ATOMIC STRUCTURE AND THE PERIODIC TABLE

AQA GCSE Chemistry revision page on atomic structure and the periodic table, explaining subatomic particles, electron arrangements, isotopes, trends in the periodic table, group patterns, and how these ideas link to reactivity and properties across elements with worked examples.
4.1.1.1 Atoms, elements and compounds
4.1.1.2 Mixtures
4.1.1.3 The development of the model of the atom
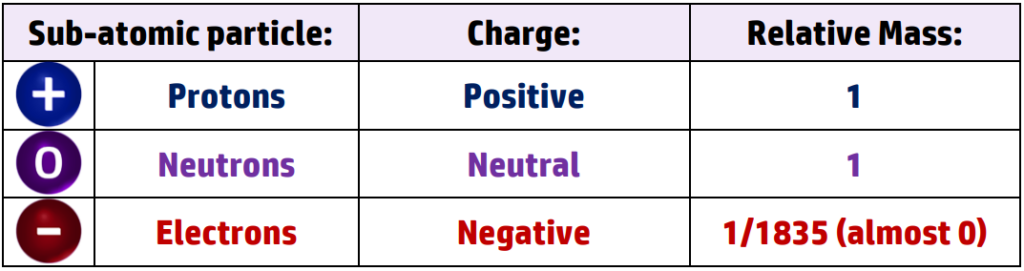
4.1.1.4 Relative electrical charges of subatomic particles
4.1.1.5 Size and mass of atoms
4.1.1.6 Relative atomic mass
4.1.1.7 Electronic structure
4.1.2.1 The periodic table
4.1.2.2 Development of the periodic table
4.1.2.3 Metals and non-metals
4.1.2.4 Group 0
4.1.2.5 Group 1
4.1.2.6 Group 7
4.1.3.1 Comparison with Group 1 elements (Separate Only!)
Atoms, Elements and Compounds
An atom is the smallest building block in the universe. All matter is made out of atoms.
When you have multiple atoms, you can have an element, a compound or a mixture.
Elements

An element is made up of all of the same atom, such as chlorine, Cl2, above.
Compounds

A compound is made up of more than one type of atom bonded together, such as water above.
Mixtures
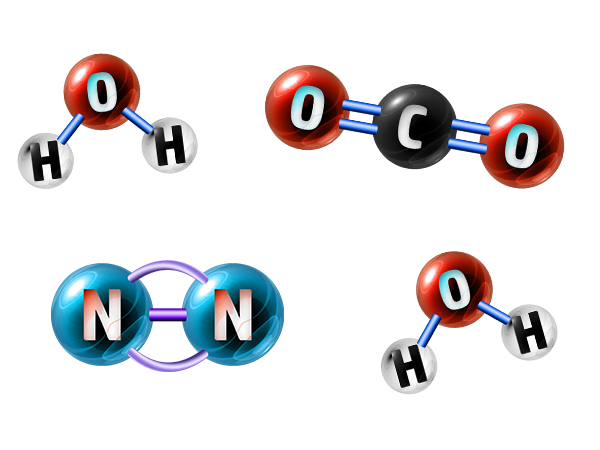
A mixture is made up of more than one element or compound not bonded together. Such as the mixture above which contains water, nitrogen and carbon dioxide.
CH27: Describe how ideas of the atom have changed
1. Dalton described atoms as indestructible spheres that could not be broken down.
2. Dalton suggested that atoms of an element were all identical.
3. JJ Thompson measured the mass of rays in a cathode tube. They were lighter than atoms – proving that the atom was not indestructible.
4. JJ Thompson proved that electrons existed – the plum pudding model – negative electrons = the plums, positive atom = the pudding.
5. Rutherford fired 20,000 alpha particles into gold foil. Most went through, some refracted and a few repelled. This proved the nucleus was positive and tiny.
6. Today’s atom, proposed by Niels Bohr, is known to contain protons and neutrons in the nucleus, with electrons orbiting shells.

CH28: Describe the structure of the atom.
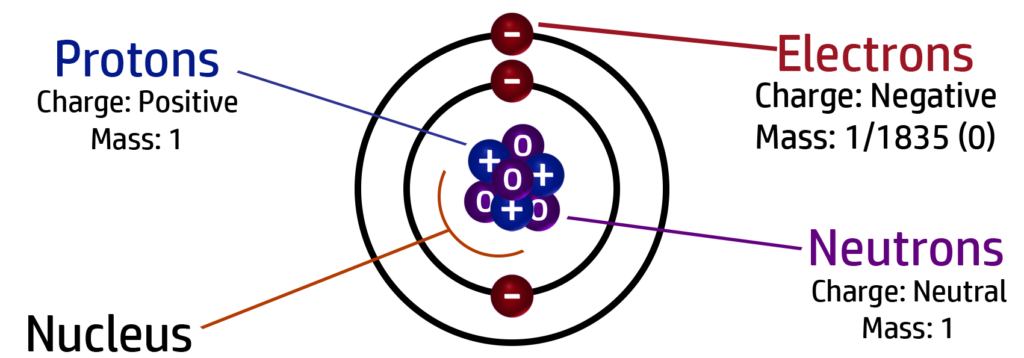
CH29: Identify the charge and mass of a proton, neutron and electron
CH31: Compare the size of the atom to the nucleus.
The nucleus is tiny.
Imagine a football field – this is the size of your atom.
Now imagine a marble on that football field. The marble is the size of the nucleus compared to the rest of the atom!

CH32: Identify where most of the mass of the atom is.
Most of the mass of an atom is found in the nucleus.
This is because the nucleus contains protons and neutrons, which both have a relative mass of 1.
Electrons have a relative mass of almost 0 (1/1835) so don’t add much mass to the atom.
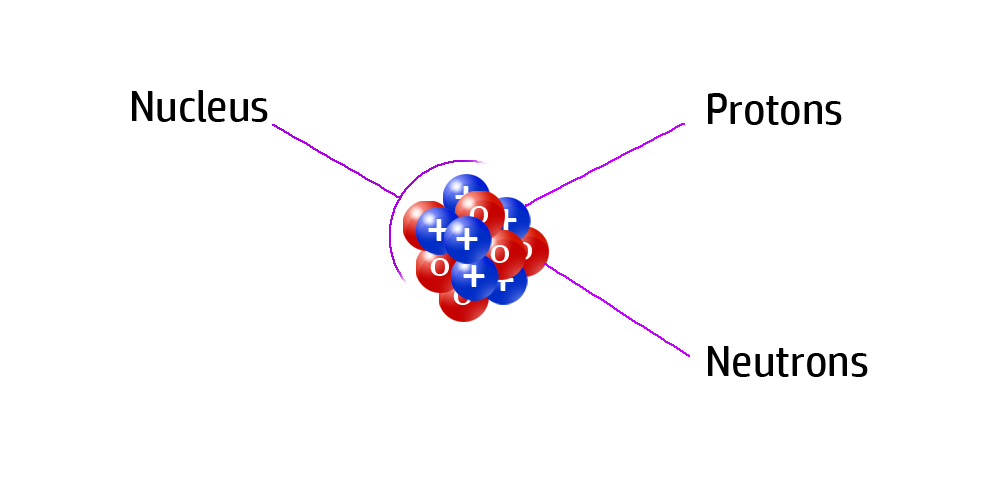
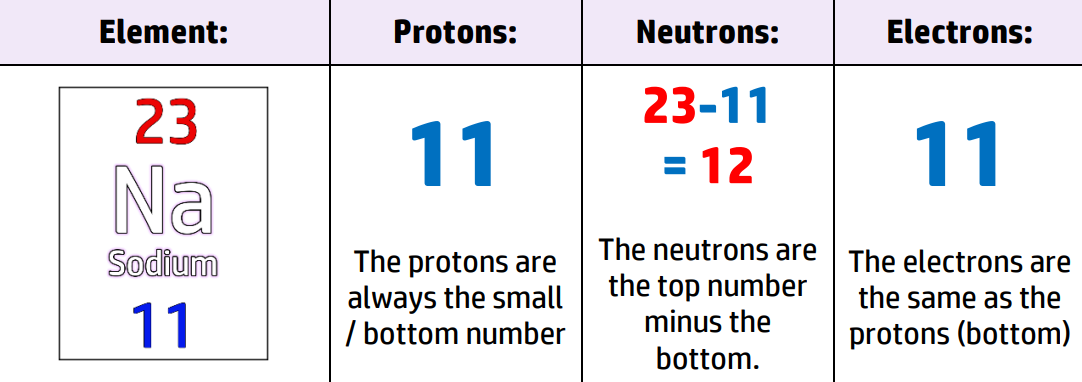
CH33: What does the mass number tell us?
The mass number is the total number of protons and neutrons inside the nucleus of an atom.
It is usually the largest number for each atom on the Periodic Table.
For example, the atom on the right has 3 positive protons and 4 neutral neutrons in the nucleus of its atom. Therefore, the mass number of this atom is 7.
CH34: Describe why the atomic number is unique.
The atomic number is the number of protons in an atom. It is also the smaller/bottom number in the Periodic Table.
This does not change (unlike neutrons and electrons). Therefore, if you have 11 protons, you always have an atom of Sodium, Na.

CH30: Explain why all atoms are neutral.
All atoms are neutral because they have the same number of protons and electrons. The charges cancel each other out giving an overall charge of 0.
For example: Lithium has 3 protons, 4 neutrons and 3 electrons:
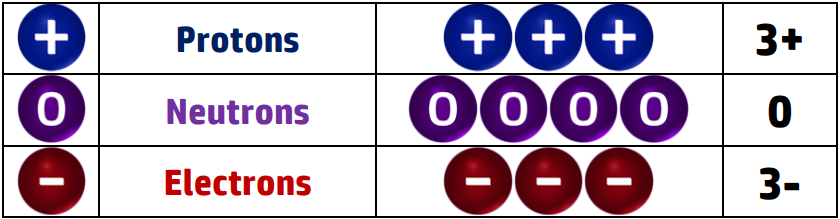
There are 3 x positive protons and 3 x negative electrons – +3-3 = 0 which is the overall charge. The neutrons don’t impact on the charge because they are neutral.
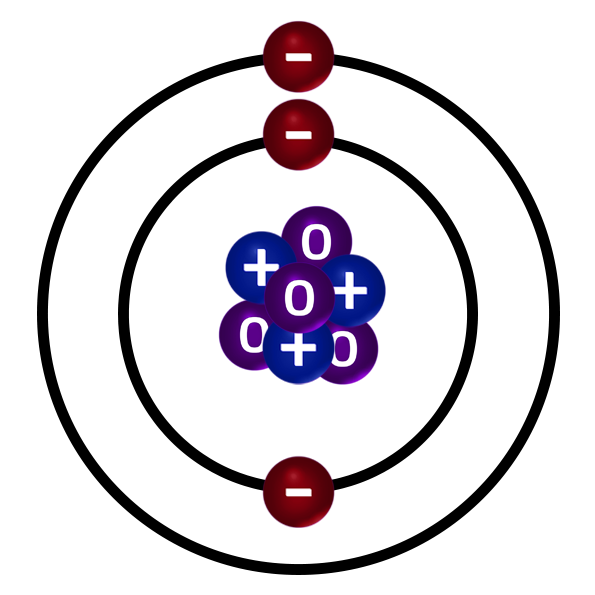
CH36: Describe how to draw the electronic configuration.

Find out the number of electrons. Aluminium has 13.
The first shell can hold 2e– and the 2nd/3rd can hold 8e–.
Fill up the shells from the inside out.
Aluminium can hold 2e– on the first shell, 8e– in the second and has 3e– left for the third shell.

CH37: Relate the electronic configuration to the periodic table
The number of electrons on the outer shell tells you the group the element is in.
Chlorine is in group 7 because it has 7 electrons in the outer shell.
The number of shells tells you the period the element is in.
Chlorine is in period 3 because it has 3 shells.

2.8.7
CH181: Identify the groups on the Periodic Table
CH71: Identify the properties of metals and non-metals
| Properties of Metals: | Properties of non-metals: |
| Good conductors of heat & electricity | Poor conductors of heat & electricity |
| Strong | Brittle (Break easily) |
| Malleable (Can be hammered into shape) | Low Melting Points & Boiling Points |
| Ductile (Can be stretched into wires) | Low Density |
| High Melting Points & Boiling Points |
CH40: Calculate the relative atomic mass of an isotope (H)
To calculate the relative atomic mass of an isotope you need to follow the following steps:

For example, if Chlorine has two isotopes – 35-Cl, with an abundance of 75% and 37-Cl with an abundance of 25%, the calculation will look like this:
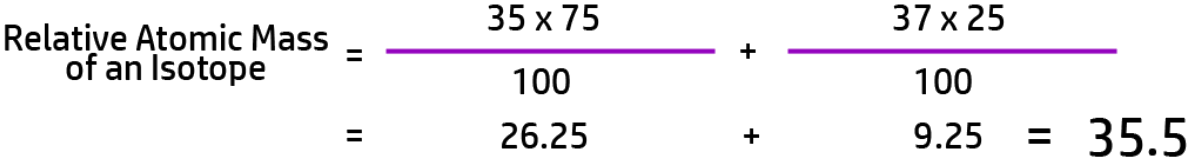
CH41: Estimate the abundance of isotopes (H)
Example: There are two isotopes of copper. 63-Cu and 65-Cu. The relative atomic mass is 63.5. Estimate the abundance of each isotope.
- Step 1: Work out the difference between the masses of each isotope. 65-63= 2.
- Step 2: Work out the difference between the isotope mass and each isotope:
- 63.5-63 = 0.5
- 65-63.5 = 1.5
- Step 3: Work out the percentage for each isotope by dividing these differences (step 2) by the total difference (step 1)
- 0.5 / 2 x 100 = 25%
- 1.5 / 2 x 100 = 75%
- Step 4: The isotope with the mass closest to the relative atomic mass is the largest abundance. So, 63-Cu is 75% abundant and 65-Cu is 25% abundant.
CH42: Describe how Mendeleev arranged his Periodic Table
Dmitri Mendeleev was a Russian Scientist who, in 1869, decided to put the elements know at the time into a Table of Elements.
He put the elements into groups based on their chemical properties – which is still the same today. He also arranged the elements in order of atomic mass. We now know that this was wrong and today’s Periodic Table is in order of atomic number.

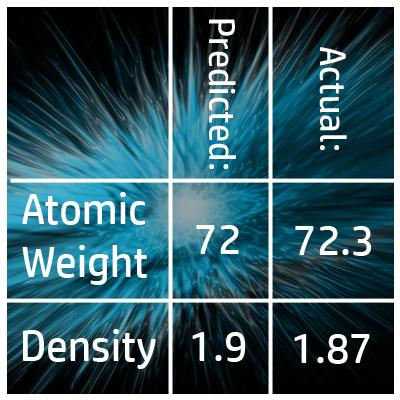
CH43: Explain how Mendeleev predicted the properties of undiscovered elements
If an element hadn’t been discovered yet, Mendeleev left gaps and used the properties from the elements in the rest of the group to predict the properties of the missing elements.
For example, he predicted the atomic weight of Germanium was 72 and the density 1.9g/dm3. Many years later, when these elements were discovered, he was almost spot on!
CH44: Explain what Mendeleev did when properties didn’t match
Because Dmitri Mendeleev’s table of the Elements was in order of atomic mass (but should have been in order of atomic number), some of the properties didn’t match up to the properties of the group.
He ended up swapping these elements so that the chemical properties matched. An example of this is iodine (group 7) and tellurium (group 6). Originally, iodine was in group 6 and tellurium was in group 7, but iodine has similar chemical properties to the rest of the halogens (group 7) – so Mendeleev swapped them.
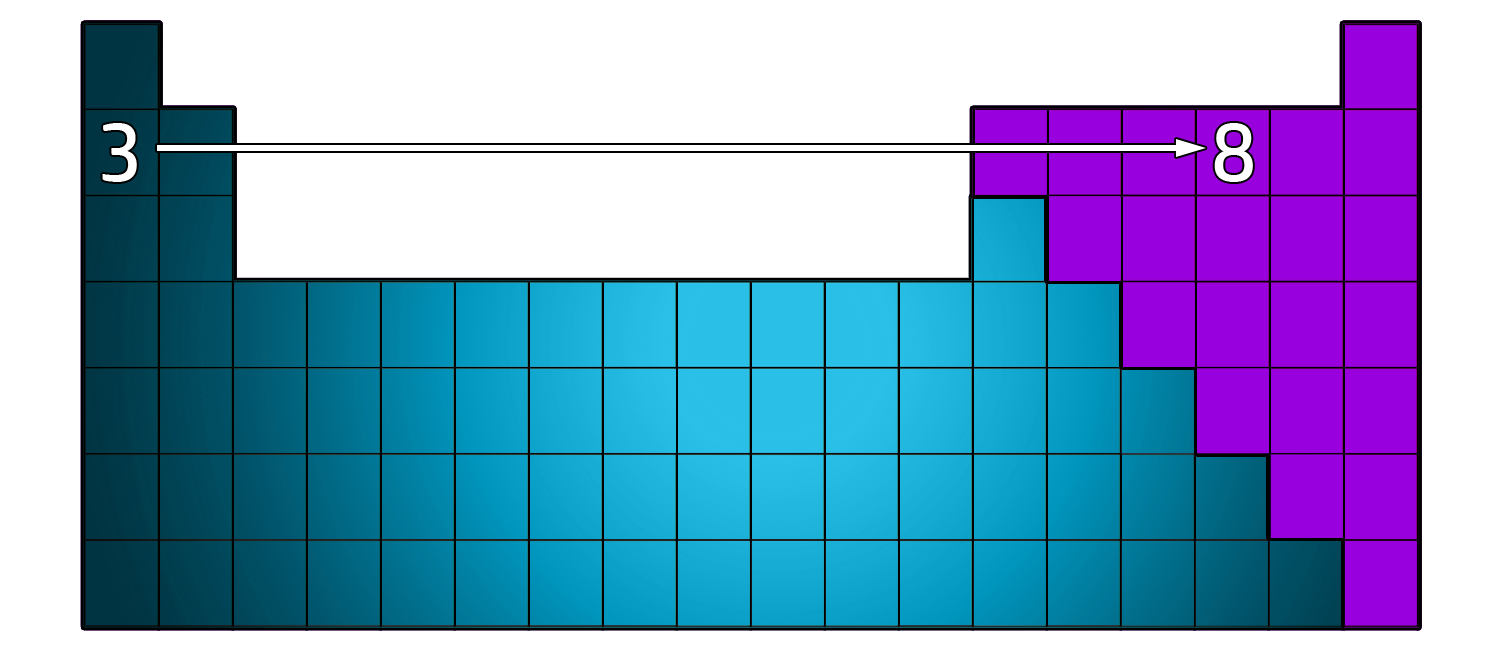
CH45: Describe how the atomic number is linked to the Periodic Table
The atomic number tells you where the atom can be found on the Periodic Table.
Starting off with Hydrogen (Period 1), the atomic number increases by one each time you go from left to right.
So, if you have an atomic number of 3 (Lithium) and move 5 places to the right, you will have an atomic number of 8 (oxygen)
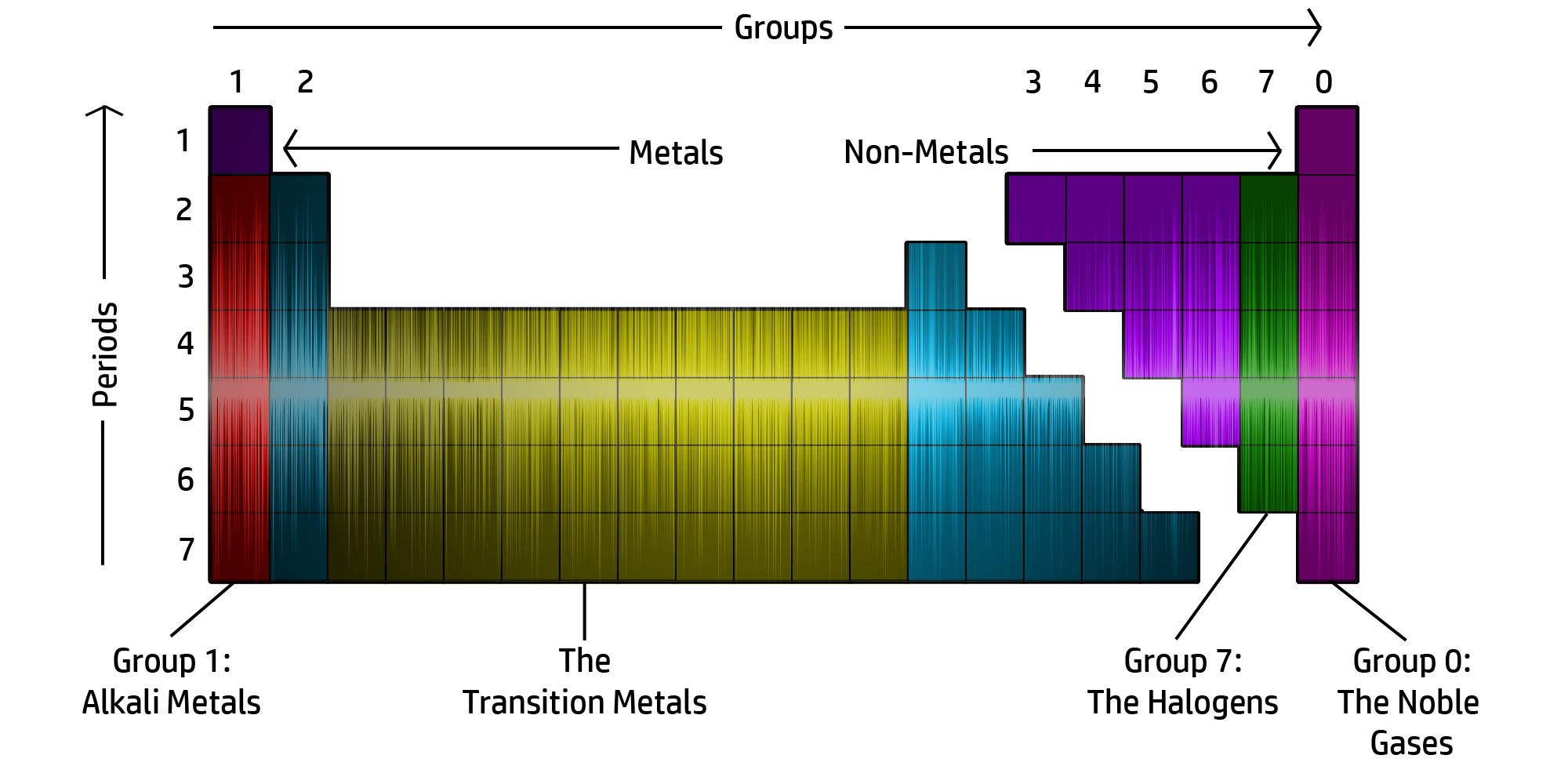
CH46: Describe the structure of the Periodic Table
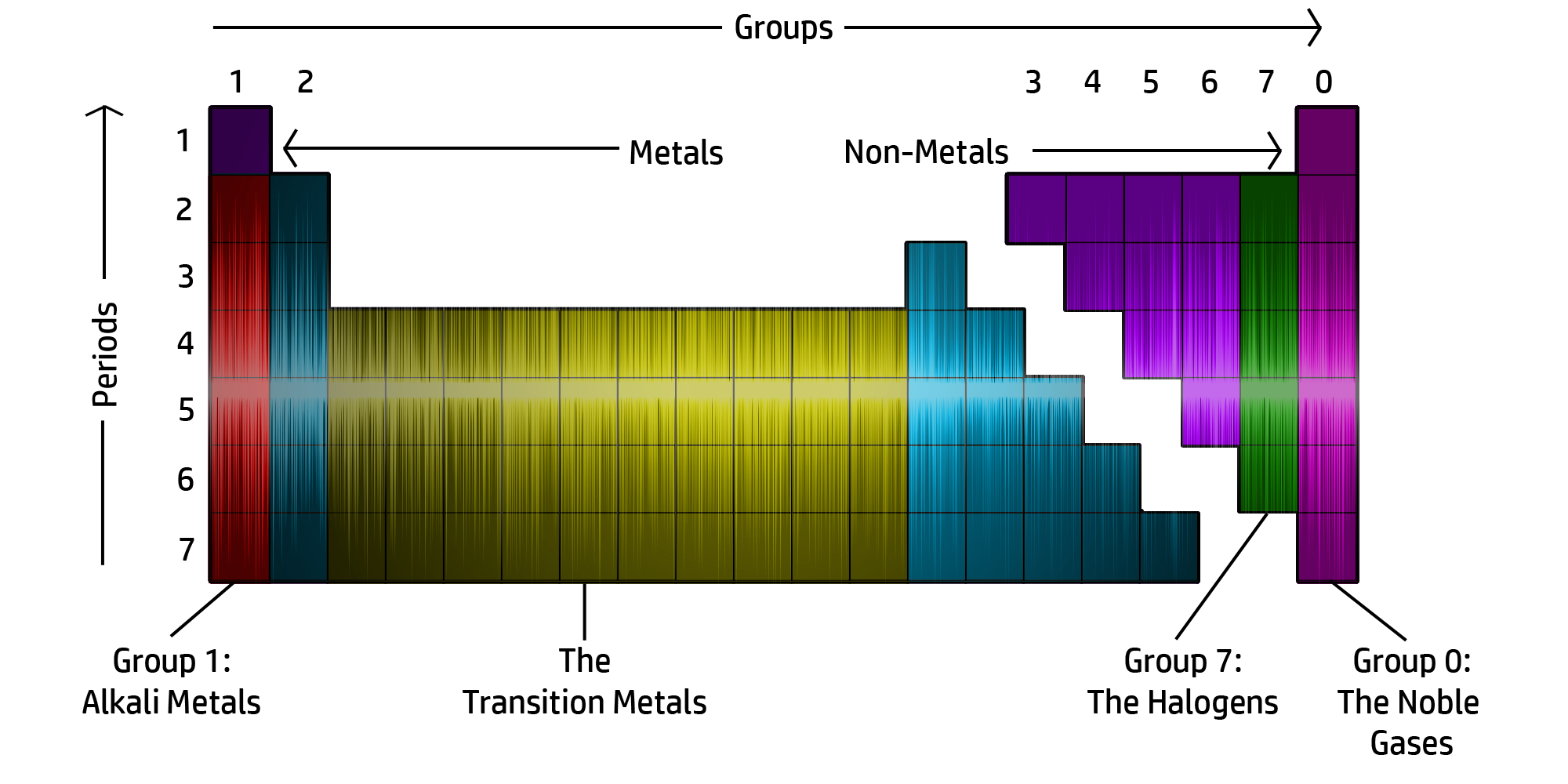
The Periodic table is now arranged in groups based on chemical properties.
The groups can be seen in the diagram above.
CH47: Identify where metals / non-metals are found on the Periodic Table
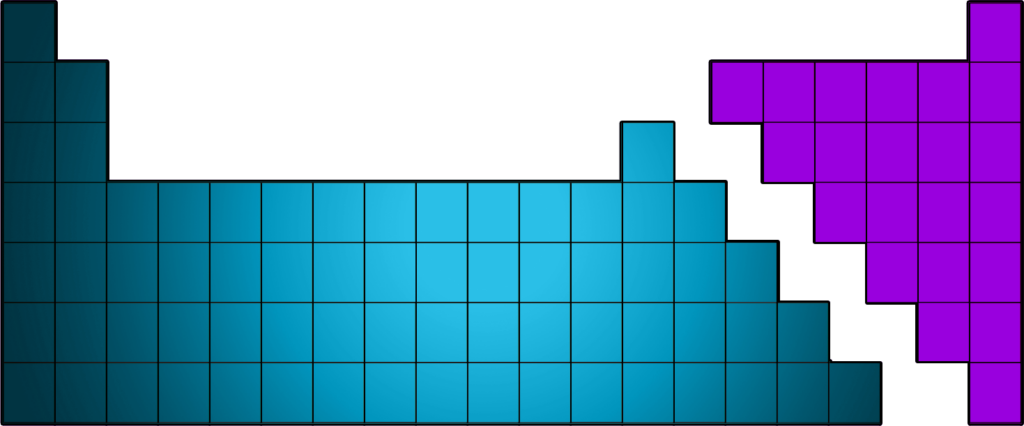
- Metals are found on the left of the zigzag (with the exception of hydrogen, which is a non-metal.
- Non-Metals are found on the right of the zigzag.
The zigzag starts between Boron and Aluminium and goes right and down like a staircase!
CH78: Calculate Relative Formula Mass
The relative formula mass is simply the atomic masses of all the atoms in a compound added together. You can find the atomic mass in the question – or the big number on the Periodic Table.
Example 1: Chloromethane has the chemical formula of CH3Cl. Calculate the relative formula mass. (Ar: C = 12, H = 1, Cl = 35.5)
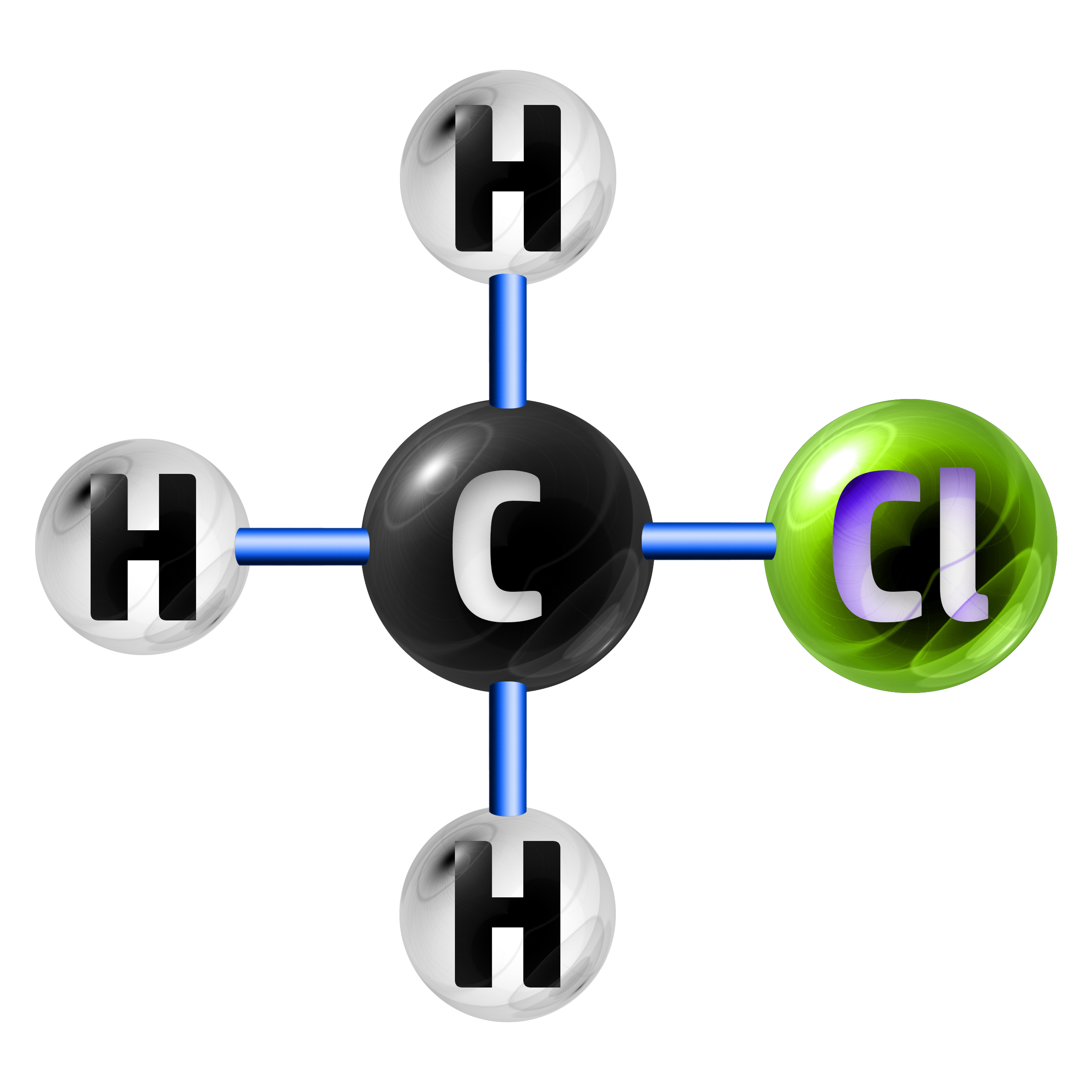
The formula CH3Cl shows me that I have 1 x C (carbon), 3 x H (hydrogen) and 1 x Cl (chlorine).
- 1 x C = 1 x 12 = 12
- 3 x H = 3 x 1 = 3
- 1 x Cl = 1 x 35.5 = 35.5
Relative Formula Mass = 12 + 3 + 35.5 = 50.5
Example 2: Magnesium nitrate has the chemical formula of Mg(NO3)2. Calculate the relative formula mass. (Ar: Mg = 24, N = 14, O = 16)
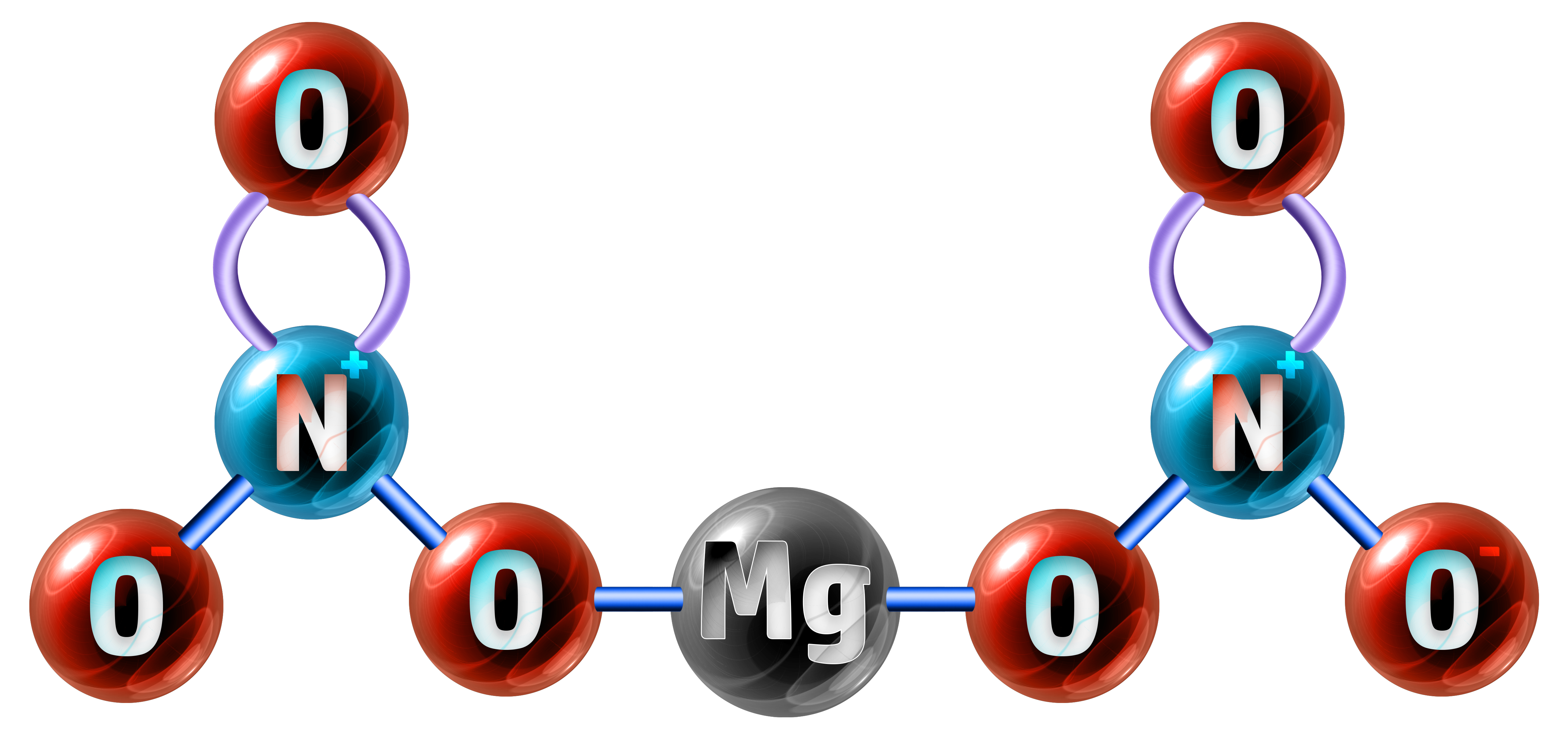
The formula Mg(NO3)2 shows me that I have 1 x Magnesium atom (Mg) connected to two lots of my nitrate ion (NO3), so I have 1×2 nitrogen’s and 3×2 oxygen’s:
- 1 x Mg = 1 x 24 = 24
- 2 x N = 2 x 14 = 28
- 6 x O = 6 x 16 = 96
Relative Formula Mass = 24 + 28 + 96 = 148
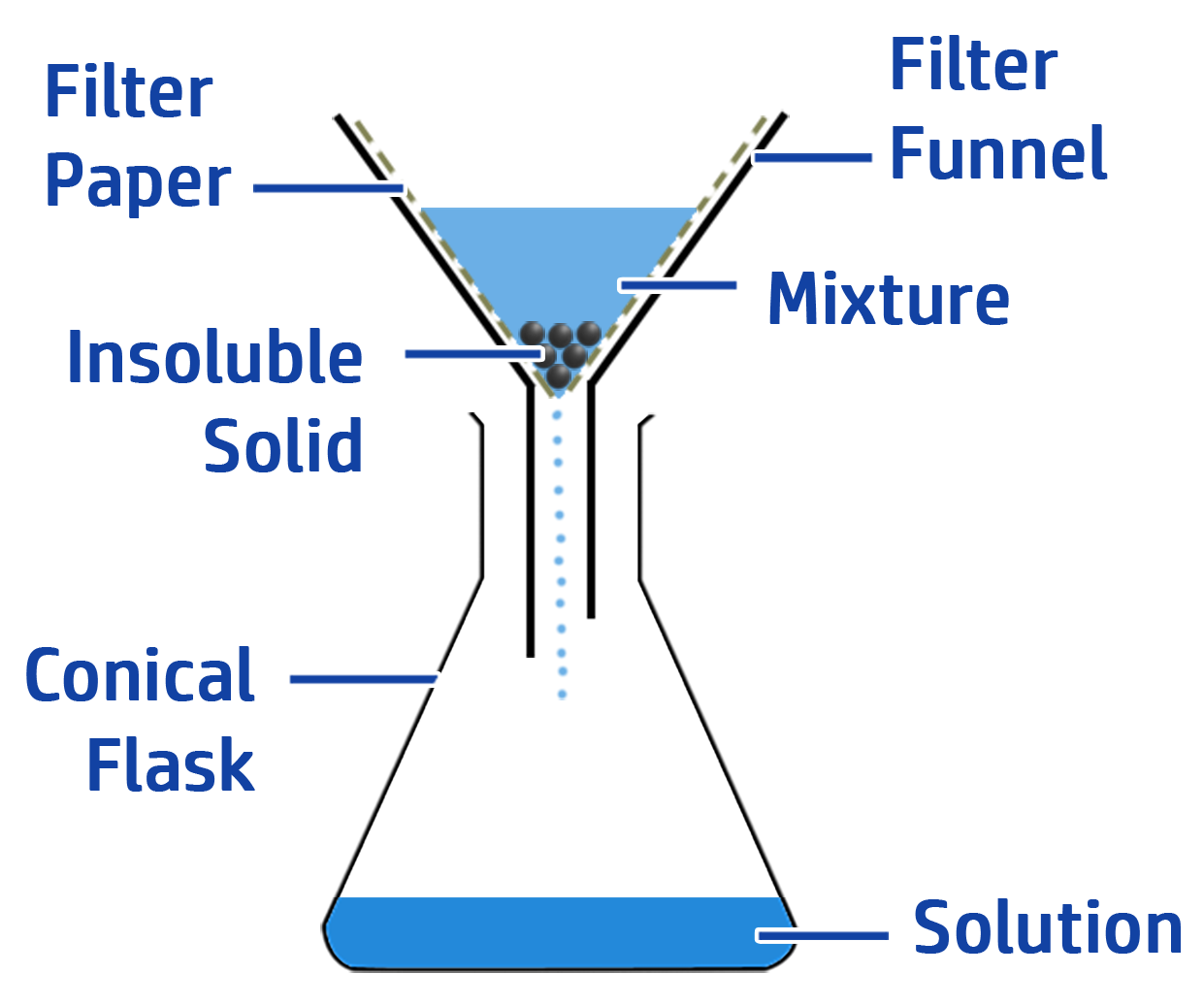
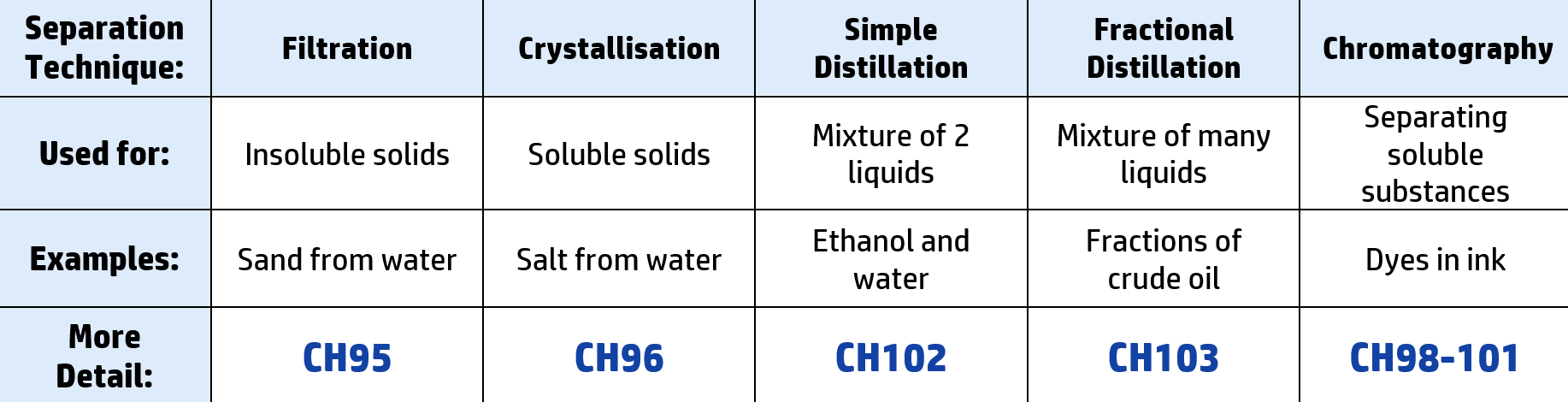
CH95: Describe how to carry out filtration
An insoluble solid is a solid that will not dissolve – such as rock/sand/copper oxide.
To separate an insoluble solid from a mixture:
- Step 1: Add water to dissolve anything that is soluble. The insoluble solid will remain as a solid.
- Step 2: Add the mixture to a filter funnel with filter paper in it.
The soluble solids and water will move through the filter paper (because their particles are small enough to fit through the holes in the filter paper). The insoluble particles will be too big, so will remain in the filter paper.
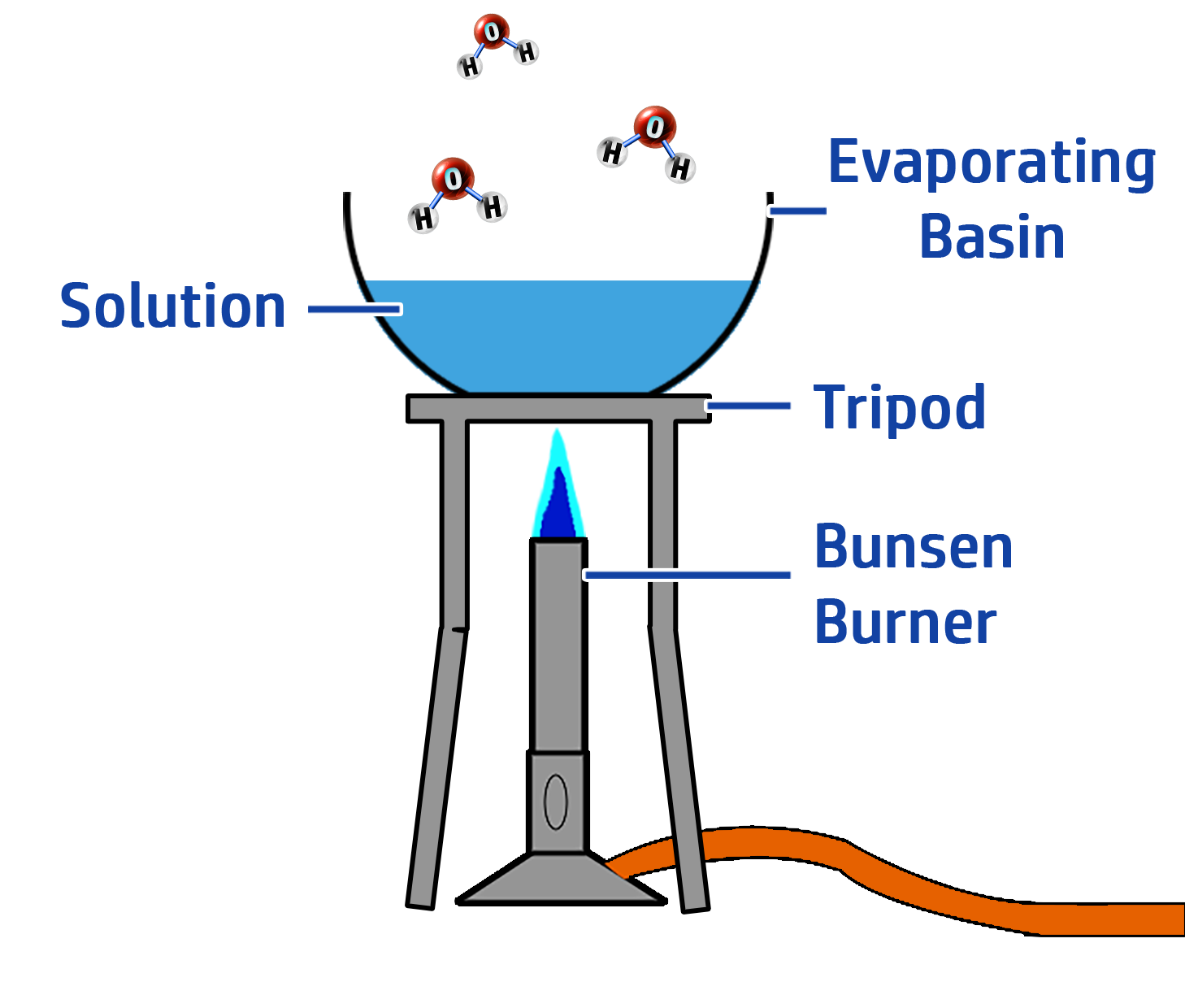
CH96: Describe how to carry out crystallisation
A soluble solid is a solid that will dissolve when added water, or other solvents. An example of a soluble salt is table salt (sodium chloride)
To separate a soluble solid from a mixture:
- Step 1: Heat the solution gently using a Bunsen Burner (see right)
- Step 2: Evaporate half of the solution
- Step 3: Leave the solution to cool.
- Step 4: Leave the crystals to dry on a windowsill.
CH97: Describe how to separate soluble and insoluble solids
If you have a mixture of a soluble and insoluble solid, you need to carry out filtration and crystallisation.
- Step 1: Dissolve the mixture with water.
- Step 2: Add the mixture to a filter funnel and filter paper. This will separate the insoluble solid from the rest of the mixture.
- Step 3: Add the solution (filtrate) to an evaporating basin and heat the solution until half of the solution has evaporated.
- Step 4: Leave to cool/evaporate – leaving crystals behind.
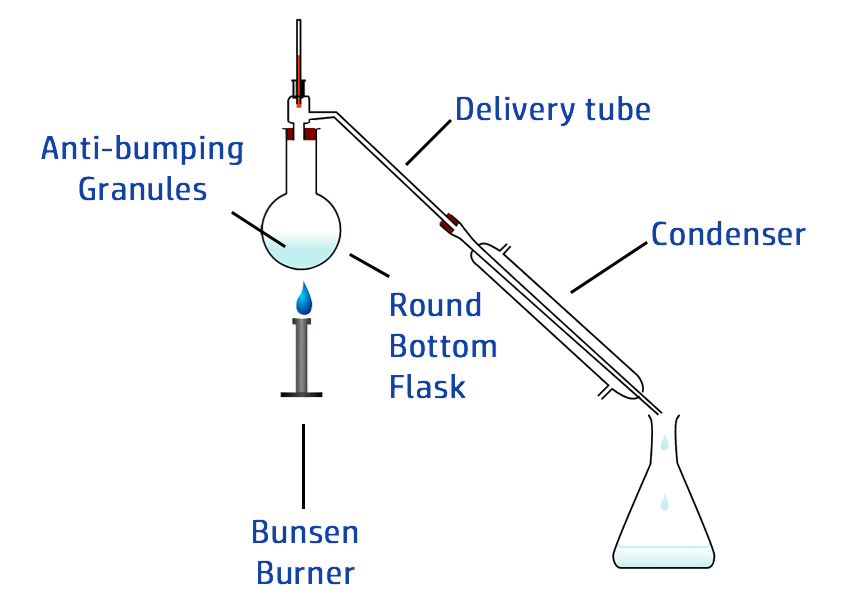
CH102: Describe how to carry out simple distillation
Simple distillation is a technique used to separate two different liquids based on their boiling points.
Example: Separating water (boils at 100oC) and ethanol (boils at 78oC).
- Heat the solution in a round bottom flask.
- Once it reaches 78oC, the ethanol will evaporate.
- The gas will rise and move into the condenser.
- The condenser is surrounded by cold water, which cools the gas down – turning it back into a liquid.
- If you stay below 100oC, the water will not evaporate.
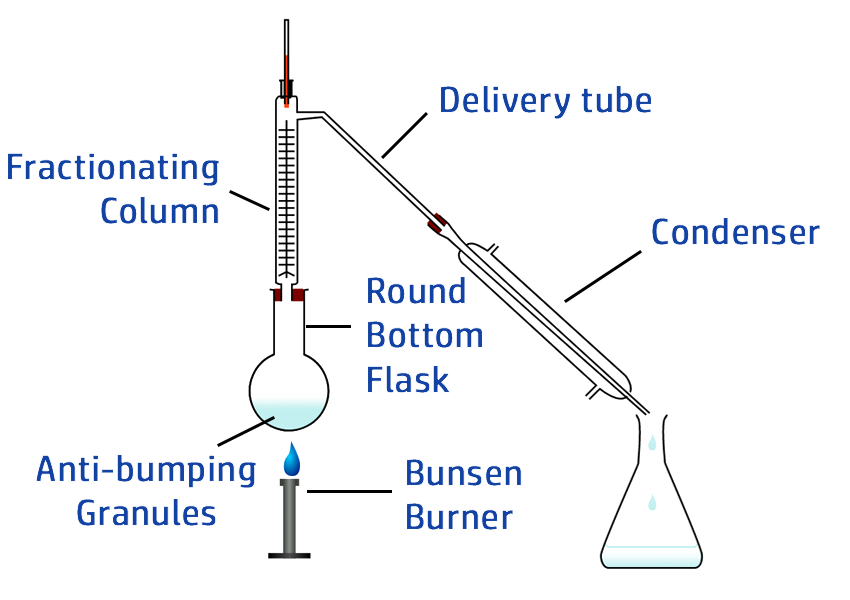
CH103: Describe how to carry out fractional distillation
Fractional distillation is used to separate a mixture that contains lots of different liquids.
As the mixture is heated, a temperature gradient will exist – where it is hotter at the bottom of the fractionating column and cooler at the top.
Once the top of the column reaches the lowest boiling point – the first gas will move into the condenser and can be collected.
As the column continues to warm up, more and more of the fractions will move into the condenser, allowing them all to be collected separately.
CH104: Suggest a suitable method to separate mixtures
CH182: Describe the physical properties of the alkali metals
Alkali metals have similar physical properties to most metals (CH71), but there are some differences:
- They have much lower melting points than most metals.
- They are very soft – can be cut easily.
- They have low densities (lithium, sodium and potassium can float on water)
CH183: Write word and chemical equations for reactions between alkali metals and water
When alkali metals are added to water, they all react the same way. They all react with water to produce a hydroxide and hydrogen gas.
The word and balanced chemical equations are identical:
Potassium + Water → Potassium Hydroxide + Hydrogen
2K (s) + H2O (l) → 2KOH (aq) + H2 (g)
You can write word and balanced chemical equations easily for the alkali metals and water by just replacing the name/symbol for potassium above with the name/symbol of the alkali metal. For more information on alkali metal word equations see CH4 and for balanced equations check CH13.
CH184: Describe the similarities and differences when the alkali metals are added to water

When the alkali metals are added to water, you need to remember the similarities:
- They all float on water.
- They all move on water.
- They all fizz.
As you go down the group (Lithium → Sodium → Potassium), the reactivity increases. We can see this because:
- Lithium only moves and fizzes a small amount.
- Sodium fizzes more, moves slightly faster than lithium, and turns into a molten ball.
- Potassium fizzes the most, sets on fire with a lilac flame, and can explode.
You can use this to predict the properties of the rest of the alkali metals – as you go down, the move more, fizz more and will most likely explode!
CH185: Explain why reactivity increases as you go down group one
As you go down group 1, reactivity increases. This is all to do with electronic configuration:
- As you go down the group, the atom has more shells of electrons.
- There is more electron shielding.
- The outer electron is further away from the nucleus.
- The force of attraction between the outer electron and the positive nucleus is weaker.
- It is easier to lose the outer electron – therefore more reactive!

CH186: Explain why the noble gases are inert

The Noble Gases are found in group 0 of the Periodic table and contain helium, neon, argon, krypton, xenon, and radon.
They are in group 0 because they all have full outer shells.
This makes all noble gases inert.
This means that they are unreactive – they don’t need to lose or gain any electrons to get a full outer shell.
They are all monatomic – which means they go around on their own.
CH187: Describe the uses and properties of the noble gases

Argon, Xenon and Krypton can be used in lightbulbs.
Argon is used in filament light bulbs
Krypton and Xenon are used in flash photography.
Explanation: Argon has a full outer shell, so doesn’t want to lose / gain / share any electrons. Therefore, it will not react with the hot filament (unlike oxygen), stopping it from breaking.
Argon is also used in welding.
Explanation: Argon is denser than air, so it stops oxygen from getting to the metal. It has a full outer shell, so doesn’t want to lose / gain / share any electrons. Therefore, it will not react with the metal (unlike oxygen), stopping it from getting impurities.


Argon is also used in wine barrels
Explanation: Argon is placed in the top of wine barrels, replacing oxygen from the air. It has a full outer shell, so doesn’t want to lose / gain / share any electrons. Therefore, it will not react with the wine (unlike oxygen), stopping it from oxidising and turning into a carboxylic acid.
Helium is used in airships, weather balloons and party balloons
Explanation: Helium is less dense than air, so will float. It is used instead of hydrogen because it has a full outer shell, so doesn’t want to lose / gain / share any electrons. Therefore, it will not set on fire (unlike hydrogen), stopping it from exploding

CH188: Describe the trend in physical properties for the noble gases
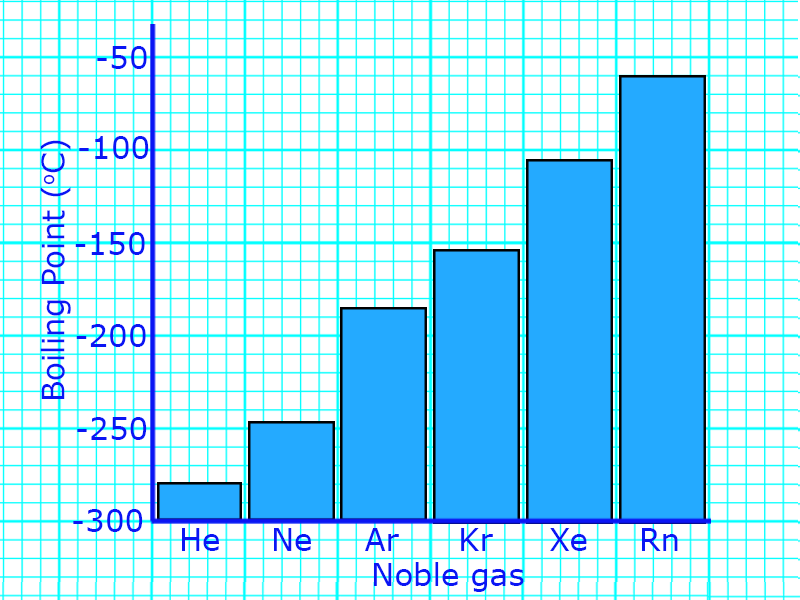
The noble gases all seem similar, but the physical properties do change as you go down the group:
- Boiling point and Melting point increases down the group
- Density increases as you go down the group
You can use the information about these densities to predict the properties of different noble gases.
CH189: Identify the state and colour of the halogens
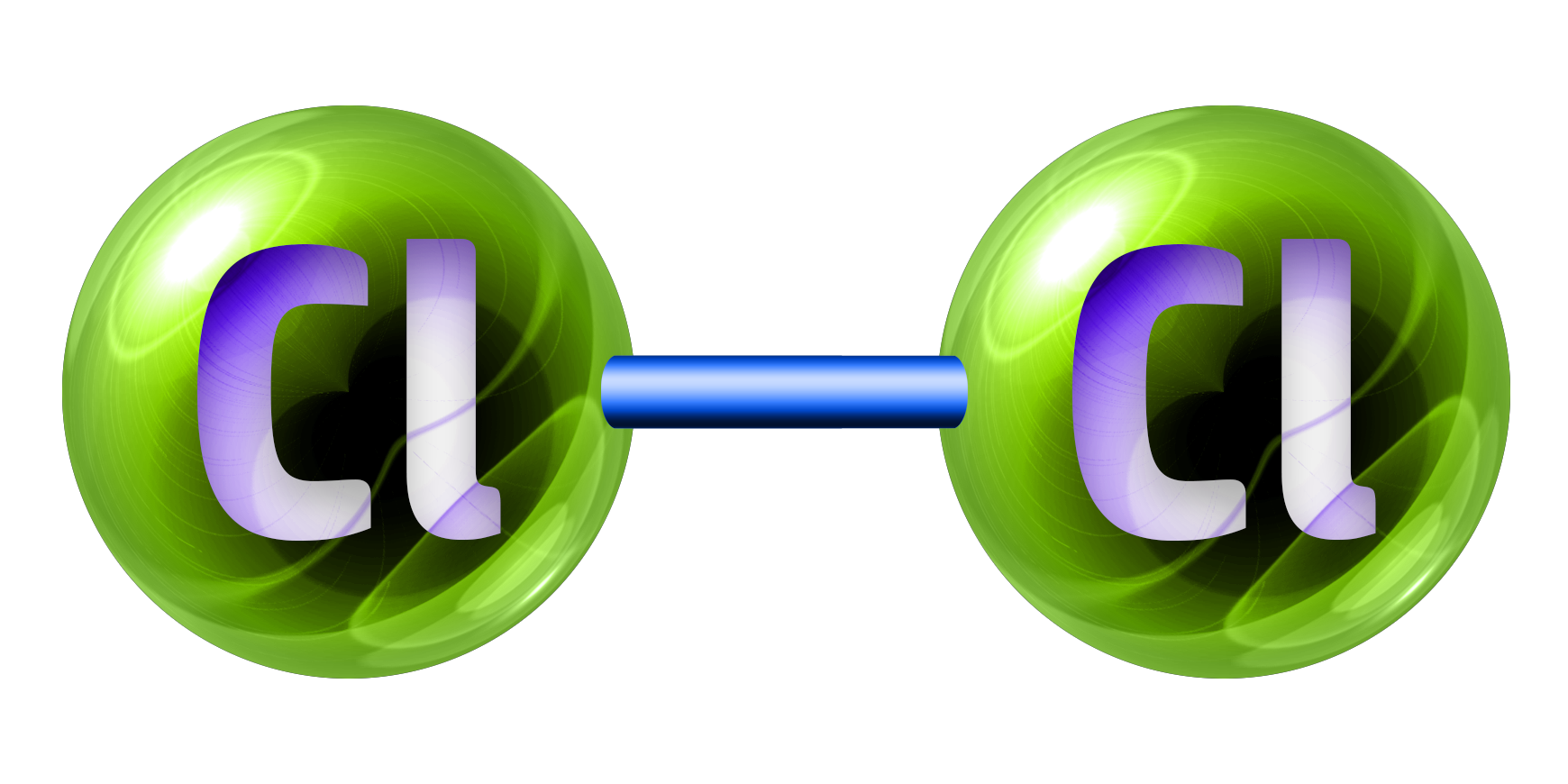
Chlorine, Cl2, is a green gas.
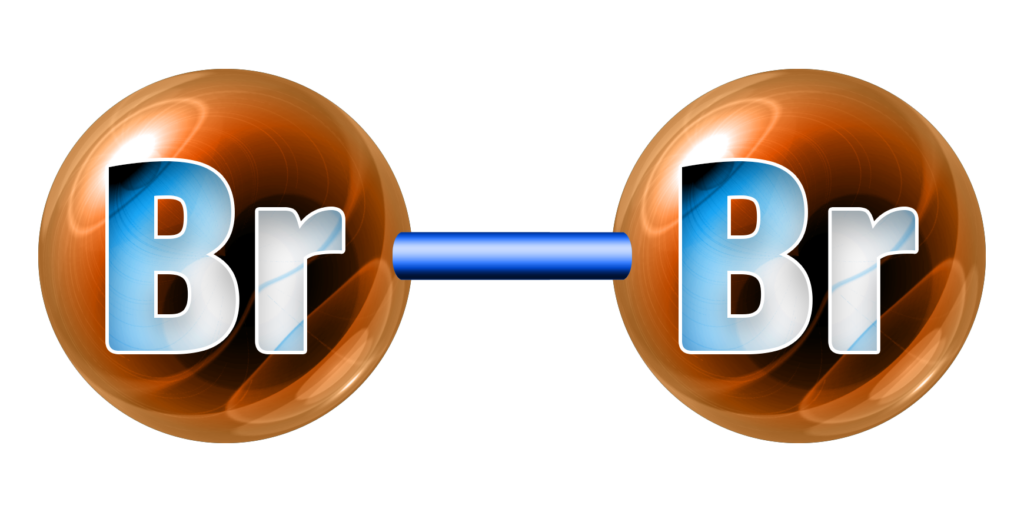
Bromine, Br2, is a red-brown liquid.
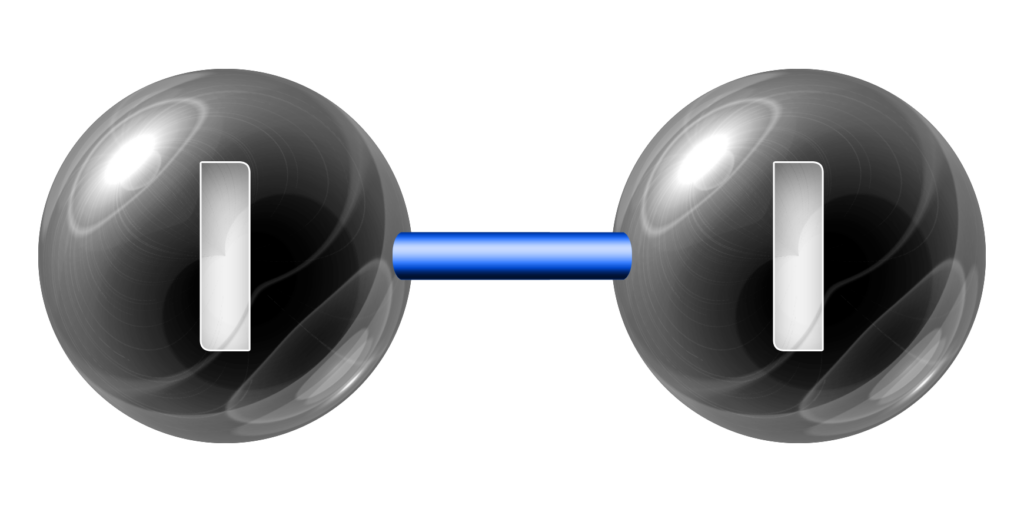
Iodine, I2, is a black solid.
CH190: Describe the trend in properties of the halogens
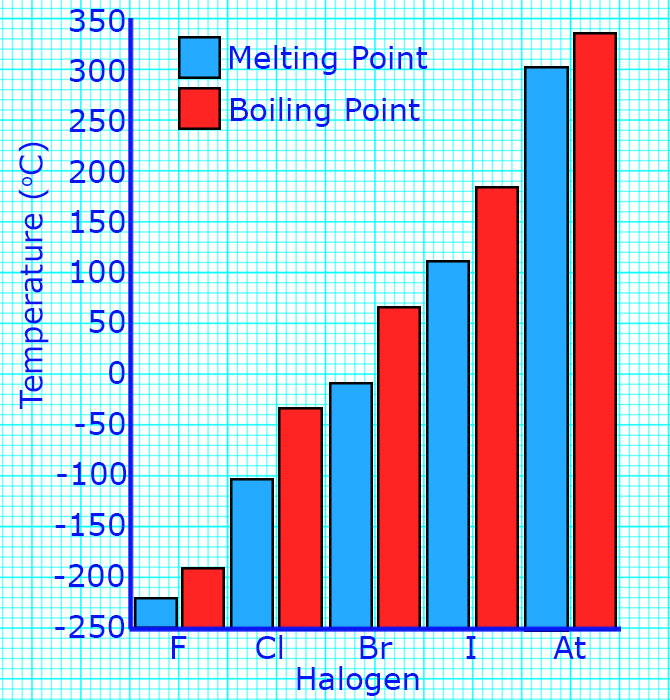
As you go down group 7, the melting points and boiling points increase.
You can tell this because the states change from gases to solids as you go down the group.
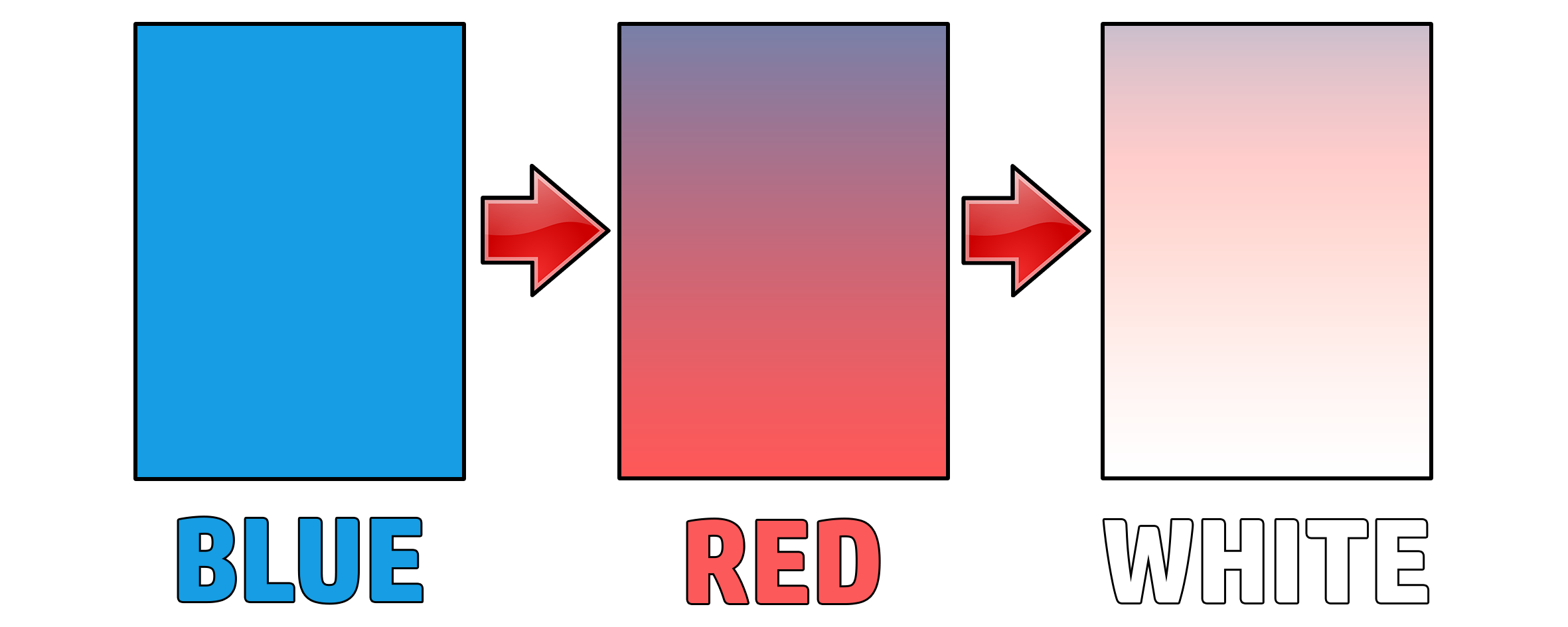
CH191: Describe the test for chlorine
If you wanted to prove that a gas was chlorine, add damp blue litmus paper to it.
Damp blue litmus paper turns red, then bleaches.
CH192: Describe the reactions of the halogens with metals
When the halogens react with metals, they form metal halides. The reactions are all similar, depending on what group you are reacting with:
- Group 1 metals give the general formula of M1H1 – one metal and one halogen.
- Group 2 metals give the general formula of M1H2 – one metal and two halogens.
- Group 3 metals give the general formula of M1H3 – one metal and three halogens.
Group 1 Example: Write the word and balanced equation for the reaction between sodium and iodine.
Sodium + Iodine → Sodium Iodide
2Na (s) + I2 (s) → 2NaI (s)
Group 2 Example: Write the word and balanced equation for the reaction between magnesium and bromine.
Magnesium + Bromine → Magnesium Bromide
Mg (s) + Br2 (l) → MgBr2 (s)
Group 3 Example: Write the word and balanced equation for the reaction between aluminium and chlorine.
Aluminium + Chlorine → Aluminium Chloride
2Al (s) + Cl2 (g) → 2AlCl3 (s)
CH193: Describe the reactions of the halogens with hydrogen
When the halogens react with hydrogen, the same reaction occurs – hydrogen halides are formed.
Example: Write the word and balanced equation for the reaction between hydrogen and chlorine.
Hydrogen + Chlorine → Hydrogen Chloride
H2 (g) + Cl2 (g) → 2HCl (g)
All the hydrogen halides are gases, and when they dissolve into water, they form an aqueous solution which is acidic.
- Hydrogen chloride, HCl (g), dissolves to form hydrochloric acid, HCl (aq).
- The hydrogen ion, H+, is what makes the solution acidic.
CH194: Investigate the reactivity of the halogens
As you go down group 7, reactivity decreases. This can be proved using displacement reactions – where the more reactive halogen will always end up in the salt.
If you take a salt containing chlorine, bromine and iodine, and react them with solutions of their elements (Cl2, Br2 and I2), the following will occur:
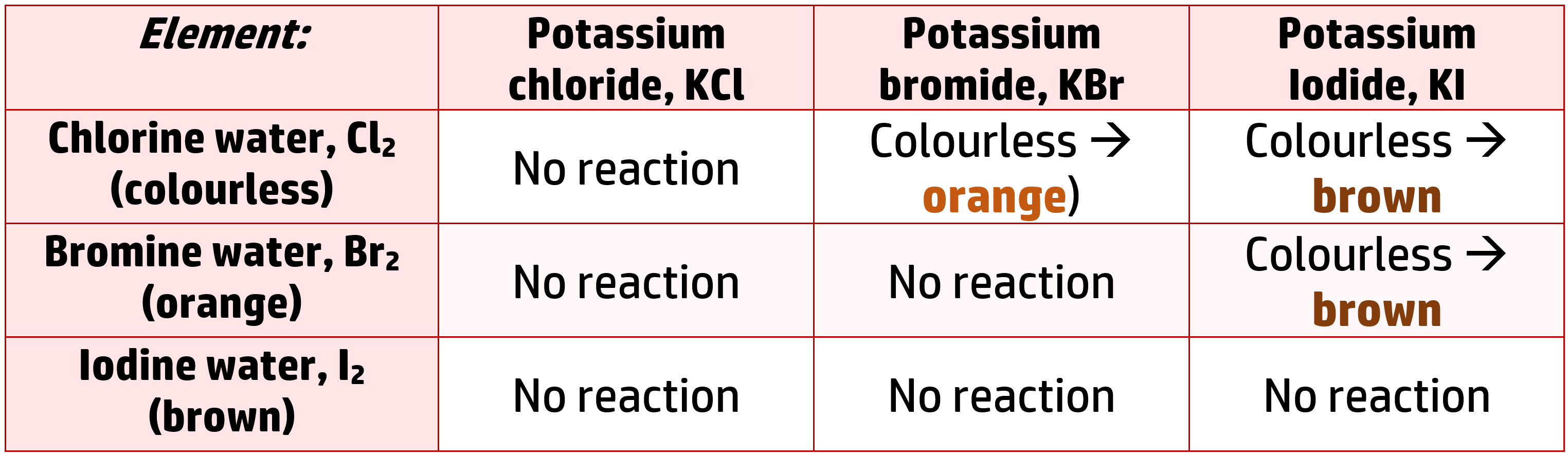
This shows us that chlorine is more reactive than both bromine and iodine because it displaces them both to form potassium chloride.
It also shows that bromine is more reactive than iodine because it displaces it (you see a reaction), but it is less reactive than chlorine (no reaction occurs)

Example of a displacement reaction:
Chlorine + Potassium Bromide → Potassium Chloride + Bromine
Cl2 + 2KBr → 2KCl + Br2
CH195: Link displacement reactions of the halogens to REDOX reactions
The displacement reactions seen in CH194 are all examples of REDOX reactions.
In the above reaction, where chlorine displaces the bromine in potassium bromide, the following reaction occurs:
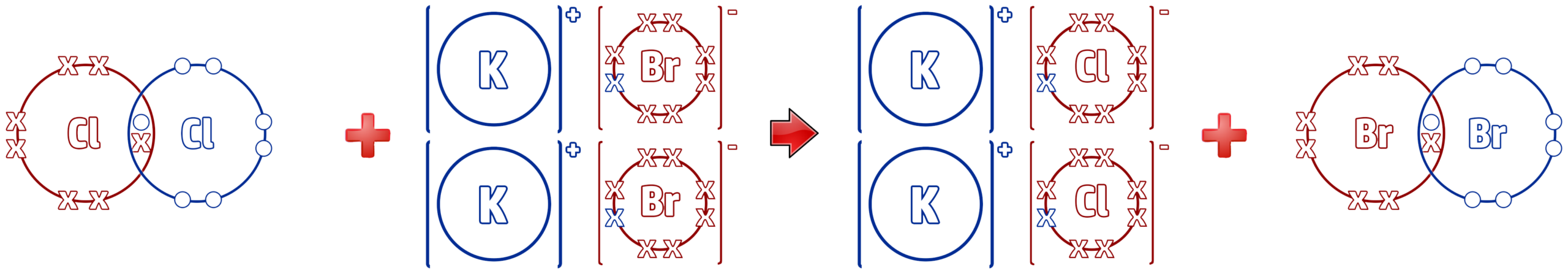
As you can see from the dot and cross diagram above, the chlorine molecule has 14 electrons on the left and the chlorine ions have 16 electrons on the right.
This means that chlorine has gained electrons – RIG – Reduction Is Gain. Chlorine is reduced.
The opposite happens to bromine – OIL – Oxidation Is Loss of electrons. The halogen (e.g. Cl2) always gains electrons (reduced) and the ionic halide (e.g. 2KBr) is always oxidised, as they lose electrons.
CH196: Explain why reactivity of the halogens decreases as you go down group 7

As you go down the group, the atom has more shells of electrons.
There is more electron shielding.
The outer electron is further away from the nucleus.
The force of attraction between the outer electron and the positive nucleus is weaker.
It is harder to gain the outer electron – therefore less reactive!
CH159: Identify the properties of the transition metals (SEPARATE ONLY)
Physical Properties:
- Malleable (can be hammered into shape)
- Ductile (can be stretched into wires)
- Good conductors of electricity and heat
- Shiny when polished.
Transition metals usually have higher melting points and densities than metals from groups 1 and 2.
There are exceptions though – mercury is a liquid at room temperature!
Chemical Properties:
One of the main chemical properties that makes transition metals different to those in groups 1 and 2 is that they form colourful compounds – such as iron (II) oxide, which is a red/brown colour.
Transition metals are used as catalysts (which speed up a chemical reaction without being changed chemically)
- Iron is the catalyst used in the Haber Process.
- Iron (III) chloride, FeCl3, is used during the manufacture of PVC
{“type”:”default”}} –>
The nucleus is tiny.
Imagine a football field – this is the size of your atom.
Now imagine a marble on that football field. The marble is the size of the nucleus compared to the rest of the atom!