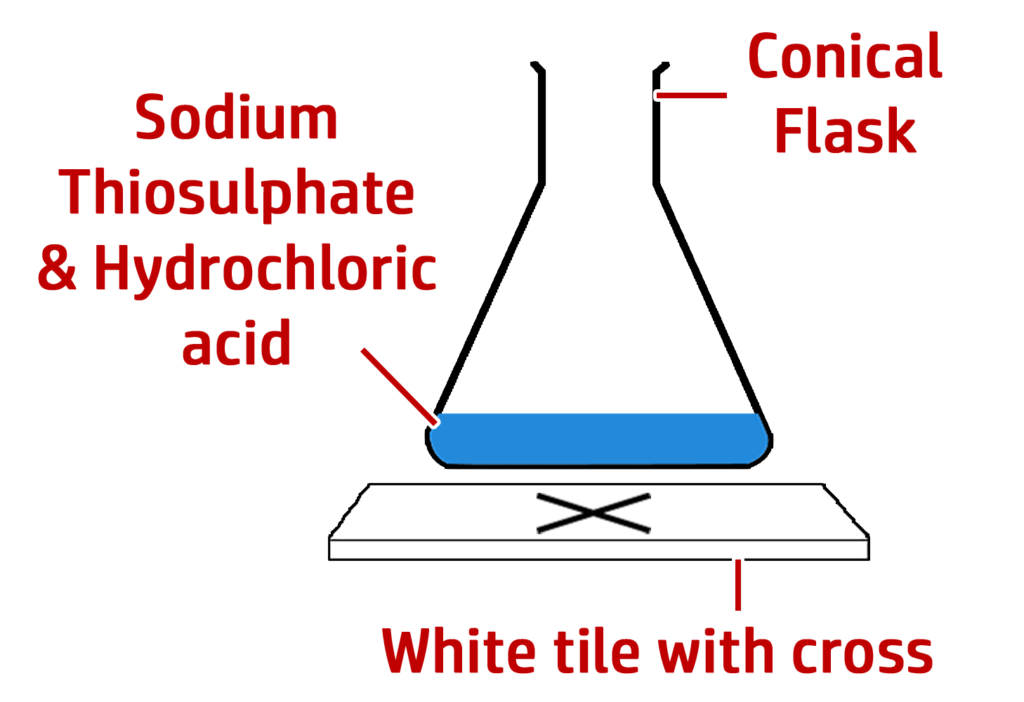
Method:
- Draw a cross on a white tile.
- Mix the acid and sodium thiosulfate and start the timer.
- Stop the timer when the cross disappears.
- Repeat for different {concentrations / temperatures}
Variables:
- Independent Variable: Change the concentration or the temperature.
- Dependent Variable: Measure the time it takes for the cross to disappear.
- Control Variable: Keep the volume of the acid and sodium thiosulfate the same, as well as whichever you didn’t change out of temperature/concentration.
Results:
Increasing the {temperature / concentration} will mean a faster reaction. This will mean that solid sulfur is produced quicker so the cross will disappear quicker.
CH198: Identify what is needed for a reaction to occur

For a chemical reaction to occur, two things need to happen:
- A collision between your reactant particles
- The collisions need to have enough energy to break bonds in the reactants. This is called the Activation Energy.
If a collision occurs between two particles without enough energy, there will be no reaction – therefore no products will be formed.
CH199: Collision Theory: Explain how to speed up a chemical reaction

Increasing Temperature:
If you increase the temperature, the particles have more kinetic energy.
There will be more successful collisions every second – meaning a faster rate of reaction.

Increasing Concentration:
If you increase the concentration, there are more particles in the same volume.
This means there are more frequent collisions – increasing the rate of reaction.
You can also increase the pressure, which means there particles will be closer together, meaning more frequent collisions.
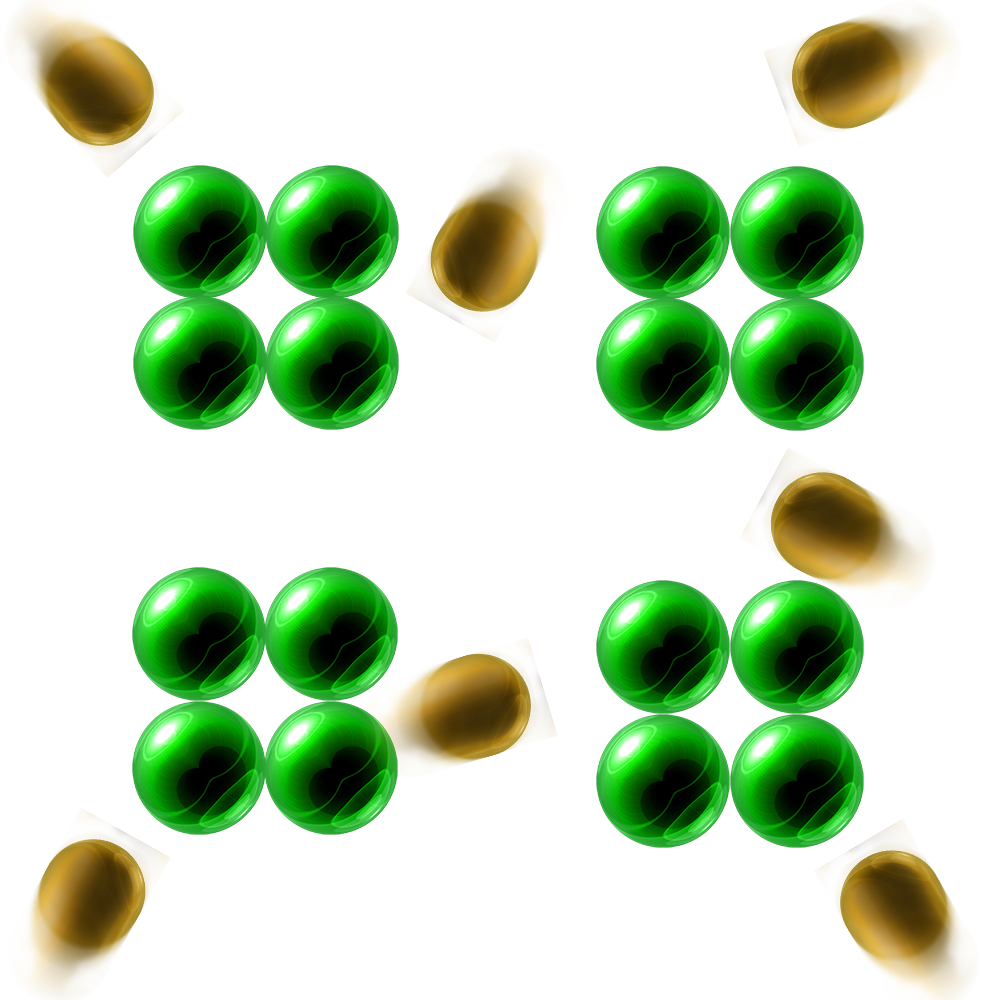
Increasing Surface Area:
If you increase the surface area of a solid, you break it up into smaller pieces.
This means that there is a larger surface area-to-volume ratio – more solid is exposed to the acid.
This will mean that there are more frequent collisions and a faster rate of reaction.
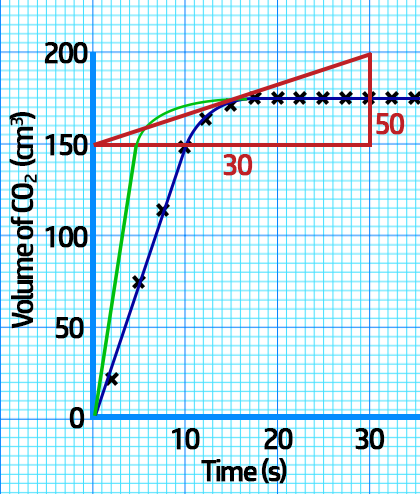
CH200: Interpreting Rate Graphs
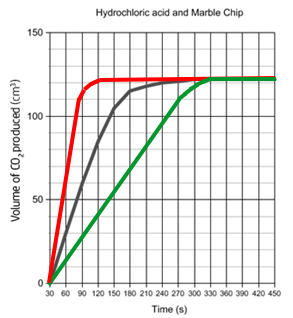
Sketching changing conditions:
If you increase the temperature / concentration / surface area, the reaction will be quicker, and the rate graph will be steeper as shown in red below.
If you decrease the temperature / concentration / surface area, the reaction will be slower, and the rate graph will be shallower as shown in green on the left.
Note that in both situations, the total volume at the end is always the same – the flat bit should always be at the same level.
The reason the rate of reaction decreases is because, as the reaction proceeds, there are less reactants, meaning less frequent collisions.
Rate calculations:
You can be asked two different ways of calculating the rate of reaction:
1. Calculating the mean / average rate of reaction: The mean rate of reaction is the average rate at a specific time. To calculate this, all you need is the time and the volume.
Example: In the first 50s of a reaction, 25cm3 of carbon dioxide was produced. Calculate the mean rate.
- Mean rate = Volume (cm3) ÷ Time (s)
- Mean rate = 25cm3 ÷ 50s = 0.5cm3/s.
2. Calculating the rate of reaction at a SPECIFIC time: To calculate this, you need to draw a tangent at the time given and then divide the change in volume (cm3) by the change in time (s).
Example: Calculate the actual rate of CO2 production at 15s.
From the tangent, you can see that at 15 seconds:
- The change in volume = 200cm3 – 150cm3 = 50cm3.
- The change in time = 30s – 0s = 30s
- Actual rate = Change in volume (cm3) ÷ Change in time (s)
- Actual rate = 50cm3 ÷ 30s = 1.7cm3/s.