CHEMICAL CHANGES

An AQA GCSE chemistry revision page showing INSERT CONTENT HERE.
CH109: Identify the ions that make something acidic

Acids always contain H+ ions. Examples include hydrochloric acid, HCl, nitric acid, HNO3, and sulfuric acid, H2SO4.
Alkalis usually contain OH– ions. Examples include sodium hydroxide, NaOH, magnesium oxide, Mg(OH)2 and aluminium hydroxide, Al(OH)3
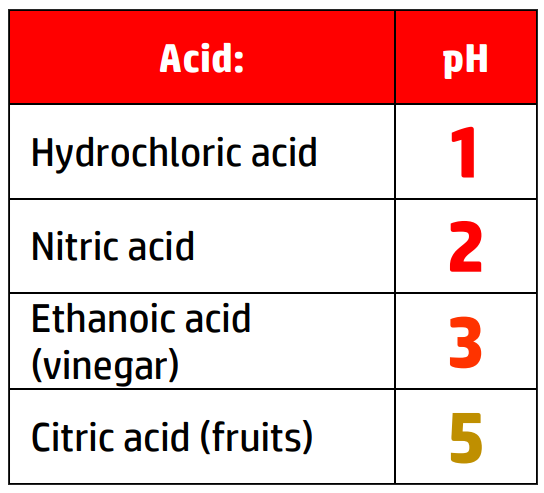
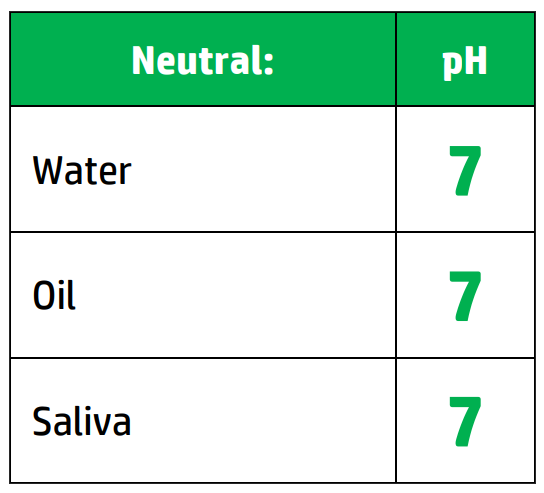
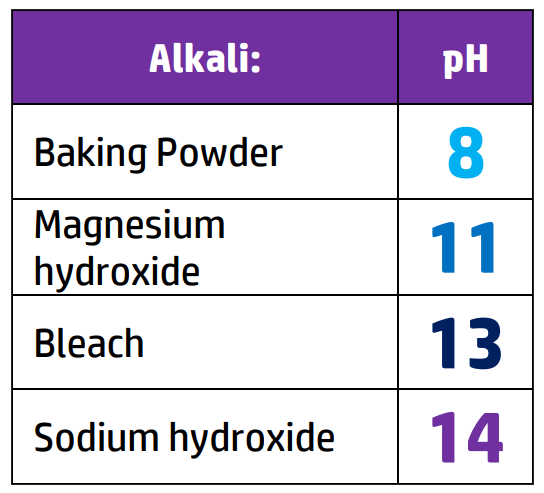
CH110: Identify the pH of acids and alkalis
CH111: Identify the colour changes in different indicators
| Indicator | Colour in acid | Colour when neutral | Colour in alkali |
|---|---|---|---|
| Phenolphthalein | Colourless | Colourless | Pink |
| Methyl Orange | Red | Orange | Yellow |
| Litmus Solution | Red | Purple | Blue |
CH112: Describe the terms ‘dilute’ and ‘concentrated’
Dilute

The more dilute an acid is, the less H+ ions there are per litre (dm3)
Concentrated

The more concentrated an acid is, the more H+ ions there are per litre (dm3)
CH113: Explain the difference between a weak and a strong acid
Strong Acids
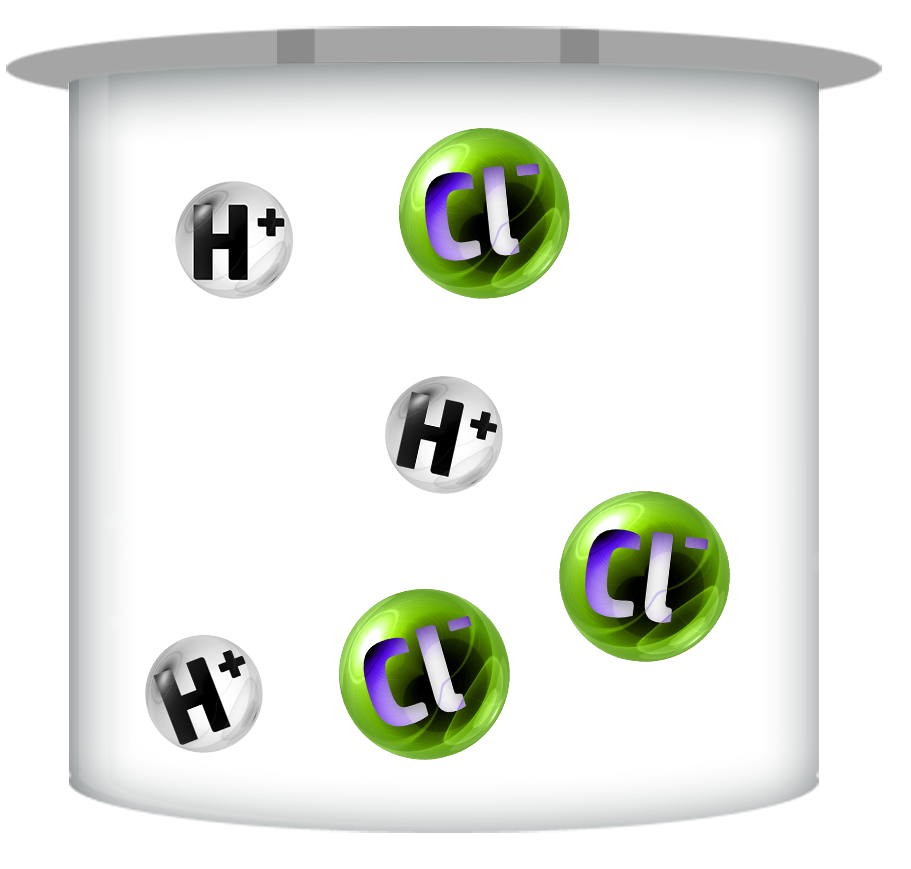
Strong acids dissociate / ionise (split up) completely – producing more H+ ions.
They have lower pH’s (0-2)
Weak Acids
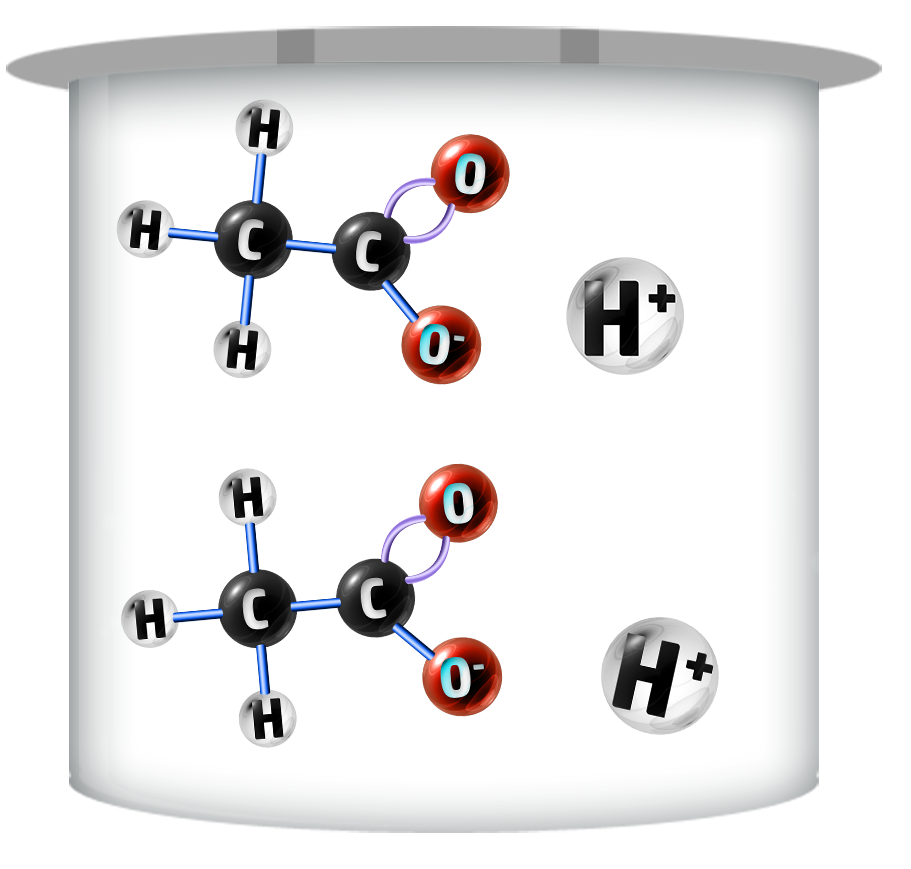
Weak acids do not fully ionise – producing fewer H+ ions.
Their pH ranges are higher (3-6)
CH114: Describe what a base is
A base is a chemical compound that can react with an acid and neutralise it.
There are two types of bases:
- Oxides/Hydroxides which produce a salt and water.
- Carbonates which produce a salt, carbon dioxide and water.
CH115: Explain the difference between an alkali and a base
Similarity: Both bases and alkalis neutralise acids.
Difference: Alkalis are soluble. Bases can be both soluble or insoluble.
Example:
- Sodium hydroxide and copper oxide both neutralise acids, so are both bases.
- Sodium hydroxide is soluble, so it is a base and an alkali.
- Copper oxide is insoluble, so it is a base, but not an alkali.
CH116: Write word equations for reactions between metals and acids
All acids react with metals to produce a salt and hydrogen:

Step 1: To name the salt, take the name of the metal (sodium) and add the salt ending, which you get from the acid:
- Hydrochloric acid produces a chloride salt.
- Nitric acid produces a nitrate salt.
- Sulfuric acid produces a sulfate salt.
Here we have nitric acid, so our salt becomes sodium nitrate.
Step 2: If you only have a metal (i.e. no hydroxide or carbonate), simply add hydrogen to the end of the word equation.
If you have a hydroxide, see CH117 and if you have a carbonate look at CH118.
CH117: Write word equations for reactions between metal hydroxides and acids
All acids react with metal hydroxides to produce a salt and water:
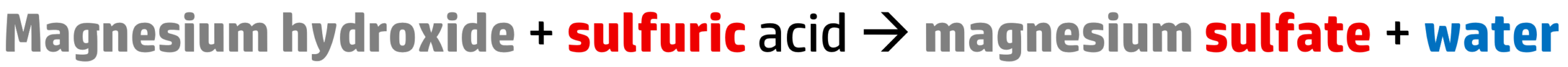
Step 1: To name the salt, take the name of the metal (magnesium) and add the salt ending, which you get from the acid:
- Hydrochloric acid produces a chloride salt.
- Nitric acid produces a nitrate salt.
- Sulfuric acid produces a sulfate salt.
Here we have sulfuric acid, so our salt becomes magnesium sulfate.
Step 2: If you have a metal hydroxide (i.e. magnesium hydroxide), simply add water to the end of the word equation.
If you just have a metal (no hydroxide or carbonate), see CH116 and if you have a carbonate look at CH118.
CH118: Write word equations for reactions between metal carbonates and acids
All acids react with metal carbonates to produce a salt, water AND carbon dioxide:

Step 1: To name the salt, take the name of the metal (calcium) and add the salt ending, which you get from the acid:
- Hydrochloric acid produces a chloride salt.
- Nitric acid produces a nitrate salt.
- Sulfuric acid produces a sulfate salt.
Here we have hydrochloric acid, so our salt becomes calcium chloride.
Step 2: If you have a metal carbonate (i.e. calcium carbonate), simply add water + carbon dioxide to the end of the word equation.
If you just have a metal (no hydroxide or carbonate), see CH116 and if you have a hydroxide look at CH117.
CH119: Describe the test for hydrogen
Hydrogen is a flammable gas.
If you take a lit splint and add it to a test tube containing hydrogen, you will hear a squeaky pop.

CH120: Describe the test for carbon dioxide
Carbon dioxide turns limewater cloudy.
Bubble the gas through limewater and if it goes cloudy/milky, carbon dioxide is present.
The carbon dioxide reacts with the limewater (calcium hydroxide) to form calcium carbonate – a white solid.
CH121: Describe what is seen during neutralisation reactions
The clue is in the state symbols. Look at the chemical equation below. What would you be able to see during this reaction?

From this reaction, you would be able to see two things:

You have a solid (s) on the left…but not on the right. You would see it disappear / dissolve


You have a gas (g) on the right…you can’t say you would see a gas! You would see bubbles/fizzing!
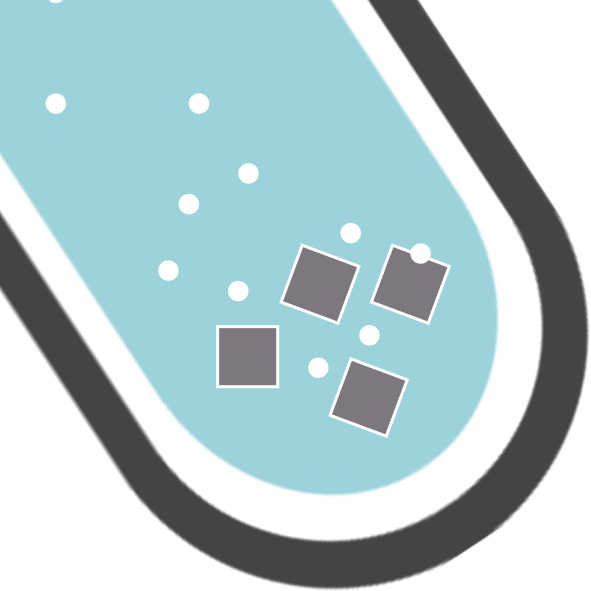
CH122: Explain, in terms of ions, what is happening during neutralisation
Every time a hydroxide (an alkali containing OH– ions) reacts with an acid (which contains H+) ions, the H+ and OH– ions react together to form water.
This happens in every acid-hydroxide reaction, giving the ionic equation of:
H+ + OH– → H2O

Every time a carbonate (a base containing CO32- ions) reacts with an acid (which contains H+) ions, the H+ and CO32- ions react together to form carbon dioxide and water.
This happens in every acid-carbonate reaction, giving the ionic equation of:
H+ + CO32- → CO2 + H2O

CH123: Link the number of H+ ions to the concentration of an acid
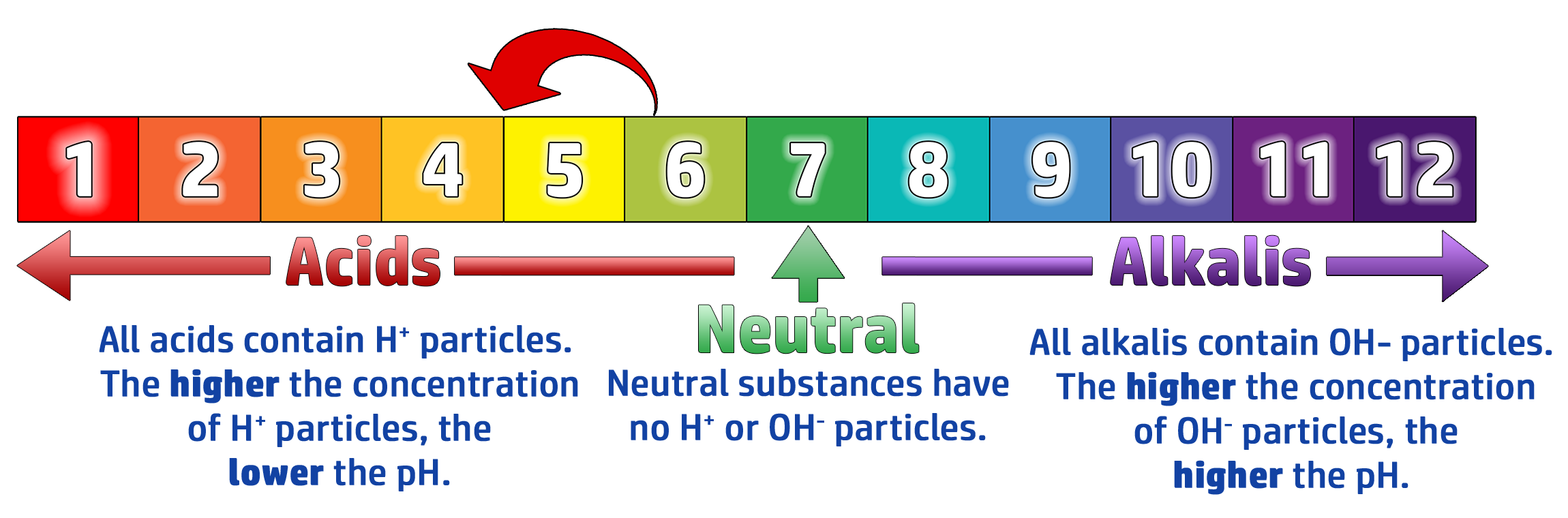
If you increase the number of H+ ions, the pH will go down towards 1.
If you increase the number of OH– ions, the pH will go up towards 14.
CH124: Calculate the change in pH and concentration
Every time the pH decreases by one, the concentration of H+ ions increases by 10 times.
Example: Calculate how much more concentrated hydrochloric acid is, with a pH of 0, than ethanoic acid, with a pH of 4.
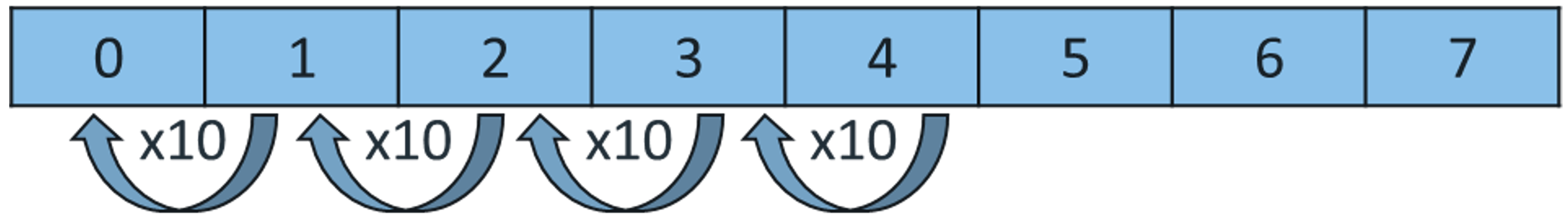
As you can see, the pH has gone down by 4, so it is 10 x 10 x 10 x 10 times more concentrated = 10,000 times more concentrated.
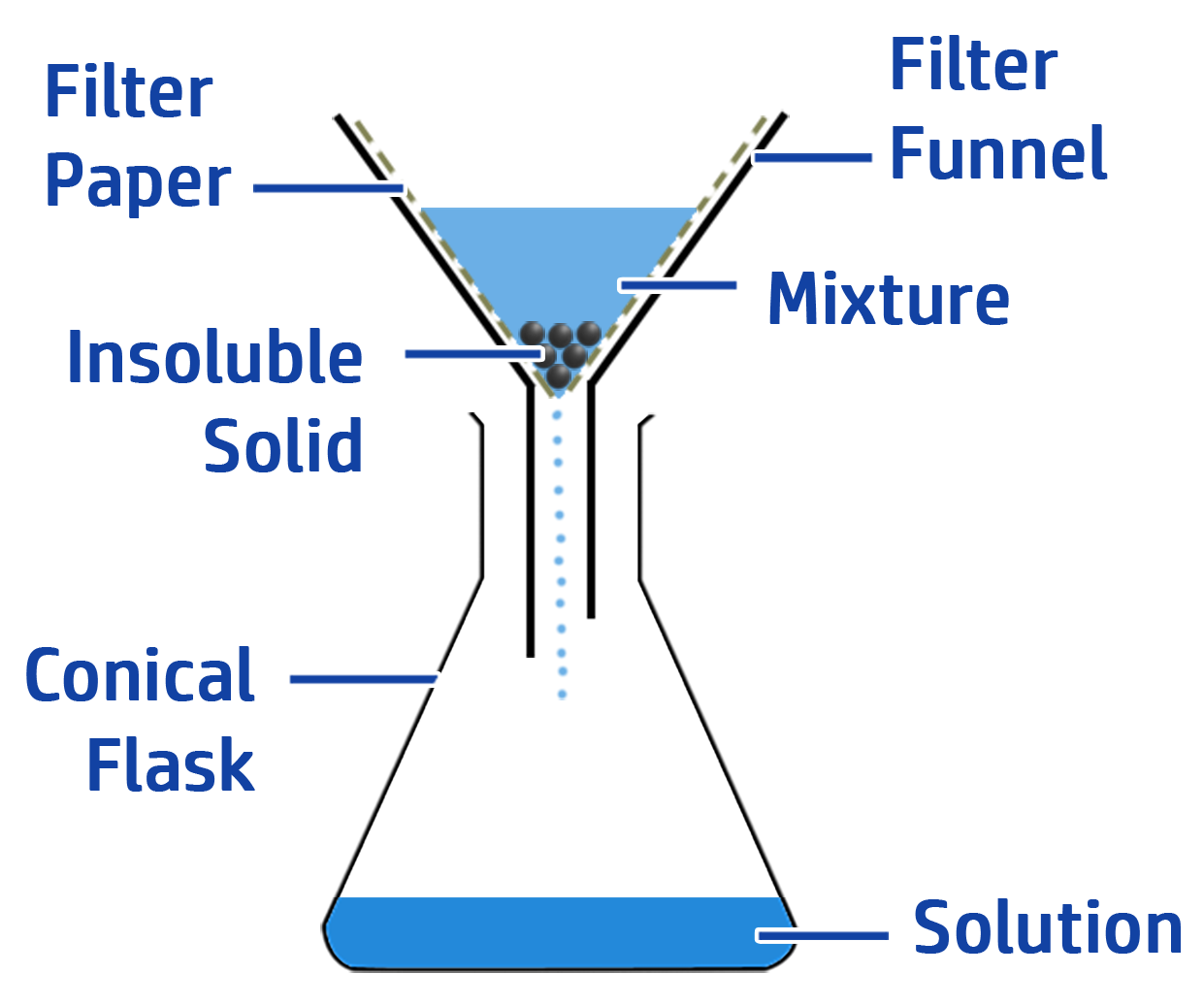
CH125: Describe how to make a salt from an insoluble reactant
If you want to make a salt from an insoluble reactant you can use filtration.
When you add enough of the insoluble reactant (e.g. copper oxide), the solution will become neutral.
If you add more copper oxide to the solution, the pH will not change because the copper oxide will not dissolve or react. Therefore, you can add in excess copper oxide to make sure the solution is neutral, then filter it to leave you with your neutral salt. See CH126 for more detail.
CH126: Core Practical: Preparing Copper Sulfate Crystals

Copper Oxide + Sulfuric Acid → Copper Sulfate + Water
Example: How can you produce soluble copper sulphate from sulphuric acid and insoluble copper oxide?
Key Steps: Mix → Neutral → Filter → Heat → Cool→ Dry
Phase One: Making the Copper Sulfate
- Mix the copper oxide with warm sulfuric acid (heating the acid speeds up the reaction).
- Continue until neutral. You can check this with pH paper.
- Filter the solution using a filter funnel and filter paper. This removes the excess copper oxide.

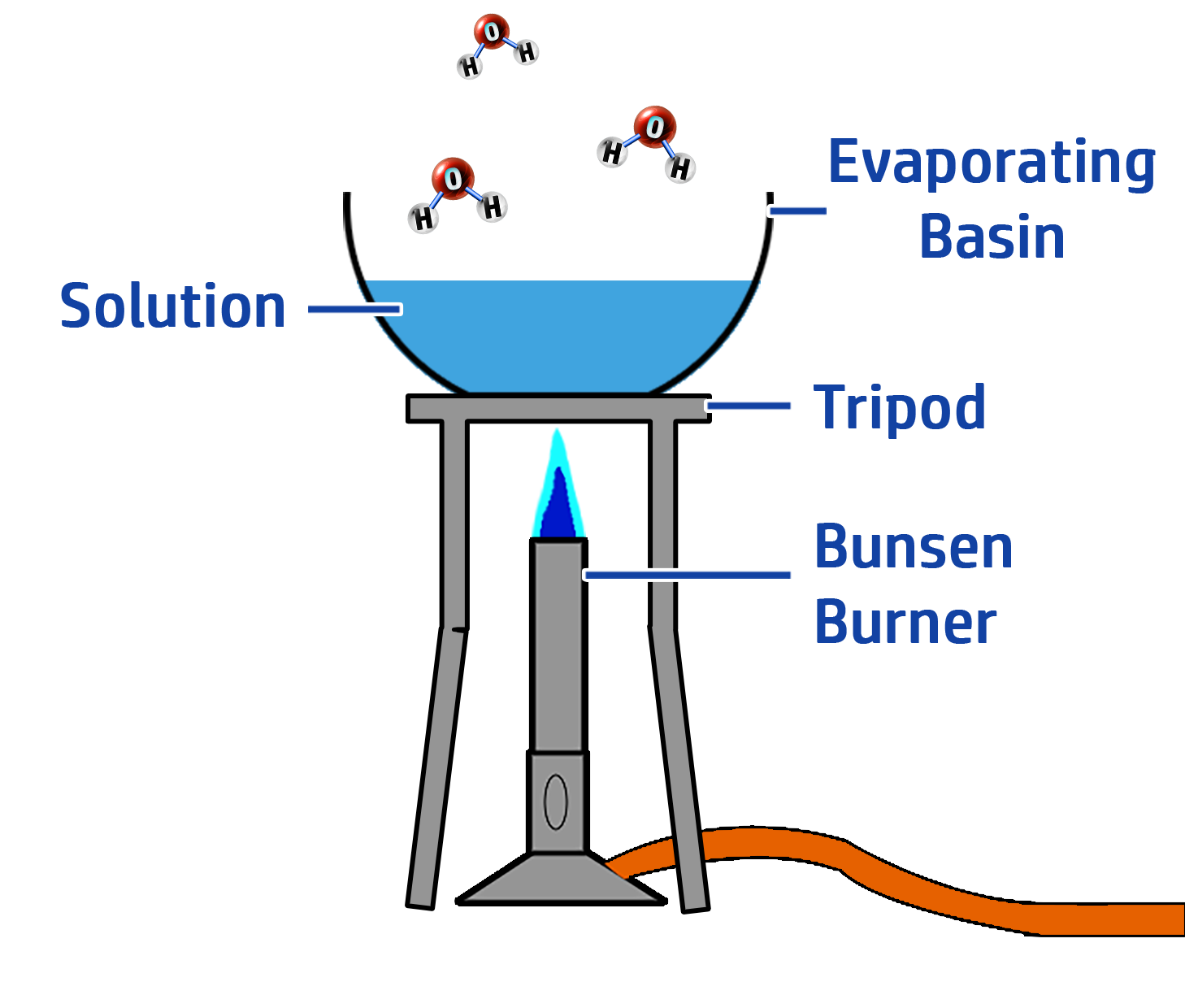
Phase Two: Preparing the Crystals
- Heat the solution and evaporate half of the water.
- Leave the solution to cool so that the rest of the water evaporates and crystals form.
- Dry the crystals between pieces of filter paper.

CH127: Core Practical: Investigating pH
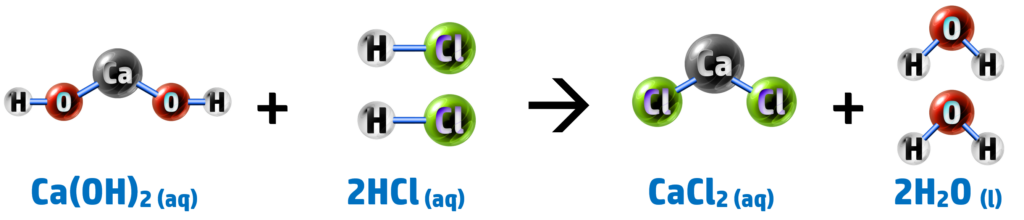
Calcium Hydroxide + Hydrochloric Acid → Calcium Chloride + Water
This is one of the big practicals that you need to know for the exam.
You could be asked how to carry it out; to analyse the results; to explain the results or to evaluate the risks. To investigate the pH of a substance, you will need to be able to do the following:
Key Steps: pH → 0.3g → Mix → pH → Neutral
Method
- Measure the pH of 50cm3 of hydrochloric acid using a pH probe (or pH paper).
- Measure out 0.3g of calcium hydroxide and add it to the beaker.
- Stir the beaker to mix the chemicals and make sure they are fully reacted.
- Check the pH of the substance again and record it in a table.
- Repeat until the pH of the solution is above neutral.

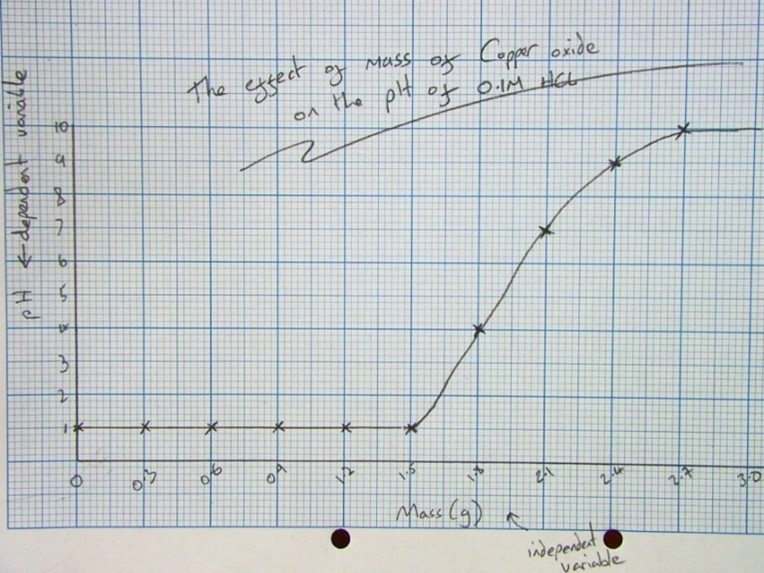
Analysing Results
- Once you have a graph of results drawn, you can draw a line across and then down from pH 7.
- From this graph, it took 2.1g of Calcium Hydroxide to completely neutralise the hydrochloric acid.
Explaining the results:
- The pH starts low as there are lots of H+ ions (it is acidic).
- It starts to rise as the hydroxide (an alkali) neutralises the acid.
- The pH continues to rise above 7 as calcium hydroxide is soluble, so the solution contains OH- ions making it alkaline.
Variables:
- Independent variable (the thing you change!): The mass of calcium carbonate, Ca(OH)2.
- Dependent variable (the thing you measure!): The pH of substance.
- Control variables (the things you keep the same!): The volume of acid, the concentration of acid, the type of tablet, the surface area of the tablet.

CH128: Describe how to make a salt from a soluble reactant
If you want to make a salt from a soluble reactant you cannot use filtration.
As you add the acid to your soluble alkali, it will reach pH 7 (neutral), but the pH will continue to rise because the alkali will start being added to your neutral solution.
To get round this, you need to use a titration (see CH129). You need to add the acid to the soluble alkali until it is exactly neutral (you can do this using an indicator) – at which point you can evaporate off the water, leaving your crystals behind.
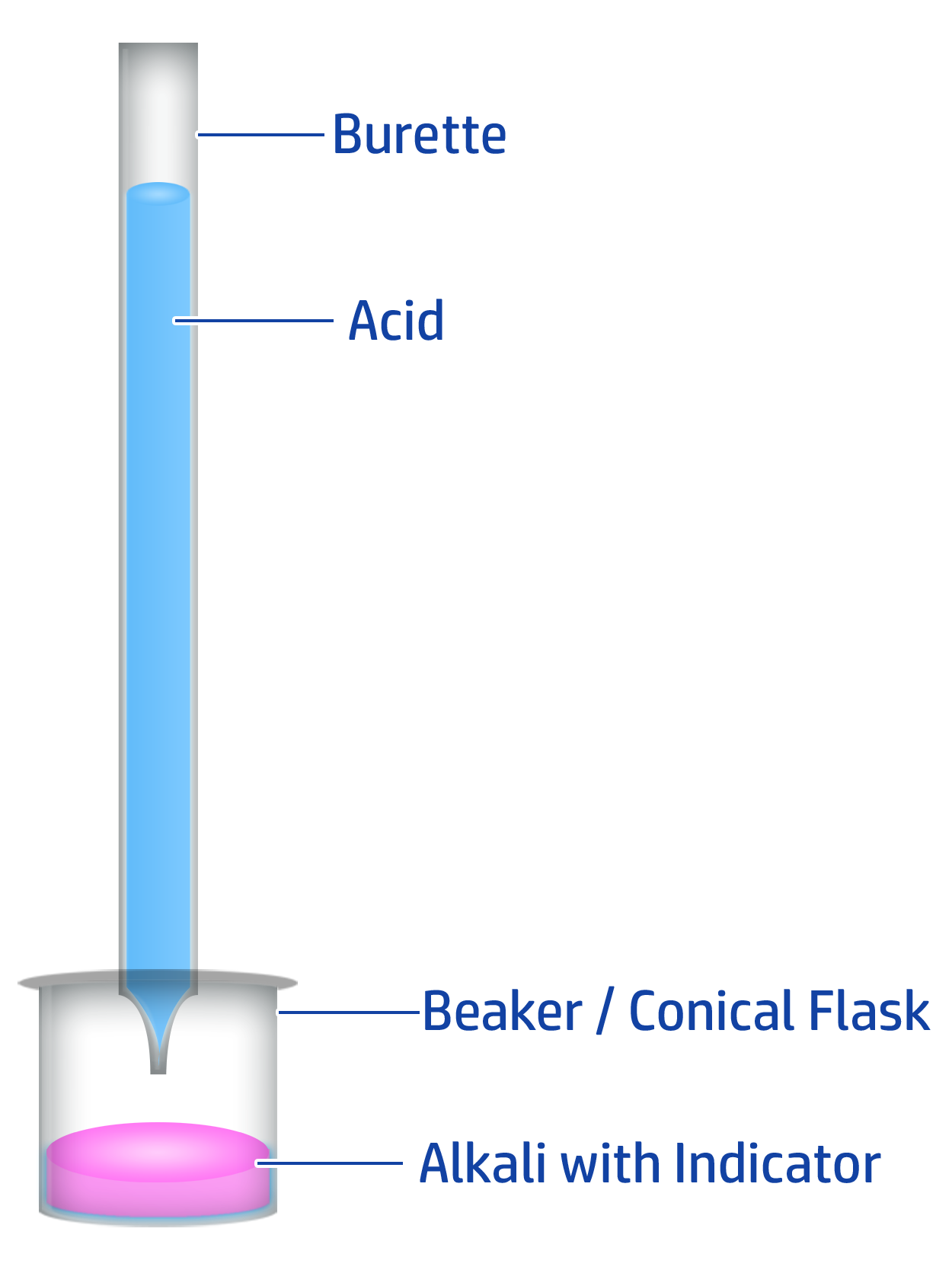
CH129: How to carry out a titration to produce a pure, dry salt
Key Steps: Burette → Pipette → Indicator → End Point
- Fill a burette to the 0.0cm3 line with hydrochloric acid.
- Add 20cm3 of your alkali (sodium hydroxide) into a conical flask using a pipette.
- Add a few drops of phenolphthalein indicator – the alkali will turn pink.
- When close to the end point, add the acid drop by drop until the indicator changes from pink to colourless.
If you are asked to produce a pure dry salt from this titration, do the following extra steps:
- Repeat the experiment without the indicator, this gives you just sodium chloride and water (pure).
- Heat the solution gently to evaporate half of the water.
- Leave the solution to cool forming crystals of sodium chloride
- Leave on a windowsill to dry.
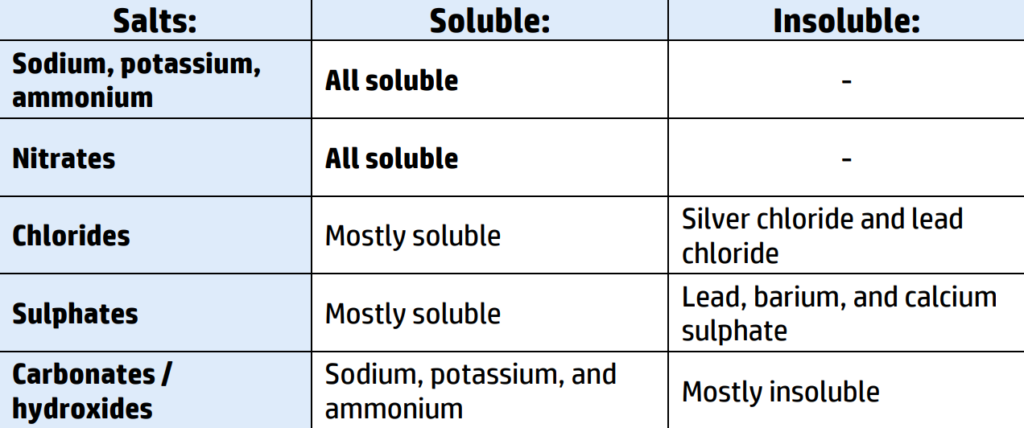
CH130: Recall the solubility rules
CH131: Predicting and naming precipitates
If you are given a word equation and asked to identify the name of the precipitate (insoluble solid) you need to look at the solubility rules (CH130)
Example: State the name of the precipitate formed in the following reaction:
Silver nitrate + copper chloride → silver chloride + copper nitrate
Start by looking at the products.
You can see from the solubility table in CH130 that most chlorides are soluble except for silver chloride and lead chloride.
Therefore, silver chloride is insoluble = your precipitate!
You then check the other product – copper nitrate – you can see that all nitrates are soluble, so copper nitrate will remain a liquid.
CH132: Describe how to produce a pure, dry precipitate
Key Steps: Dissolve → Mix → Filter → Wash → Dry
If you wanted to prepare a pure dry precipitate of lead chloride from the above reaction, there are 4 steps you need to follow:
- Dissolve the solid copper chloride and lead nitrate.
- Mix them together to produce your copper nitrate and lead chloride.
- Filter the solution to give you lead chloride in the filter paper.
- Wash the filter paper to remove the soluble copper nitrate.
- Dry the filter paper in a desiccator / oven to leave you with your pure, dry precipitate.

CH133: Describe what an electrolyte is

An electrolyte is any liquid that contains ions (atoms that have gained or lost electrons to become charged particles).
The liquid can be molten (melted) or aqueous (dissolved).

CH134: Describe what electrolysis is
Electrolysis is the breaking down of an electrolyte (CH133) using electrical energy.
The electrical energy needed is direct current.
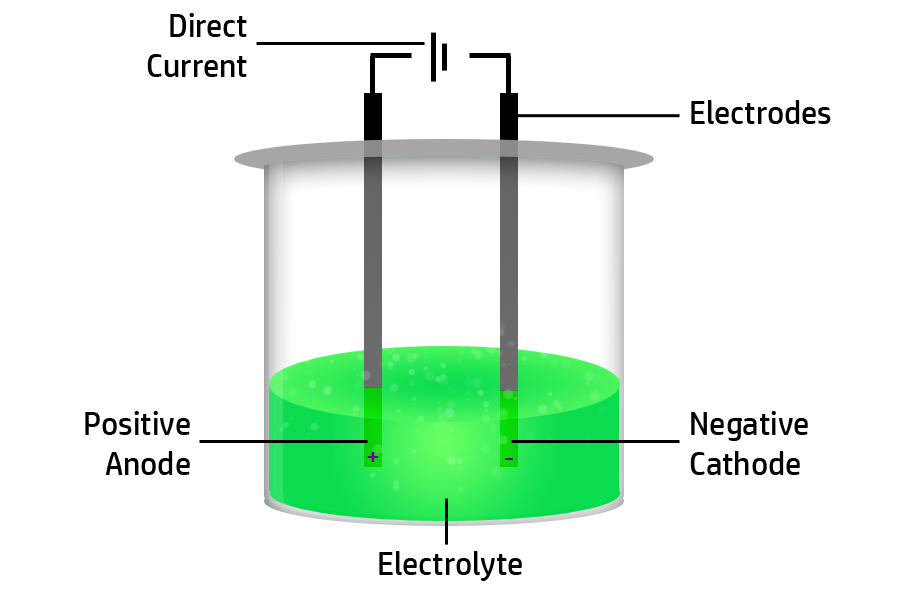
CH135: Describe the movement of ions during electrolysis
Firstly, remember that opposites attract – so positive ions will move to the negative electrode and negative ions will move to the positive electrode.
Secondly, remember the names of the electrodes by remembering the Mnemonic on the right: Positive Anode, Negative Is Cathode …so remember to PANIC in your exam!
Therefore, positive cations (paw-sitive!) will move to the negative cathode and negative anions will move to the positive anode

CH136: Describe how pure copper can be made using electrolysis
If you want to make copper pure, you can use electrolysis.
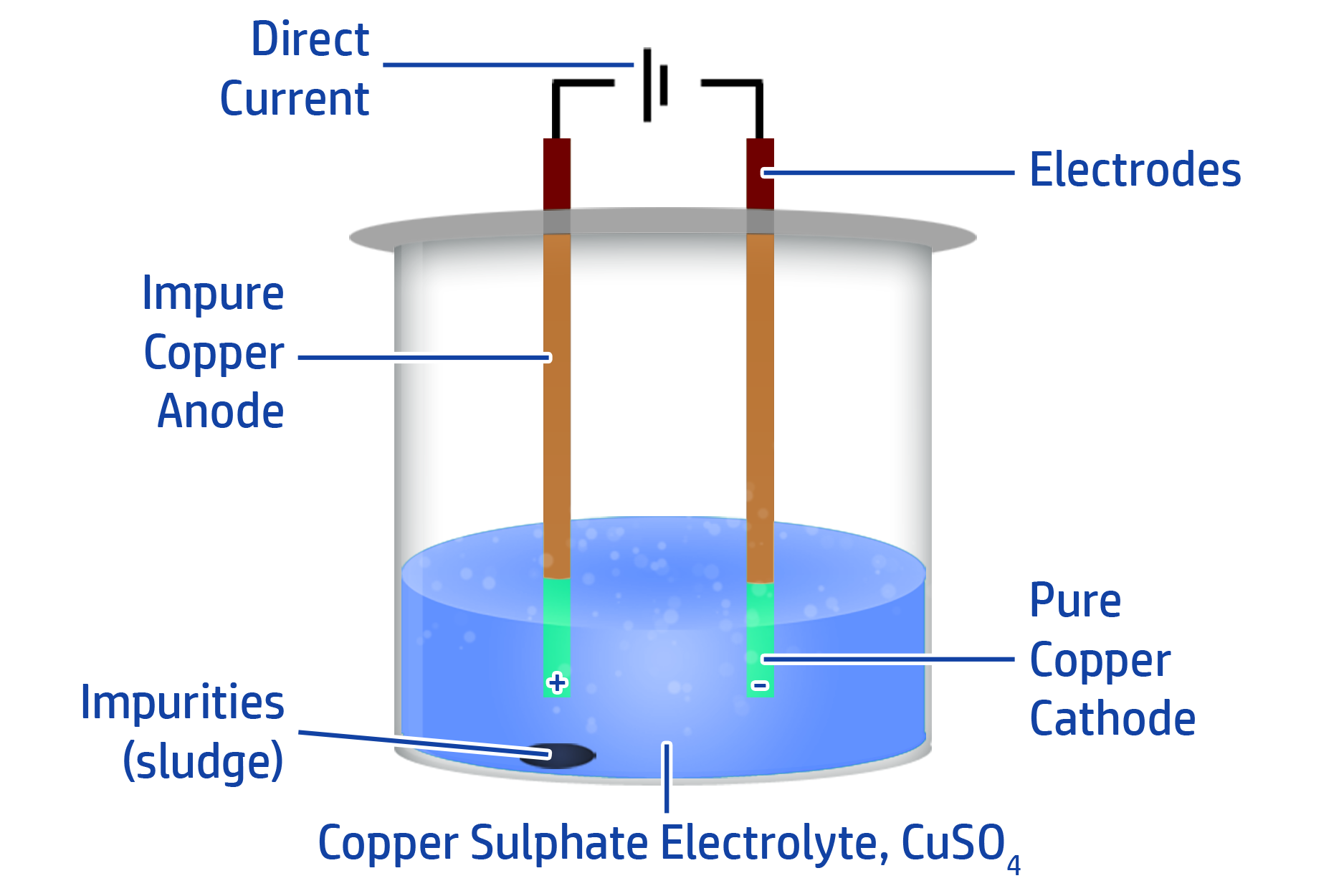
To do this, add your impure copper to the positive anode and a pure copper electrode onto the negative cathode.
You then need to have a solution that contains copper ions as your electrolyte (such as copper sulfate).
When you turn the D.C power supply on, the copper from the impure electrode will turn into copper cations (Cu2+). They will then move towards the negative cathode and turn back into copper atoms.
This removes the copper from the impure electrode and adds it to the pure electrode. You will be left with sludge in the solution – which is the ‘dirt’ from your impure electrode.
CH137: Core Practical: Investigating the electrolysis of copper sulfate

Electrolysis of CuSO4 with copper electrodes:
Why does the mass of the anode decrease?
- During the electrolysis, the copper atoms in the impure copper anode lose two electrons and turn into Cu2+ ions (oxidation).
- (H) Anode half equation (see CH139): Cu → Cu2+ + 2e–
Why does the mass of the anode decrease?
- The copper cations in the electrolyte move to the cathode and gain two electrons (reduction) to turn back into copper atoms.
- (H) Cathode half equation (see CH139): Cu2+ + 2e– → Cu
Why is the change in mass different?
- The anode mass will decrease more than the cathode mass will increase.
- This is because there are impurities on the anode that fall off during electrolysis.
- The impurities will fall to the bottom of the beaker as sludge.
Method:
Key Steps: Mass → Variable Resistor → Electrolyte → Wash → Dry → Mass
You will need to be able to explain how to investigate the change of current on the mass of the electrodes in the above set up.
- Measure the mass of the cathode and anode.
- Use a variable resistor to set the current to 0.2A.
- Add the electrodes to the electrolyte and leave for 20 minutes.
- Wash the electrodes to remove impurities
- Dry the electrodes by either gently patting them or dipping them in propanone. Do not scrub them or the copper will be wiped off.
- Measure the final mass of the electrodes and calculate the change in mass.
What happens if we use Inert Electrodes?
If you carry out electrolysis of copper sulphate using inert (unreactive) electrodes instead of copper electrodes, you see different things:
- The cathode will be coated in copper:
- Half equation (H) (see CH139): Cu2+ + 2e– → Cu
- Oxygen will form at the anode, which will be seen as bubbles:
- Half equation (H) (see CH139): 4OH– → O2 + 2H2O + 4e–
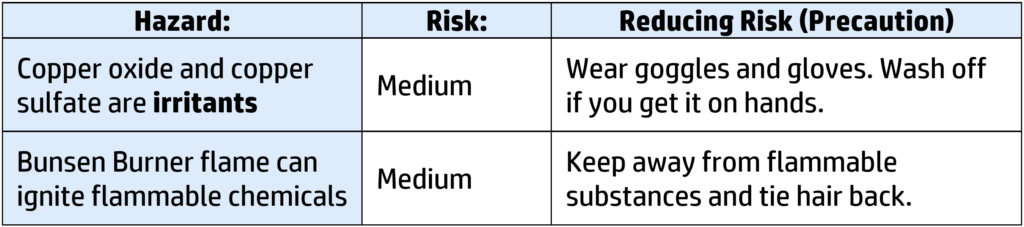
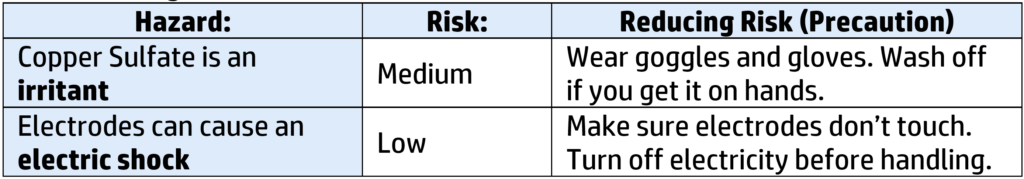
Risks and Management:
Variables:
- Independent variable (the thing you change!): The current
- Dependent variable (the thing you measure!): The change in mass of the electrodes.
- Control variables (the things you keep the same!): The volume of copper sulfate, the concentration of copper sulfate, how far the electrodes are dipped into the copper sulfate.
CH138: Predicting the products of electrolysis
Molten electrolytes: Example – Lead Bromide, PbBr2 (l)
- When you have a molten electrolyte, you only have two ions in the liquid – the metal and the non-metal.
- At the cathode, lead ions (Pb2+) will gain electrons (reduction) and turn back into lead atoms.
- At the anode, bromide ions will lose electrons (oxidation) and turn back into bromine molecules, Br2.
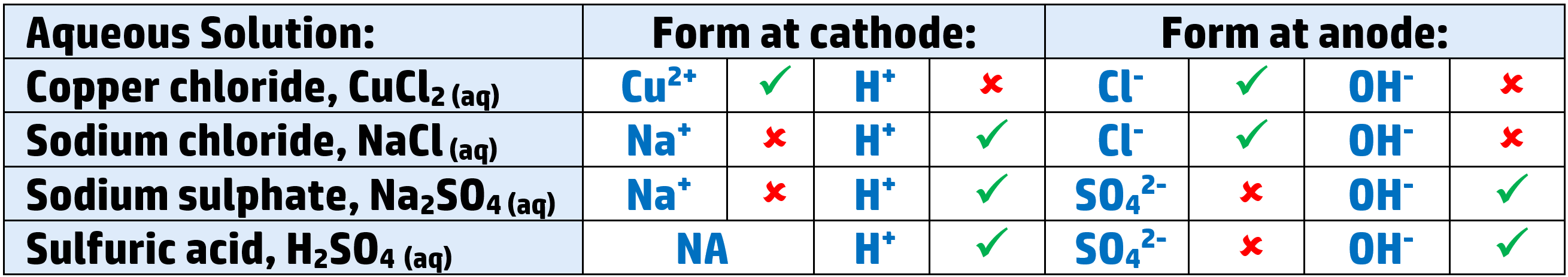
Aqueous electrolytes:
- When you electrolyse an aqueous solution, you have H+ and OH– ions as well as the metal and non-metal ions.
- For example: copper chloride, CuCl2, will have Cu2+ and H+ cations as well as Cl– and OH– anions.
- You need to be able to work out what will form. There are two rules to know:
- Cathode: The least reactive ion will form.
- Anode: If there is a halide (group 7), that will form – if not, the hydroxide will go there, and form water and bubbles of oxygen.
CH139: Writing Half Equations for electrolysis (H)
Once you know what forms at each electrode (CH138), you need to be able to write half equations. This involves looking at the change in electrons on each side.
Example: When molten zinc chloride is electrolysed, zinc ions, Zn2+, form zinc atoms. Write the half equation for this reaction.
Step 1: Write out the start of your half equation. The question tells you that the zinc ions are turning into zinc atoms, so it goes in this order: Zn2+ → Zn
Step 2: Work out what is happening to the electrons. Zn2+ has a full outer shell and is going back to its natural atom. To do this it needs to gain electrons back (it had lost 2 to become positively charged).
It will gain two electrons to get from Zn2+ to Zn – which is written like this:

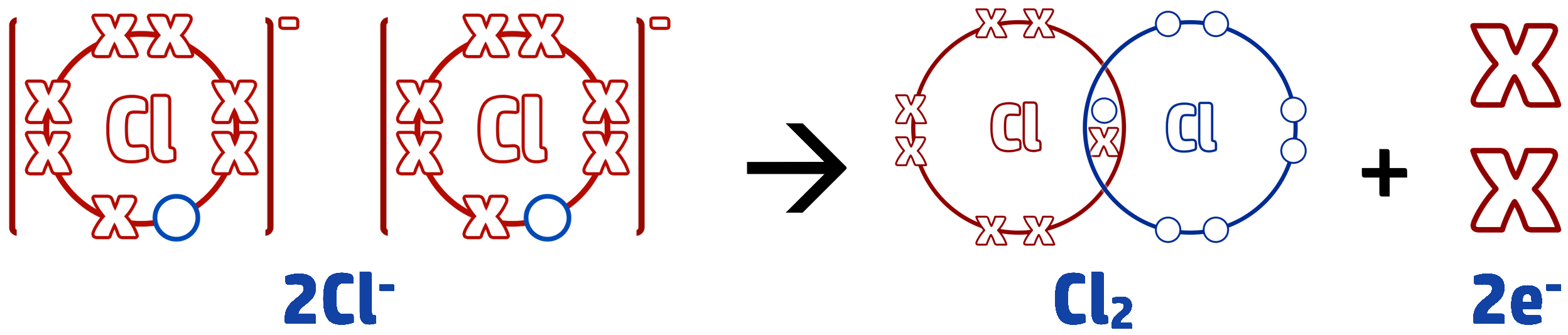
Example 2: When molten zinc chloride is electrolysed, chloride ions, Cl–, form chlorine molecules, Cl2. Write the half equation for this reaction.
Step 1: Write out the start of your half equation. The question tells you that the copper ions are turning into copper molecules, so it goes in this order: Cl– → Cl2
Step 2: Work out what is happening to the electrons. Cl– has a full outer shell and is going back to a molecule. To do this each chloride ion needs to lose an electron (they had gained 1 to become negatively charged). We write the loss of electrons on the right side of the arrow: Cl– → Cl2 + e
Step 3: We need to balance the half equation. To form Cl2, we need 2 chloride ions. Therefore, both of the chloride ions will need to lose an electron, so the balanced equation looks like this:
CH140: Electrolysis and REDOX reactions: Oxidation and Reduction (H)
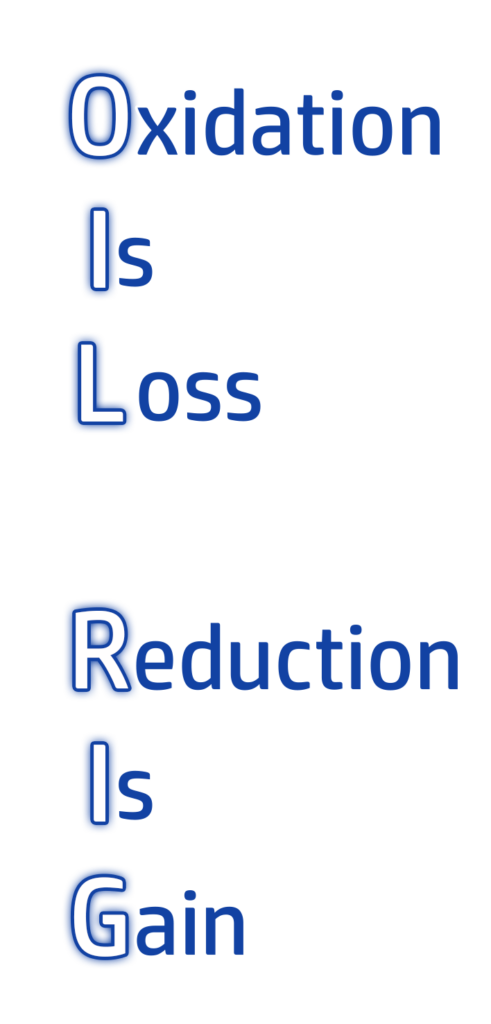
A REDOX reaction is any reaction that involves reduction and oxidation.
You may have come across oxidation when talking about the addition of oxygen, and reduction as the loss of oxygen, but there is another definition involving electrons:
- Oxidation is the loss of electrons.
- Reduction is the gain of electrons.
The order can be remembered by using the mnemonic OIL RIG (see left!)
CH141: Using REDOX to explain what happens at the electrodes (H)
When an ion goes back to its atom during electrolysis, it will either gain or lose electrons.
In the electrolysis of molten sodium chloride:
- The sodium (Na+) ions gain electrons to turn back into sodium metal. Reduction is occurring (see CH140).
- The chloride (Cl–) ions lose electrons and turn back into chlorine gas, Cl2. Oxidation is occurring (see CH140).


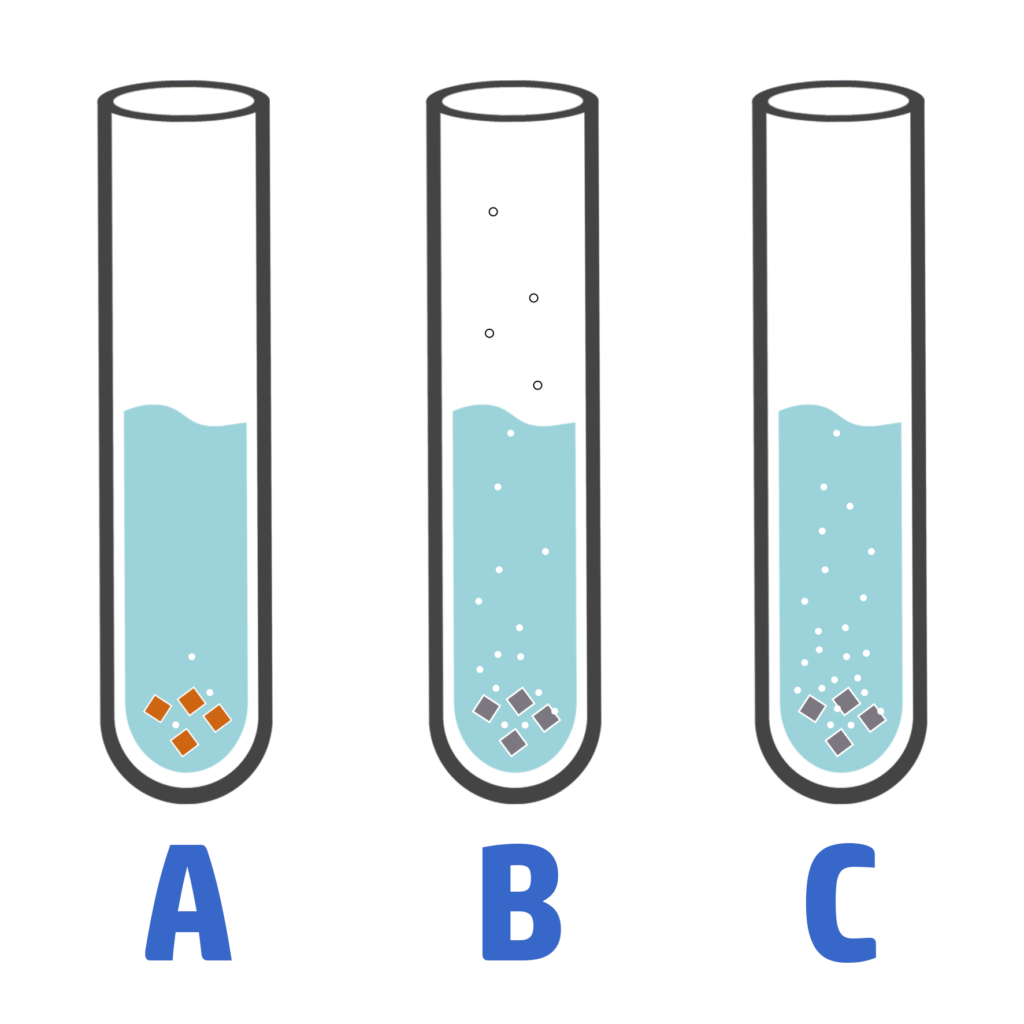
CH142: Linking observations and reactivity
When you react a metal with water and different acids, you can use the observations to work out how reactive they are:
- The more bubbles / fizzing / effervescence there is, the more reactive the metal.
- On the right, you can see that C is the most reactive metal because it produces the most bubbles.
- A is the least reactive as it has the least bubbles.
You can also look at temperature changes. The bigger the temperature change, the more reactive the metal.
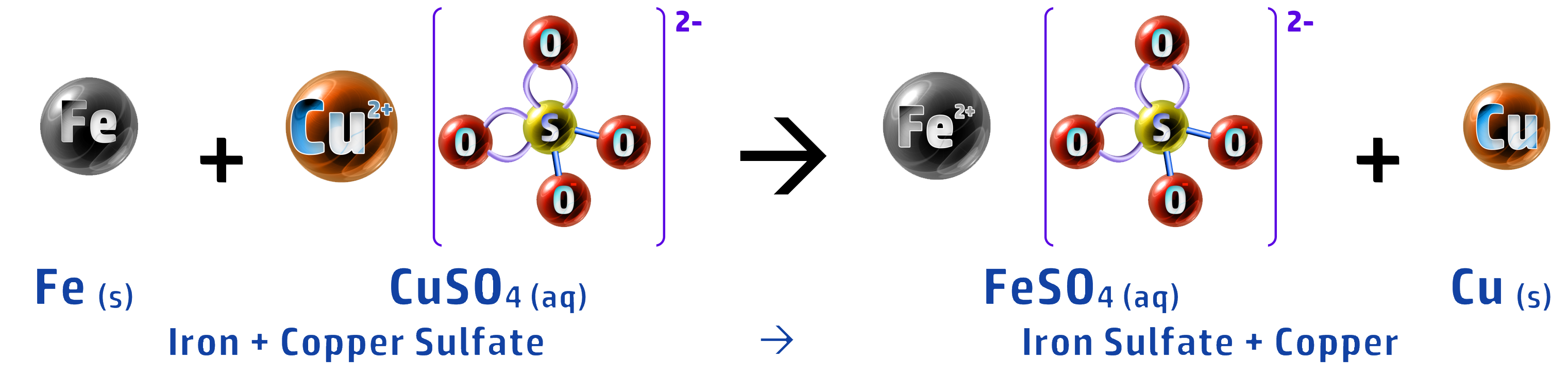
CH143: Displacement reactions and REDOX (H)
When you add a metal to a solution containing a metal, you can predict whether a displacement reaction will occur or not (where a more reactive metal swaps places with a less reactive metal in a compound).
The more reactive metal wants to be part of the compound (see CH142) – leaving the least reactive as the stand-alone metal.
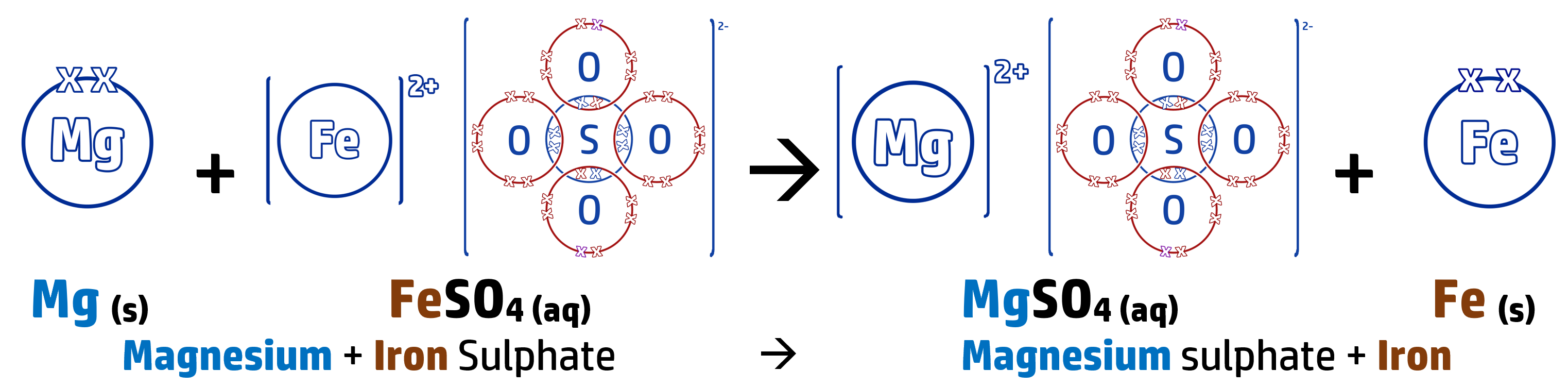
In the above reaction, magnesium is more reactive than iron so swaps places with it.
This is an example of a REDOX reaction – where oxidation and reduction are both occurring during the reaction:
Magnesium turns from an atom into an ion by losing two electrons – this means that the magnesium is oxidised (OIL – see CH140).
Mg → Mg2+ + 2e–
The iron ion, which is displaced, gains two electrons to turn back into an iron atom. The iron is therefore reduced (RIG – see CH140).
Fe2+ + 2e– → Fe
CH144: Investigate the reactivity of metals
You can work out the reactivity series for metals by seeing how they react with cold water, steam, and acids. The results below allow you to put the metals in order of reactivity:
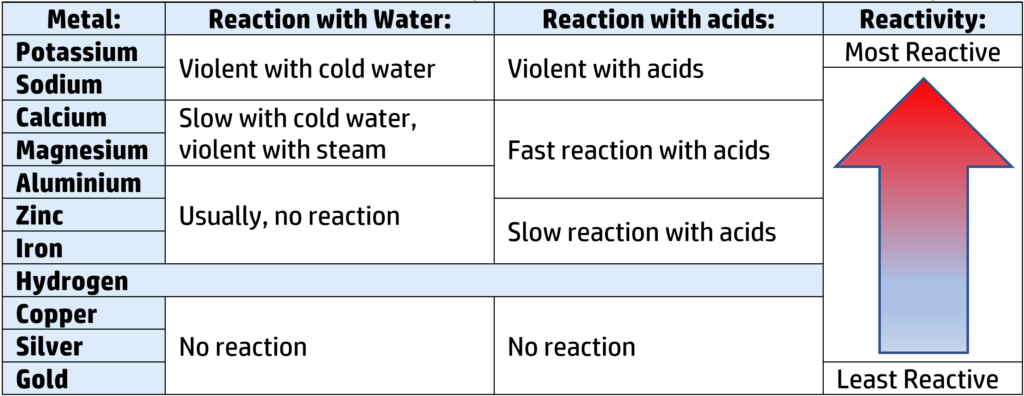
Reactions of metals with water:
If a metal reacts with water, if will always form a metal hydroxide and hydrogen:
Potassium + water → Potassium hydroxide + hydrogen
Reactions of metals with steam:
If a metal reacts with steam, if will usually form a metal oxide and hydrogen:
Calcium + water → Calcium oxide + hydrogen
Reactions of metals with acids:
If a metal reacts with an acid, if will usually form a salt and hydrogen (see CH116):
Calcium + nitric acid → Calcium nitrate + hydrogen
CH145: Linking electronic configuration to reactivity
For a metal to oxidise, it needs to lose its electrons / react with oxygen.
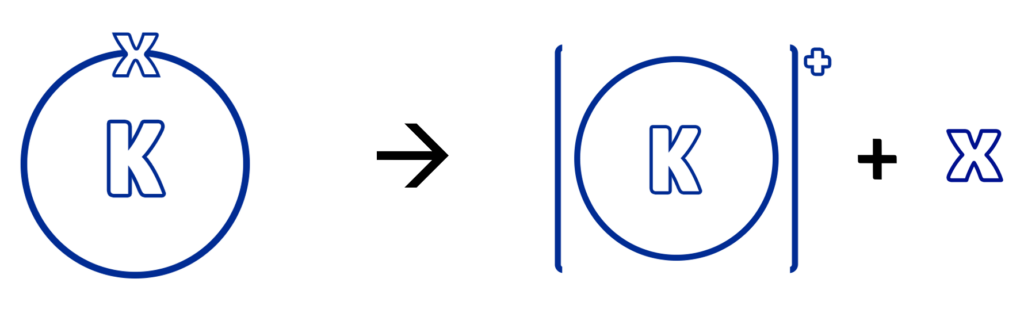
The more reactive a metal is, the quicker it is to lose its electrons/react with oxygen.
Therefore:
- If a metal is highly reactive, such as potassium, it will oxidise/lose its electrons very quickly.
- If a metal is less reactive, such as iron, it will oxidise slowly.
- If a metal is unreactive, such as gold, it will not oxidise at all.
CH146: Describe what an ore is

An ore is a rock that contains enough metal to make it financially worthwhile to extract. If it isn’t going to give a profit to extract it – it is not an ore!
Most metals are found in ores, such as bauxite – aluminium oxide, Al2O3, and need to be extracted by either heating with carbon or electrolysis (see CH149)
Some metals, such as gold and silver, are found uncombined – these are the metals that don’t react – so are not found in ores.
CH147: Describe Oxidation and Reduction in terms of oxygen
There are two definitions of oxidation and reduction – one involving electrons (see CH140) and one involving oxygen, which states that:
- Oxidation is the addition of oxygen.
- Reduction is the removal of oxygen.
For example: The reduction of iron oxide, FeO, using carbon:
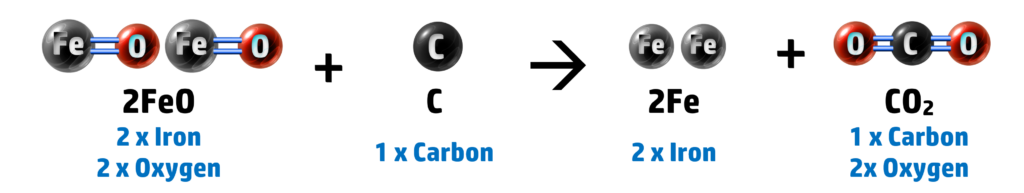
If we look at the two atoms that are not oxygen, we have iron and oxygen:
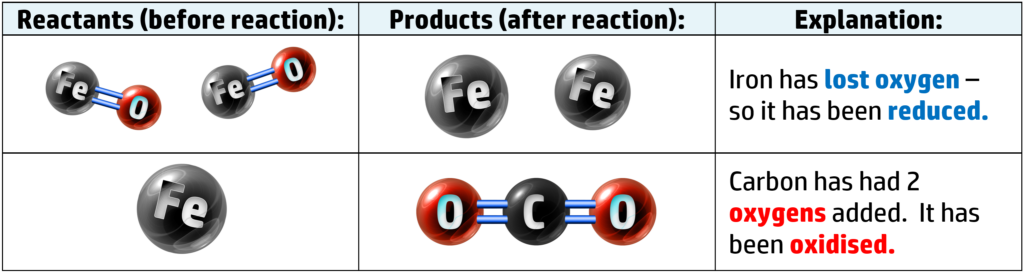
When metals are oxidised, they corrode. When iron rusts, it isn’t just reacting with oxygen, but water as well.
CH148: Link metal extraction to oxidation and reduction
Regardless of which type of metal extraction you look at (either electrolysis or heating with carbon – see CH149), reduction is always occurring.
Reduction is the removal of oxygen and the gain of electrons.
1. Electrolysis: 2Al2O3 → 4Al + 3O2
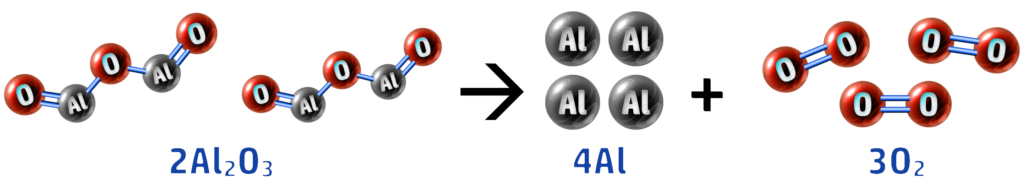
Here you can see that our aluminium oxide has lost oxygen (forming aluminium and oxygen) – therefore aluminium oxide is reduced.
2. Heating with Carbon: 2FeO + C → 2Fe + CO2

Here you can see that our iron oxide has lost oxygen (forming iron and carbon dioxide) – therefore iron oxide is reduced. Carbon has also been oxidised to form carbon dioxide (see CH147).
CH149: Explain how to extract metals based on cost and reactivity

There are three ways of removing metals from the ground:
Electrolysis is used to extract highly reactive elements from their ore. These elements are more reactive than carbon.
Heating with carbon is used to extract slightly reactive metals from their ore. These elements are less reactive than carbon.
Unreactive metals, such as gold and silver, can just be removed by digging.
CH150: Evaluate Bioleaching and Phytoextraction (H)
There are two other methods of extracting metals which are used to extract rocks with small amounts of metals in – called low grade ores: Bioleaching and Phytoextraction.
Bioleaching:
- Bioleaching uses bacteria to break down low-grade ores into a solution called a leachate.
- This solution is acidic and contains the metal ions you want.
- An example of this is collecting copper, from a copper sulfate leachate, using scrap iron.
- Iron is more reactive than copper so displaces it:
| Advantages: | Disadvantages: |
|---|---|
| Does not require higher temperatures. No harmful gases Less damage to landscape than mining Conserves supplies of higher-grade ores | Very slow. Toxic substances and sulphuric acid can be produced – damaging the environment |
Phytoextraction
- Plants absorb mineral ions from the soil.
- The metal ions become concentrated in the plant cells.
- The plants are burnt, leaving behind the metal in the ash.
| Advantages: | Disadvantages: |
|---|---|
| Can extract metals from contaminated soil. No harmful gases Less damage to landscape than mining Conserves supplies of higher-grade ores | Very slow. Can only grow in certain climates |
