QUANTITATIVE CHEMISTRY

An AQA GCSE chemistry revision page showing INSERT CONTENT HERE.
CH78: Calculate Relative Formula Mass
The relative formula mass is simply the atomic masses of all the atoms in a compound added together. You can find the atomic mass in the question – or the big number on the Periodic Table.
Example 1: Chloromethane has the chemical formula of CH3Cl. Calculate the relative formula mass. (Ar: C = 12, H = 1, Cl = 35.5)
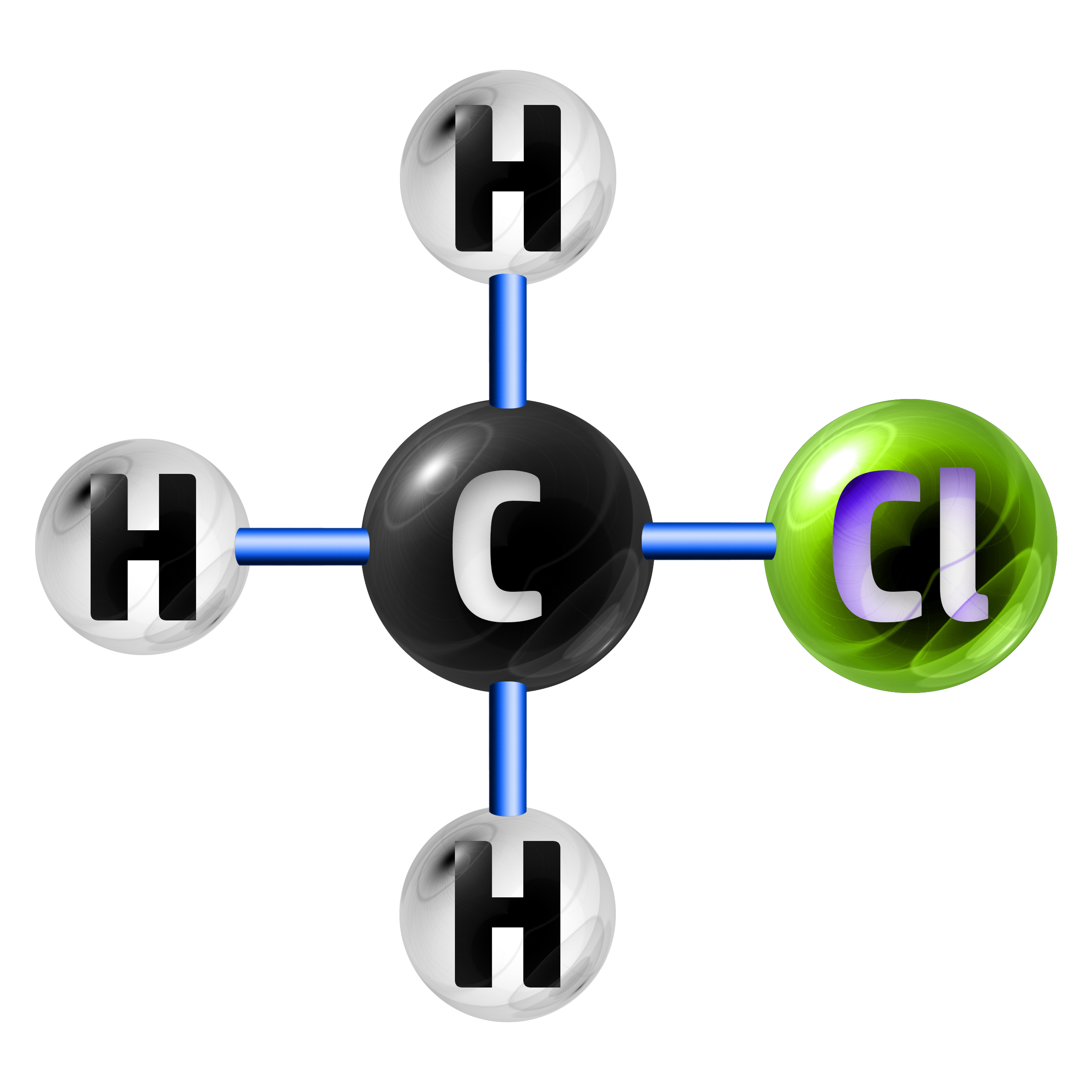
The formula CH3Cl shows me that I have 1 x C (carbon), 3 x H (hydrogen) and 1 x Cl (chlorine).
- 1 x C = 1 x 12 = 12
- 3 x H = 3 x 1 = 3
- 1 x Cl = 1 x 35.5 = 35.5
Relative Formula Mass = 12 + 3 + 35.5 = 50.5
Example 2: Magnesium nitrate has the chemical formula of Mg(NO3)2. Calculate the relative formula mass. (Ar: Mg = 24, N = 14, O = 16)
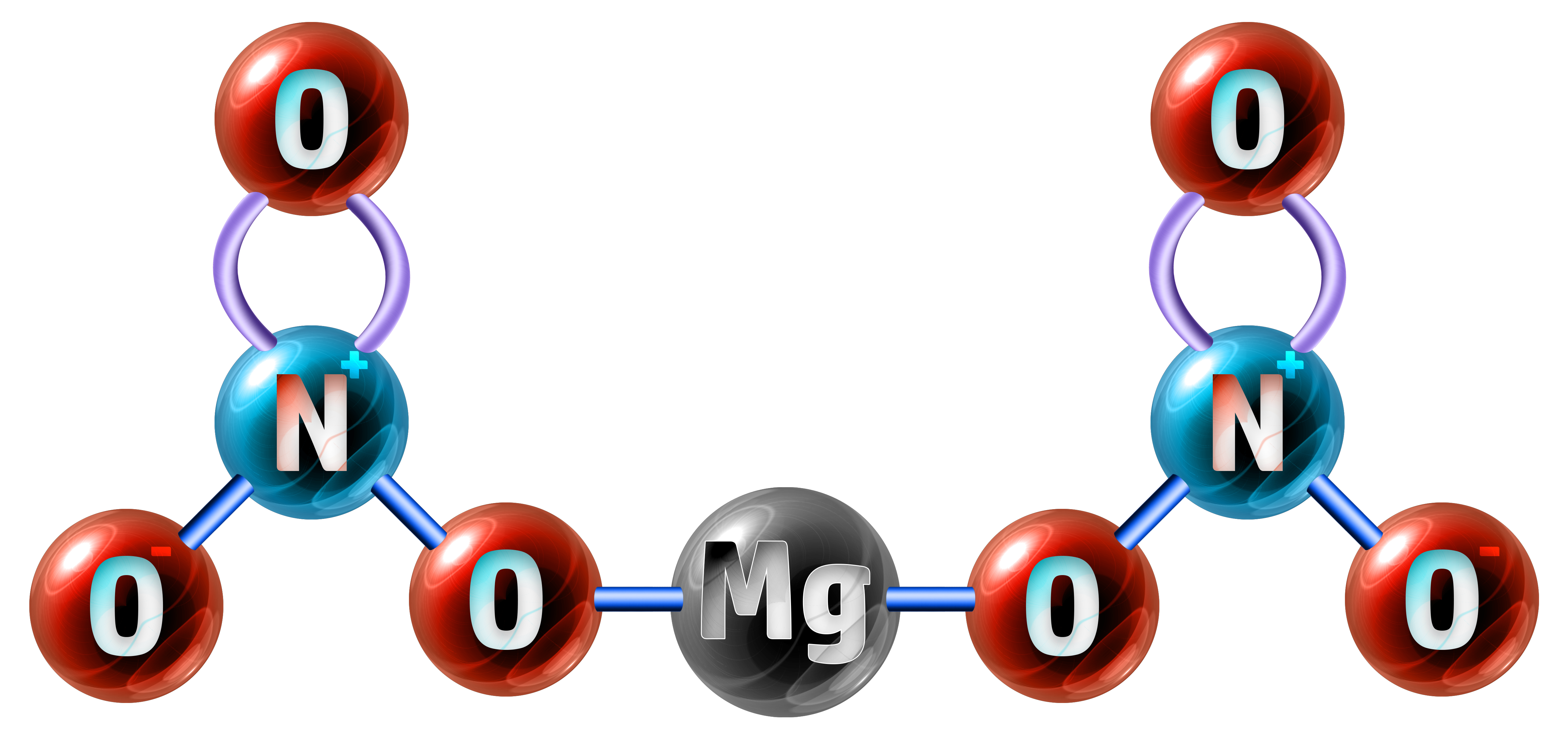
The formula Mg(NO3)2 shows me that I have 1 x Magnesium atom (Mg) connected to two lots of my nitrate ion (NO3), so I have 1×2 nitrogen’s and 3×2 oxygen’s:
- 1 x Mg = 1 x 24 = 24
- 2 x N = 2 x 14 = 28
- 6 x O = 6 x 16 = 96
Relative Formula Mass = 24 + 28 + 96 = 148
CH82: Explain the conservation of mass
The conservation of mass states that the total mass of reactants is always the same as the total mass of products.
So, if you added 5g of sodium to 100g of water you would end up with 105g of sodium hydroxide and hydrogen gas combined.
The mass may appear to go down in the above reaction – and if it does it is because a gas (such as hydrogen) has escaped.
To prove that the mass stays the same use a sealed container. I.e., Add a lid!
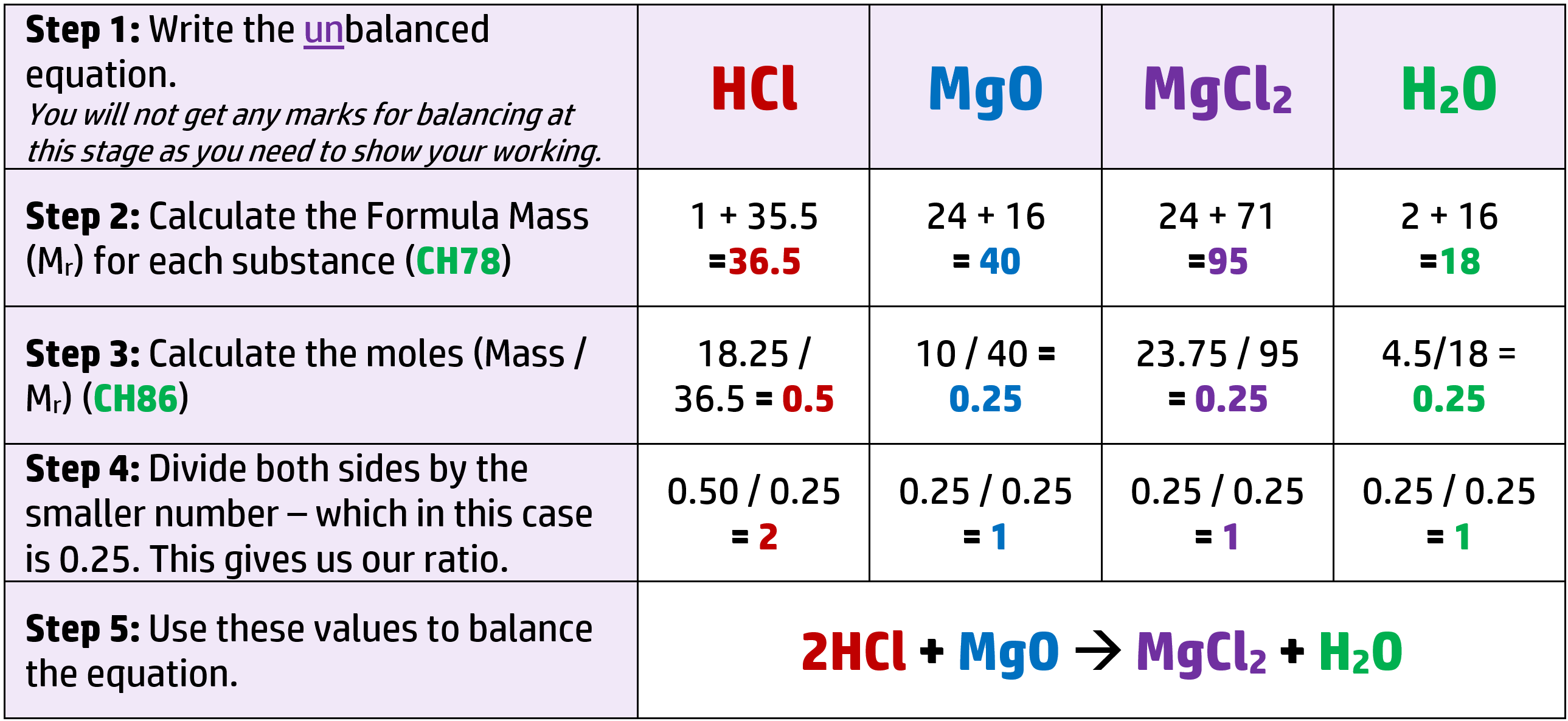
CH84: Calculate concentration in gdm-3
To calculate the concentration of a solution in grams per decimetre cubed (gdm-3), you need two things: the mass in grams, and the volume in dm3 (cm3 / 1000)
Example: 20g of sodium hydroxide dissolves in 750cm3 of water. Calculate the concentration in gdm-3
- Volume = 750cm3 / 1000 = 0.75dm3
- Concentration = mass / volume = 20g / 0.75dm3 = = 26.7gdm-3
CH85: Moles and Avogadro’s Constant (H)
Every time you see a balanced equation you are seeing a ratio – how much of each substance is needed:
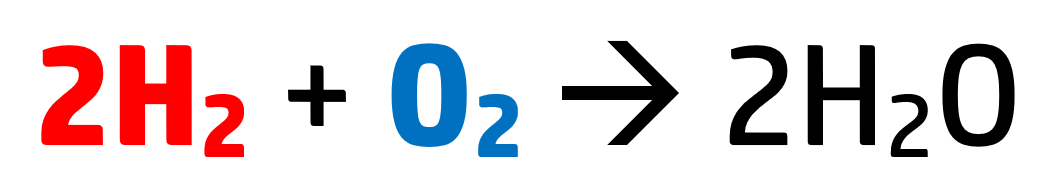
This reaction shows that for every molecule of oxygen, you need two molecules of hydrogen.
These numbers represent moles, and a mole represents 6.02×1023 {atoms / molecules / particles} of that substance.
For example, in the above reaction, we have 2 moles of hydrogen – meaning we have 2 x 6.02×1023 molecules of hydrogen (which is 1.204×1024 particles of hydrogen).
Oxygen just has 1 mole, so has 6.02×1023 particles. This number – 6.02×1023 – is known as Avogadro’s constant.
CH86: Calculate moles, mass and formula mass (H)
Moles (along with Avogadro’s constant – CH85) represent how many particles of a substance you have.
This can be calculated by using the triangle on the right.
For example: How many moles of sodium chloride, NaCl, are there in 5g of the compound? (Ar: Na = 23, Cl = 35.5)
- Step 1: Work out the formula Mass (CH78) = 23 + 35.5 = 58.5
- Step 2: Divide the mass (g) by the formula mass = 5g / 58.5 = 0.085 mol
You can also work out the exact number of particles of each substance.
In the above example, we have worked out that we have 0.085 moles of sodium chloride.
To work out the particles, just multiply this number by Avogadro’s constant – which they will always give you in the question (6.02×1023) = 0.085 x 6.02×1023 = 5.12×1022 particles of sodium chloride.
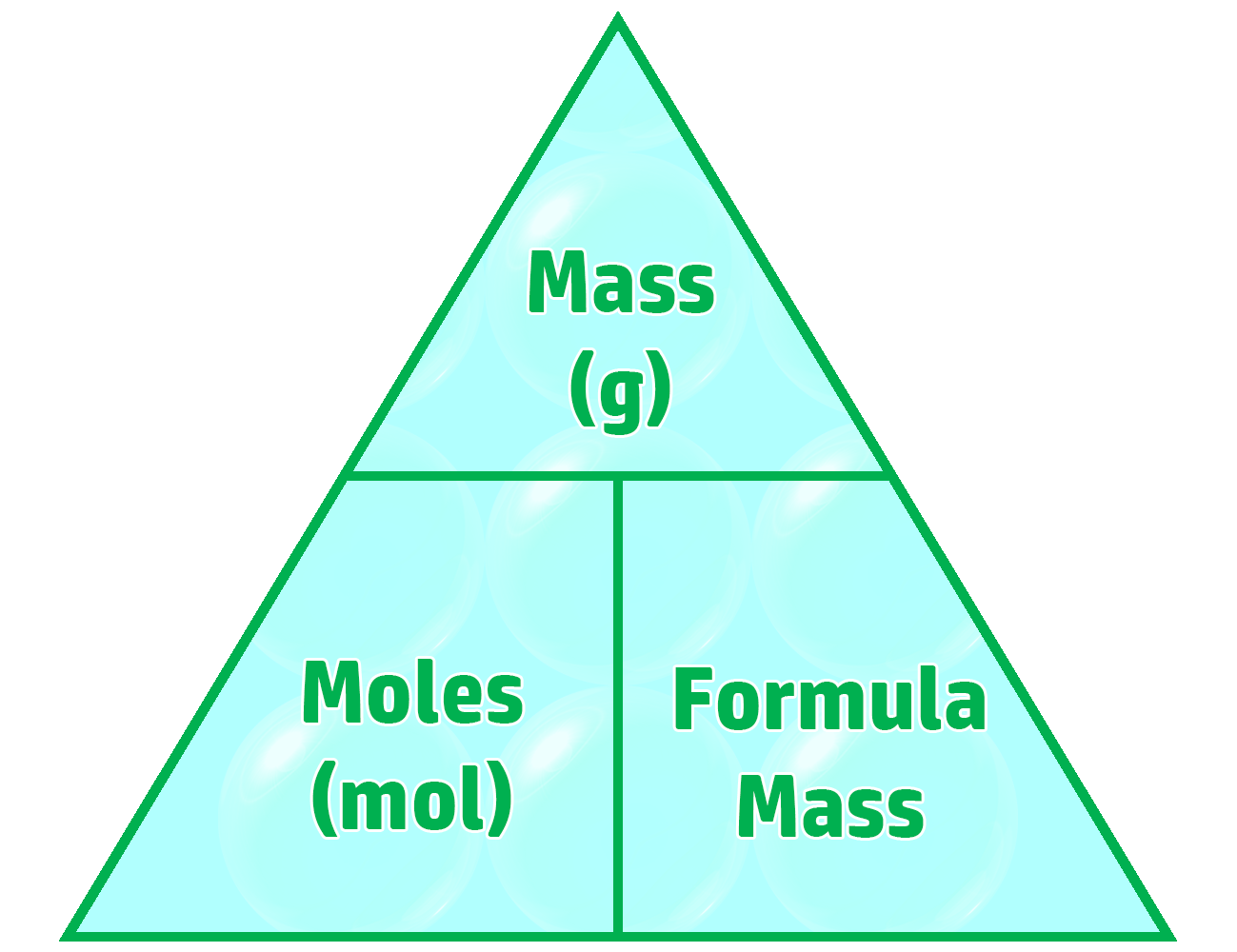
CH87: Calculate the limiting reactant for a reaction (H)
The limiting reactant is the reactant that you will run out of first. Once this runs out, the reaction will stop.
Example: 1.50g of ammonium chloride reacts with 4.0g of calcium hydroxide to form ammonia.

Work out the limiting reactant. (Ar: N = 14, H = 1, Cl = 35.5, Ca = 40, O = 16)
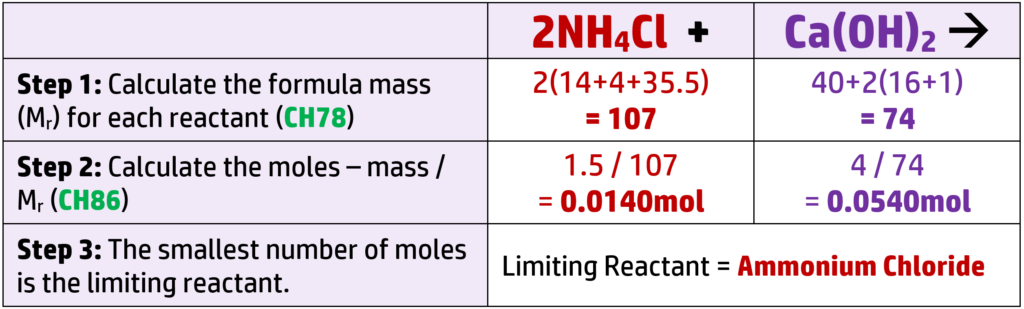
CH88: Balance chemical equations using stoichiometry (H)
You won’t normally see the term “stoichiometry” in the exam, but will see a question similar to the below:
Example 1: 18.25g of hydrochloric acid, HCl, reacts with 10g of Magnesium oxide, MgO, to form 23.75g of magnesium chloride, MgCl2 and 4.5g of water. Deduce the balanced equation for the reaction.
You must show your working. (Ar: H = 1, Cl = 35.5, Mg = 24, O = 16)
CH165: Calculating and converting between gdm-3 and moldm-3
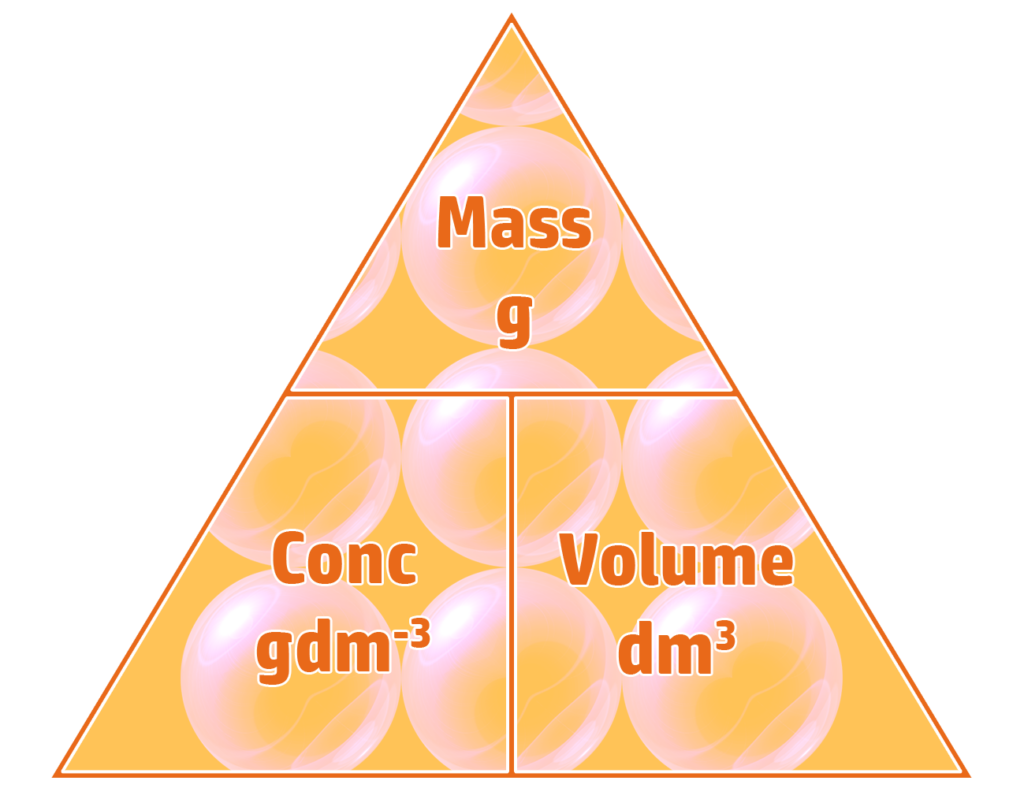
Part 1: Calculating Concentration in gdm-3
To calculate the concentration of a solution in gdm-3, you simply divide the mass (g) by the volume (dm3).
Example: 20g of NaOH is dissolved in 750cm3 of water. Calculate the concentration in gdm-3.
- Volume = 750cm3 ÷ 1000 = 0.75dm3
- Concentration = mass ÷ volume
- Concentration = 20g ÷ 0.75dm3 = 26.7gdm-3
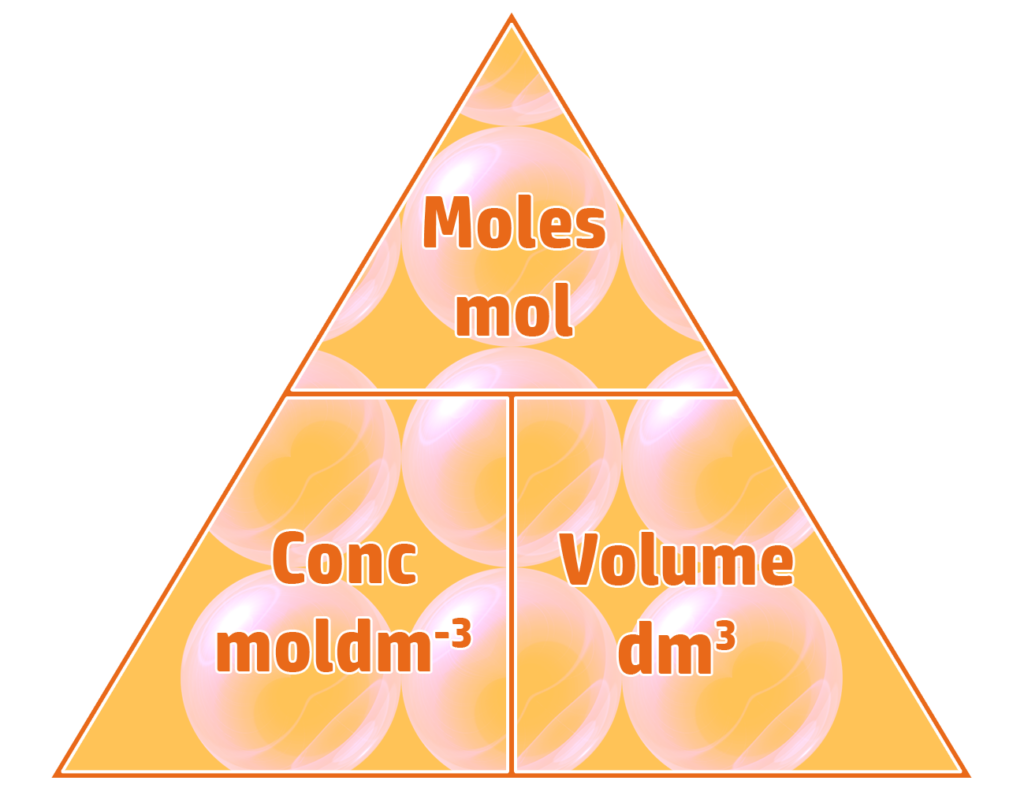
Part 2: Calculating Concentration in moldm-3
To calculate the concentration of a solution in moldm-3 you divide the moles (see CH86) by the volume (dm3).
Example: 0.5 moles of NaOH are dissolved into 250cm3 of water. Calculate the concentration of the sodium hydroxide solution formed in moldm-3
- Volume = 250cm3 ÷ 1000 = 0.25dm3
- Concentration = moles ÷ volume
- Concentration = 0.5mol ÷ 0.25dm3 = 2moldm-3

Part 3: Converting between gdm-3 and moldm-3
If you know either the concentration in gdm-3 or moldm-3, as well as the formula of the substance, you can convert between gdm-3 and moldm-3 and back using the triangle on the left.
Example: A 26.7gdm-3 solution of sodium hydroxide, NaOH, is produced by dissolving 20g of sodium hydroxide in 0.75dm3 of water. Calculate the concentration in moldm-3. (Ar: Na = 23, O = 16, H = 1)
Step 1: Calculate the Relative Formula Mass, Mr (CH78)
- You have one Na, so 1×23=23
- You have one O, so 1×16=16
- You have one H, so 1×1=1.
- Therefore the Mr is 23+16+1 = 40
Step 2: moldm-3 = gdm-3 ÷ formula mass.
- Concentration = 26.7gdm-3 / 40 = 0.67mol/dm-3
CH168: Calculating Theoretical Yields and Percentage Yields
Key word definitions:
- Actual Yield: The mass of a desired product actually produced in a chemical reaction.
- Theoretical Yield: The maximum calculated mass that could be produced.
- Percentage Yield: A measure of how much product you actually make in a chemical reaction compared to how much you should make in theory, expressed as a percentage.

Example: In a chemical reaction, 5.3g of sodium carbonate is reacted with excess hydrochloric acid. It was calculated that 5.85g of sodium chloride should be produced, but only 4.2g of sodium chloride was actually formed. Calculate the percentage yield for the reaction.
- Percentage yield = Actual Yield ÷ Theoretical Yield x 100.
- Percentage yield = 4.2g ÷ 5.85g x 100 = 71.8%
You might also be asked to calculate the theoretical yield – this is the same as a maximum mass calculation (see CH83)
CH169: Why is % Yield less than 100%
It is very rare that a reaction produces 100% yield. There are three main reasons for this:
- Some of the products is lost when transferred between containers.
- Side reactions may have occurred.
- The reaction may not have finished. (One of the reactants may have run out.)

If the yield is ever above 100%, it is normally because there is still water left in the product.
CH170: Calculate Atom Economy
Atom Economy is the percentage of mass converted into useful products and is calculated using the equation on the right.

Example: Ammonium chloride reacts with calcium hydroxide to form ammonia, calcium chloride and water. Work out the atom economy for the formation of calcium chloride in this reaction. (Ar: N = 14, H = 1, Cl = 35.5, Ca = 40, O = 16)
2NH4Cl + Ca(OH)2 → 2NH3 + CaCl2 + 2H2O
Step 1: Find out the formula mass (CH78) for calcium chloride, CaCl2.
- 1 x Ca = 1 x 40 = 40
- 2 x Cl = 2 x 35.5 = 71
- Relative Formula Mass = 40 + 71 = 111.
Step 2: Find out the total formula mass of all of the products combined:
- 2NH3: 2 x (14 + 3) = 34
- 2H2O: 2 x (2 + 16) = 36
- Total formula mass of products = 34 + 36 + 111 = 181
Step 3: Use these two values to calculate the percentage of useful products:
- Atom Economy = formula mass of useful product (calcium chloride) ÷ total formula mass of all products x 100
- Atom economy = 111 ÷ 181 x 100 = 61.3%
CH172: Avogadro’s Law and Gases
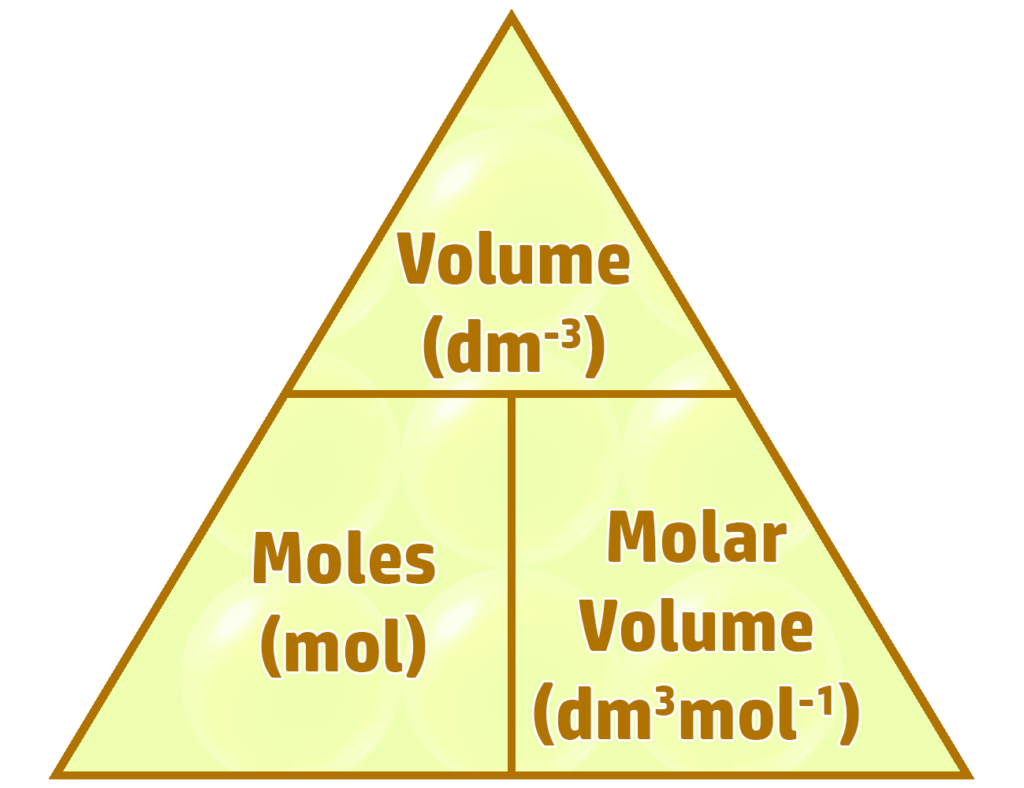
The molar volume is the volume of a gas when you have one mole of molecules.
At room temperature (20oC) and pressure (1atm), the
molar volume for 1 mole is always 24dm3mol-1.
You can use this to work out masses and volumes for any balanced equation:
Example 1: Calculate the volume of 0.50 mol of oxygen at room temperature & pressure. (Molar volume = 24dm3mol-1)
- Volume = moles x molar volume
- = 0.5 x 24 = 12dm3
Example 2: In an experiment, hydrogen gas is made by reacting 20.0g of sodium with excess water. Calculate the maximum volume of hydrogen that could be formed.
2Na (s) + 2H2O(l) → 2NaOH(s) + H2 (g)
- Step 1: Find out the formula mass (CH78) for one sodium = 23
- Step 2: Find out the moles (CH86) for sodium = mass ÷ Mr = 20 / 23 = 0.870 moles
- Step 3: The ratio of sodium to hydrogen is 2:1, so you have 0.87/2 = 0.435 moles of hydrogen.
- Step 4: Volume = moles of H2 x molar volume = 0.435 moles x 24 = 10.4dm3
CH173: Molar Volume Calculations
Avogadro’s law states that if the temperature and pressure are the same,the volume will be the same if you have the same number of molecules. You can use this to work out the volumes from balanced equations:
Example: 200dm3 of hydrogen reacts with oxygen to form water vapour. What volume of oxygen would completely react with the hydrogen?
2H2 (g) + O2 (g) → 2H2O(g)
The ratio of moles between hydrogen and oxygen is 2:1 – so the volume of oxygen is half of the volume of hydrogen:
- 200dm3 / 2 = 100dm3.
The volume of water produced would be 200dm3 because the ratio is now 2:2.
