ENERGY CHANGES

An AQA GCSE chemistry revision page showing INSERT CONTENT HERE.
CH202: Describe exothermic and endothermic reactions in terms of heat and temperature

Exothermic:
- In an exothermic reaction, heat is given out to the surroundings.
- This means that:
- The temperature increases.
- Heat energy is given out to the surroundings (so the surroundings get hotter).
- The products have less energy than the reactants. (See CH203)

Endothermic:
- In an endothermic reaction, heat is taken in from the surroundings.
- This means that:
- The temperature decreases.
- Heat energy is taken in from the surroundings (so the surroundings get colder).
- The products have more energy than the reactants. (See CH203)
CH203: Draw reaction profiles for exothermic and endothermic reactions
EXOTHERMIC
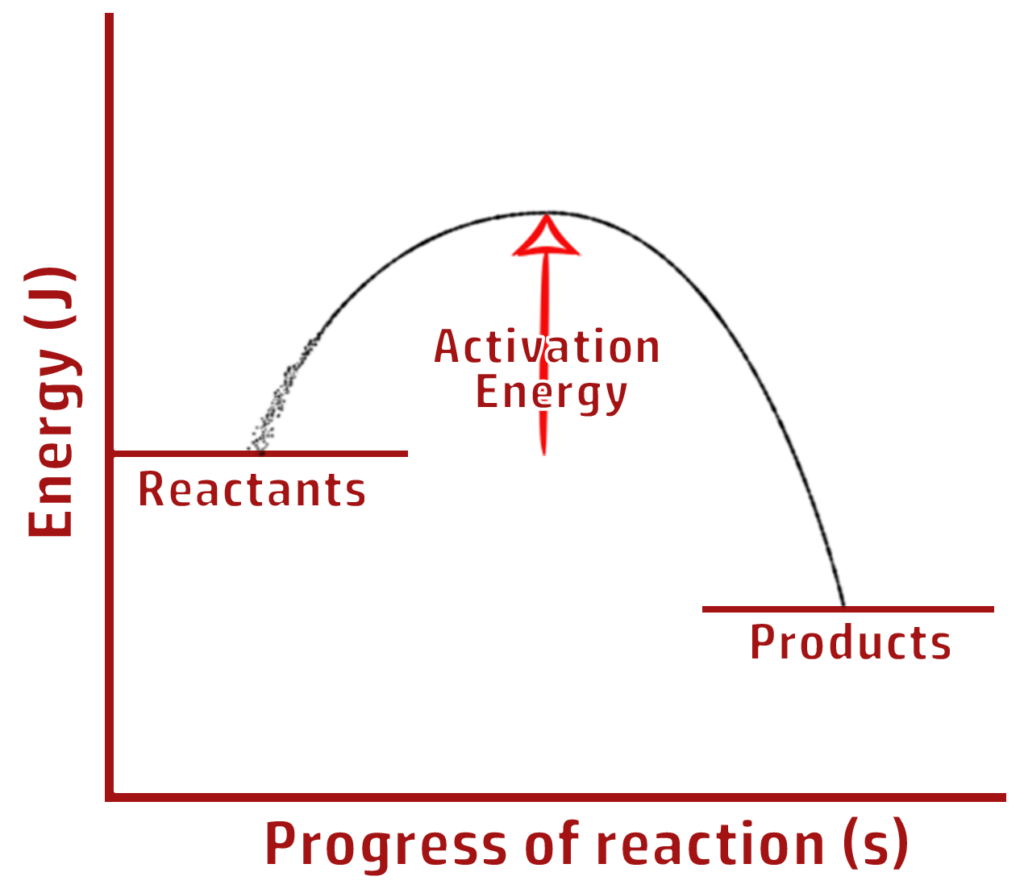
Clue: More people have exited the shop (products have less energy) so it is EXOthermic.
ENDOTHERMIC
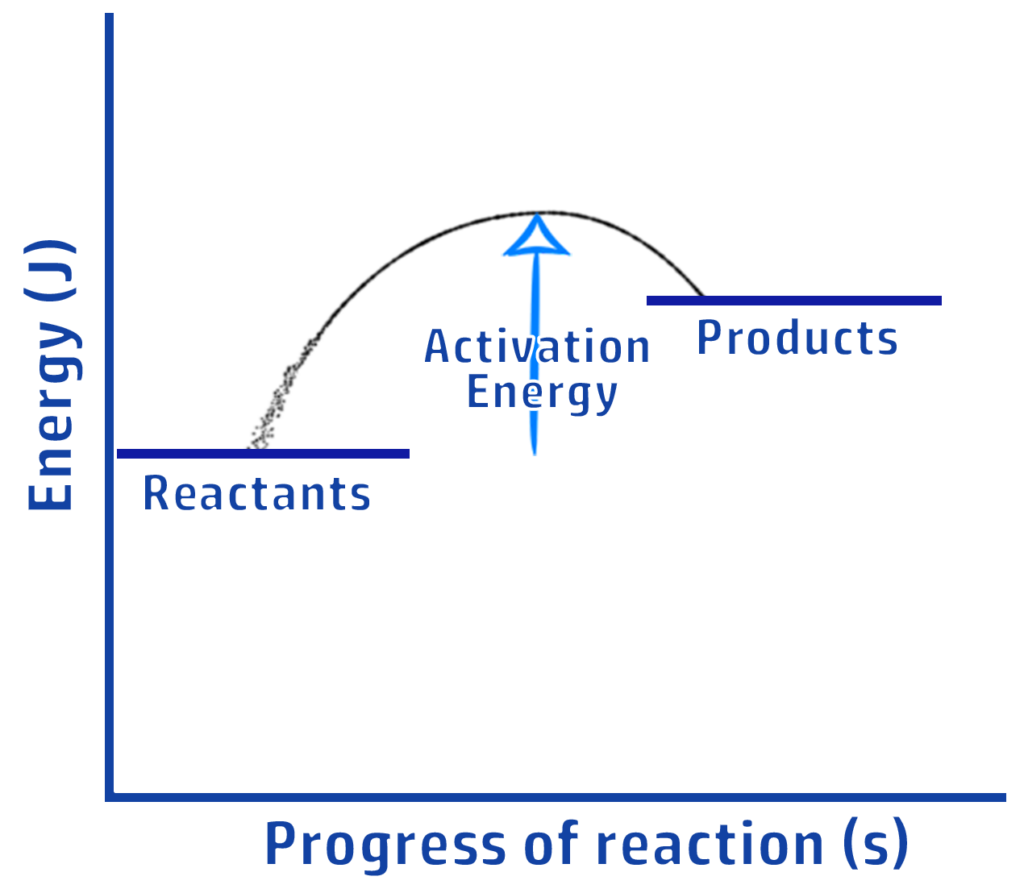
Clue: More people have entered the shop (products have more energy) so it is ENDOthermic.
CH204: Describe bond breaking and bond forming
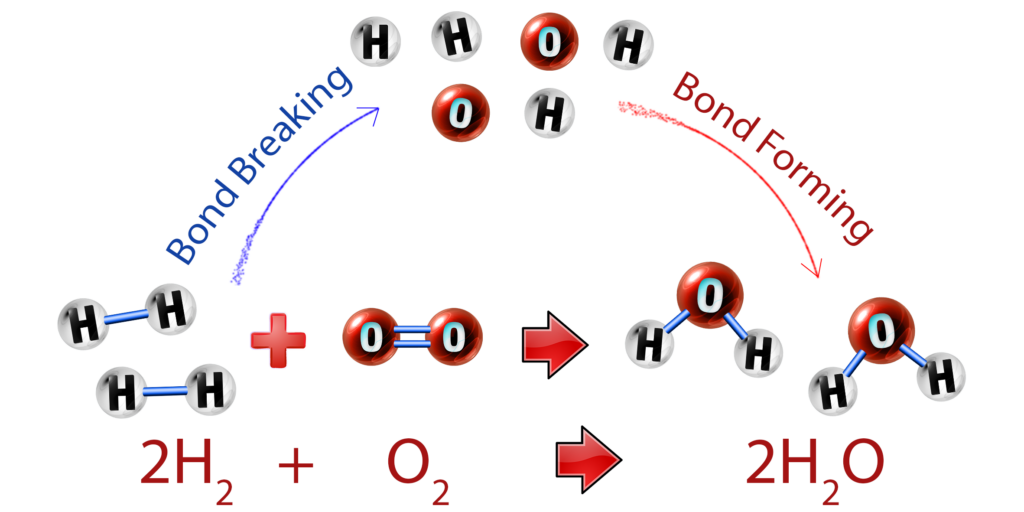
There are two key things to remember that occur in every chemical reaction:
- Bond breaking is endothermic – heat energy is taken in to break the bonds.
- Bond forming is exothermic – heat energy is given out to the surroundings when new bonds form.
CH205: Explain what makes a reaction exothermic overall
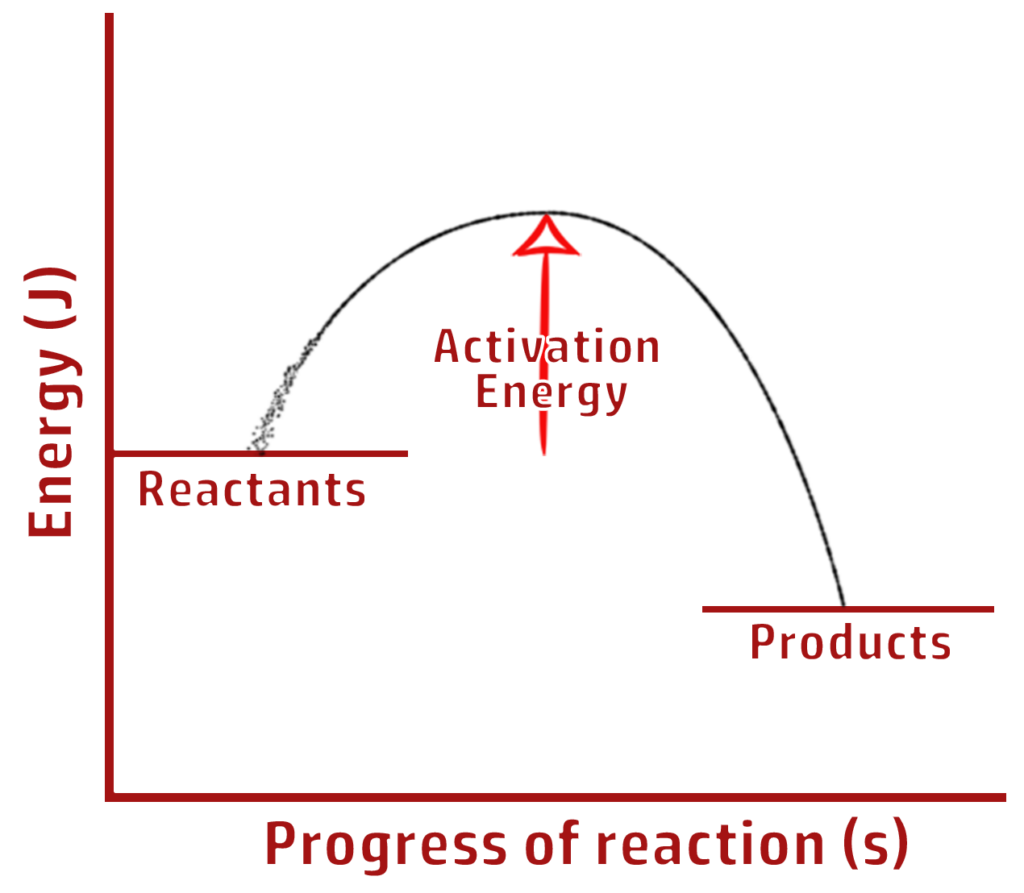
For a reaction to be exothermic overall, more energy must be given out to the surroundings (when bonds form) than taken in (when bonds are broken).

For a reaction to be endothermic overall, more heat energy must be taken in from the surroundings (when bonds are broken) than given out (when new bonds are formed).
CH206: Calculating Energy Changes (H)
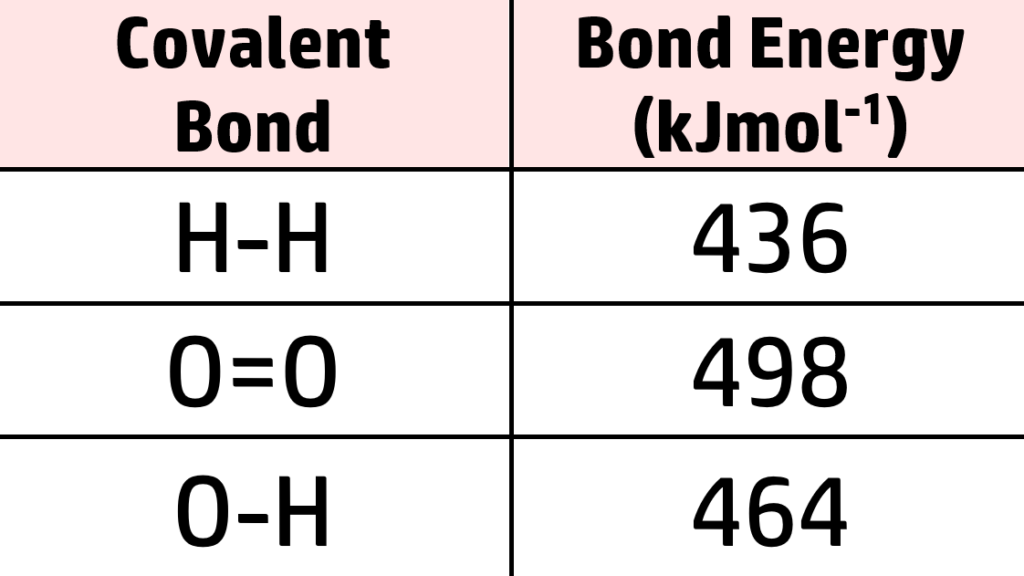
In a chemical reaction, you need a certain amount of energy to break bonds (endothermic) and a certain amount of energy is given out to form new bonds (exothermic). You can, therefore, use a reference table (such as the one on the right) to work out if a reaction is exothermic or endothermic.
Example: Calculate the energy change in the reaction between hydrogen and oxygen to form water:
2H2 + O2 → 2H2O
Step 1: Work out the energy needed to break the bonds in H2 and O2:
2H2 + O2 →
We have 2 hydrogen molecules and one oxygen molecule.
From the table above, you can see that:
- 436kJmol-1of energy is needed to break the bonds in each hydrogen molecule. We have 2 molecules of hydrogen, so 2 x 436 = 872kJmol-1
- We have one oxygen molecule, so 498kJmol-1 is needed.
Therefore, the total energy to break the bonds is 872 + 498 = 1370kJmol-1 is needed to break all bonds.
Step 2: Work out the energy needed to form the bonds in H2O:
→ 2H2O
Each water molecule has 2 hydrogens attached to one oxygen. Therefore, each H2O has two O=H bonds.
- 464kJmol-1of energy is needed to break the O- H bond.
- Each water has 2xO-H bonds, so each water = 928kJmol-1.
We have 2 water molecules, so 928×2 = 1856kJmol-1 needed to break all bonds in 2H2O.

Step 3: Work out the energy change, which is bond breaking (reactants) – bond forming (products):
- 1370kJmol-1 – 1856kJmol-1 = -486kJmol-1
- Make sure to put the sign (+ or -) – this is worth one mark!
